Abstract
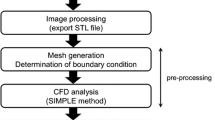
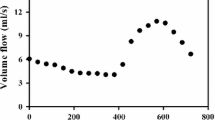
Modeling of flow in intracranial aneurysms (IAs) requires flow information at the model boundaries. In absence of patient-specific measurements, typical or modeled boundary conditions (BCs) are often used. This study investigates the effects of modeled versus patient-specific BCs on modeled hemodynamics within IAs. Computational fluid dynamics (CFD) models of five IAs were reconstructed from three-dimensional rotational angiography (3DRA). BCs were applied using in turn patient-specific phase-contrast-MR (pc-MR) measurements, a 1D-circulation model, and a physiologically coherent method based on local WSS at inlets. The Navier–Stokes equations were solved using the Ansys®-CFX™ software. Wall shear stress (WSS), oscillatory shear index (OSI), and other hemodynamic indices were computed. Differences in the values obtained with the three methods were analyzed using boxplot diagrams. Qualitative similarities were observed in the flow fields obtained with the three approaches. The quantitative comparison showed smaller discrepancies between pc-MR and 1D-model data, than those observed between pc-MR and WSS-scaled data. Discrepancies were reduced when indices were normalized to mean hemodynamic aneurysmal data. The strong similarities observed for the three BCs models suggest that vessel and aneurysm geometry have the strongest influence on aneurysmal hemodynamics. In absence of patient-specific BCs, a distributed circulation model may represent the best option when CFD is used for large cohort studies.







Similar content being viewed by others
References
Boussel, L., V. Rayz, et al. Aneurysm growth occurs at region of low wall shear stress: patient-specific correlation of hemodynamics and growth in a longitudinal study. Stroke 39(11):2997–3002, 2008.
Castro, M. A., C. M. Putman, et al. Computational fluid dynamics modeling of intracranial aneurysms: effects of parent artery segmentation on intra-aneurysmal hemodynamics. AJNR Am. J. Neuroradiol. 27(8):1703–1709, 2006.
Castro, M. A., C. M. Putman, et al. Patient-specific computational fluid dynamics modeling of anterior communicating artery aneurysms: a study of the sensitivity of intra-aneurysmal flow patterns to flow conditions in the carotid arteries. AJNR Am. J. Neuroradiol. 27(10):2061–2068, 2006.
Castro, M. A., C. M. Putman, et al. Patient-specific computational modeling of cerebral aneurysms with multiple avenues of flow from 3D rotational angiography images. Acad. Radiol. 13(7):811–821, 2006.
Cebral, J. R., M. A. Castro, et al. Efficient pipeline for image-based patient-specific analysis of cerebral aneurysm hemodynamics: technique and sensitivity. IEEE Trans. Med. Imaging 24(4):457–467, 2005.
Cebral, J. R., M. A. Castro, et al. Characterization of cerebral aneurysms for assessing risk of rupture by using patient-specific computational hemodynamics models. AJNR Am. J. Neuroradiol. 26(10):2550–2559, 2005.
Cebral, J. R., M. A. Castro, et al. Flow–area relationship in internal carotid and vertebral arteries. Physiol. Meas. 29(5):585–594, 2008.
Cebral, J. R., S. Hendrickson, et al. Hemodynamics in a lethal basilar artery aneurysm just before its rupture. AJNR Am. J. Neuroradiol. 30(1):95–98, 2009.
Chatziprodromou, I., V. D. Butty, et al. Pulsatile blood flow in anatomically accurate vessels with multiple aneurysms: a medical intervention planning application of computational haemodynamics. Flow Turbul. Combust. 71:333–346, 2003.
Cheng, C., F. Helderman, et al. Large variations in absolute wall shear stress levels within one species and between species. Atherosclerosis 195(2):225–235, 2007.
Dempere-Marco, L., E. Oubel, et al. CFD analysis incorporating the influence of wall motion: application to intracranial aneurysms. Med. Image Comput. Comput. Assist. Interv. 9(Pt 2):438–445, 2006.
Fedorov, B. M., T. V. Sebekina, et al. [Stress and blood circulation in man]. Kosm. Biol. Aviakosm. Med. 24(3):35–40, 1990.
Ferns, S. P., J. J. Schneiders, et al. Intracranial blood-flow velocity and pressure measurements using an intra-arterial dual-sensor guidewire. AJNR Am. J. Neuroradiol. 31(2):324–326, 2010.
Ford, M. D., H. N. Nikolov, et al. PIV-measured versus CFD-predicted flow dynamics in anatomically realistic cerebral aneurysm models. J. Biomech. Eng. 130(2):021015, 2008.
Formaggia, L., J. F. Gerbeau, et al. On the coupling of 3d and 1d Navier–Stokes equations for flow problems in compliant vessels. Comput. Methods Appl. Mech. Eng. 191:561–582, 2001.
Gonzalez-Alonso, J., M. K. Dalsgaard, et al. Brain and central haemodynamics and oxygenation during maximal exercise in humans. J Physiol 557(Pt 1):331–342, 2004.
Gwilliam, M. N., N. Hoggard, et al. MR derived volumetric flow rate waveforms at locations within the common carotid, internal carotid, and basilar arteries. J. Cereb. Blood Flow Metab. 29(12):1975–1982, 2009.
Hajjar, I., M. Selim, et al. The relationship between nighttime dipping in blood pressure and cerebral hemodynamics in nonstroke patients. J. Clin. Hypertens. (Greenwich) 9(12):929–936, 2007.
Hassan, T., M. Ezura, et al. Computational simulation of therapeutic parent artery occlusion to treat giant vertebrobasilar aneurysm. AJNR Am. J. Neuroradiol. 25(1):63–68, 2004.
Hop, J. W., G. J. Rinkel, et al. Case-fatality rates and functional outcome after subarachnoid hemorrhage: a systematic review. Stroke 28(3):660–664, 1997.
Ide, K., and N. H. Secher. Cerebral blood flow and metabolism during exercise. Prog. Neurobiol. 61(4):397–414, 2000.
Isoda, H., Y. Ohkura, et al. In vivo hemodynamic analysis of intracranial aneurysms obtained by magnetic resonance fluid dynamics (MRFD) based on time-resolved three-dimensional phase-contrast MRI. Neuroradiology 52(10):921–928, 2010.
Jou, L. D., D. H. Lee, et al. Wall shear stress on ruptured and unruptured intracranial aneurysms at the internal carotid artery. AJNR Am. J. Neuroradiol. 29(9):1761–1767, 2008.
Jou, L. D., C. M. Quick, et al. Computational approach to quantifying hemodynamic forces in giant cerebral aneurysms. AJNR Am. J. Neuroradiol. 24(9):1804–1810, 2003.
Jou, L. D., G. Wong, et al. Correlation between lumenal geometry changes and hemodynamics in fusiform intracranial aneurysms. AJNR Am. J. Neuroradiol. 26(9):2357–2363, 2005.
Karmonik, C., G. Benndorf, et al. Wall shear stress variations in basilar tip aneurysms investigated with computational fluid dynamics. Conf. Proc. IEEE Eng. Med. Biol. Soc. 1:3214–3217, 2006.
Kulikov, V. P., V. N. Grechishnikov, et al. [Response of cerebral hemodynamics to combined stress impacts]. Patol. Fiziol. Eksp. Ter. 1:7–9, 2005.
Lagana, K., G. Dubini, et al. Multiscale modelling as a tool to prescribe realistic boundary conditions for the study of surgical procedures. Biorheology 39(3–4):359–364, 2002.
Mantha, A., C. Karmonik, et al. Hemodynamics in a cerebral artery before and after the formation of an aneurysm. AJNR Am. J. Neuroradiol. 27(5):1113–1118, 2006.
Marzo, A., P. Singh, et al. Influence of inlet boundary conditions on the local haemodynamics of intracranial aneurysms. Comput. Methods Biomech. Biomed. Eng. 12(4):431–444, 2009.
Mitsos, A. P., N. M. Kakalis, et al. Haemodynamic simulation of aneurysm coiling in an anatomically accurate computational fluid dynamics model: technical note. Neuroradiology 50(4):341–347, 2008.
Moyle, K. R., L. Antiga, et al. Inlet conditions for image-based CFD models of the carotid bifurcation: is it reasonable to assume fully developed flow? J. Biomech. Eng. 128(3):371–379, 2006.
Oshima, M., H. Sakai, et al. Modelling of inflow boundary conditions for image-based simulation of cerebrovascular flow. Int. J. Numer. Methods Fluids 47:603–617, 2005.
Quarteroni, A., and L. Formaggia. Mathematical modelling and numerical simulation of the cardiovascular system. In: Handbook of Numerical Analysis, Vol. 12, edited by P. G. Ciarlet, and J. L. Lions. Amsterdam: Elsevier, 2004, pp. 3–127.
Radaelli, A. G., L. Augsburger, et al. Reproducibility of haemodynamical simulations in a subject-specific stented aneurysm model—a report on the Virtual Intracranial Stenting Challenge 2007. J. Biomech. 41(10):2069–2081, 2008.
Rayz, V. L., L. Boussel, et al. Numerical simulations of flow in cerebral aneurysms: comparison of CFD results and in vivo MRI measurements. J. Biomech. Eng. 130(5):051011, 2008.
Reymond, P., F. Merenda, et al. Validation of a one-dimensional model of the systemic arterial tree. Am. J. Physiol. Heart Circ. Physiol. 297(1):H208–H222, 2009.
Sekhar, L. N., and R. C. Heros. Origin, growth, and rupture of saccular aneurysms: a review. Neurosurgery 8(2):248–260, 1981.
Shimogonya, Y., T. Ishikawa, et al. Can temporal fluctuation in spatial wall shear stress gradient initiate a cerebral aneurysm? A proposed novel hemodynamic index, the gradient oscillatory number (GON). J. Biomech. 42(4):550–554, 2009.
Shojima, M., M. Oshima, et al. Magnitude and role of wall shear stress on cerebral aneurysm: computational fluid dynamic study of 20 middle cerebral artery aneurysms. Stroke 35(11):2500–2505, 2004.
Shojima, M., M. Oshima, et al. Role of the bloodstream impacting force and the local pressure elevation in the rupture of cerebral aneurysms. Stroke 36(9):1933–1938, 2005.
Singh, P. K., A. Marzo, et al. Effects of heparin on the hemodynamic characteristics of intracranial aneurysms. J. Neuroradiol. Apr 5.
Steinman, D. A., J. S. Milner, et al. Image-based computational simulation of flow dynamics in a giant intracranial aneurysm. AJNR Am. J. Neuroradiol. 24(4):559–566, 2003.
Venugopal, P., D. Valentino, et al. Sensitivity of patient-specific numerical simulation of cerebral aneurysm hemodynamics to inflow boundary conditions. J. Neurosurg. 106(6):1051–1060, 2007.
Zhao, M., S. Amin-Hanjani, et al. Regional cerebral blood flow using quantitative MR angiography. AJNR Am. J. Neuroradiol. 28(8):1470–1473, 2007.
Acknowledgments
The @neurIST project is funded by the European Commission, VI Framework Programme, Priority 2, Information Society Technologies (Oxfordshire Research Ethics Committee—A Study Number: 07/Q1604/53).
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Peter E. McHugh oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Marzo, A., Singh, P., Larrabide, I. et al. Computational Hemodynamics in Cerebral Aneurysms: The Effects of Modeled Versus Measured Boundary Conditions. Ann Biomed Eng 39, 884–896 (2011). https://doi.org/10.1007/s10439-010-0187-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-010-0187-z