Abstract
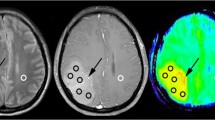
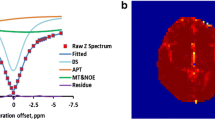
The aim of the study was to investigate the ability of 1HMRS to reflect proliferative activity of diffuse low-grade gliomas (WHO grade II). Between November 2002 and March 2007, a prospective study was performed on consecutive patients with suspected supratentorial hemispheric diffuse low-grade tumors. All the patients underwent MR examination using uniform procedures, and then surgical resection or biopsy within 2 weeks of the MR examination. Proliferative activity of the tumors was assessed by Ki-67 immunochemistry (Mb-1) on paraffin embedded tumor sections. Spectroscopic data was compared with Ki-67 labeling index and other histologic data such as histological subtype, cellular atypia, cellular density using univariate and multivariate analysis. 82 of 97 consecutive patients had histologically confirmed WHO grade 2 gliomas. Ki-67 proliferation index (PI) was correlated with specific spectral patterns: (1) low PI (<4%) was associated with increased Cho/Cr and absence of both free lipids or lactates; (2) intermediate PI (4–8%) was associated with resonance of lactates; and (3) high PI (>8%) was characterized by a resonance of free lipids. On multivariate analysis, resonance of lactates and resonance of free lipids appeared as independent predictors of intermediate PI (P < 0.001) and high PI (P < 0.001), respectively; moreover, free lipids resonance was correlated with cellular atypia (P < 0.05). This study suggests that 1HMRS is a reliable tool to evaluate the proliferation activity of WHO grade 2 glioma and to identify potentially more aggressive clinical behavior.

Similar content being viewed by others
References
Alger JR et al (1990) Metabolism of human gliomas: assessment with H-1 MR spectroscopy and F-18 fluorodeoxyglucose PET. Radiology 177(3):633–641
Ott D, Hennig J, Ernst T (1993) Human brain tumors: assessment with in vivo proton MR spectroscopy. Radiology 186(3):745–752
Michaelis T et al (1993) Absolute concentrations of metabolites in the adult human brain in vivo: quantification of localized proton MR spectra. Radiology 187(1):219–227
Miller BL (1991) A review of chemical issues in 1H NMR spectroscopy: N-acetyl-l-aspartate, creatine and choline. NMR Biomed 4(2):47–52
Urenjak J et al (1993) Proton nuclear magnetic resonance spectroscopy unambiguously identifies different neural cell types. J Neurosci 13(3):981–989
Kemp GJ (2000) Non-invasive methods for studying brain energy metabolism: what they show and what it means. Dev Neurosci 22(5–6):418–428
Croteau D et al (2001) Correlation between magnetic resonance spectroscopy imaging and image-guided biopsies: semiquantitative and qualitative histopathological analyses of patients with untreated glioma. Neurosurgery 49(4):823–829
Dowling C et al (2001) Preoperative proton MR spectroscopic imaging of brain tumors: correlation with histopathologic analysis of resection specimens. Am J Neuroradiol 22(4):604–612
Vuori K et al (2004) Low-grade gliomas and focal cortical developmental malformations: differentiation with proton MR spectroscopy. Radiology 230(3):703–708
Herminghaus S et al (2003) Determination of histopathological tumor grade in neuroepithelial brain tumors by using spectral pattern analysis of in vivo spectroscopic data. J Neurosurg 98(1):74–81
Yang D et al (2002) Cerebral gliomas: prospective comparison of multivoxel 2D chemical-shift imaging proton MR spectroscopy, echoplanar perfusion and diffusion-weighted MRI. Neuroradiology 44(8):656–666
Mandonnet E et al (2003) Continuous growth of mean tumor diameter in a subset of grade II gliomas. Ann Neurol 53(4):524–528
Hoyt JW et al (1995) Analysis of proliferative grade in glial neoplasms using antibodies to the Ki-67 defined antigen and PCNA in formalin fixed, deparaffinized tissues. J Neurooncol 24(2):163–169
Steck K, el-Naggar AK (1994) Comparative flow cytometric analysis of Ki-67 and proliferating cell nuclear antigen (PCNA) in solid neoplasms. Cytometry 17(3):258–265
Kleihues P, Sobin LH (2000) World Health Organization classification of tumors. Cancer 88(12):2887
Czernicki Z et al (2000) Malignancy of brain tumors evaluated by proton magnetic resonance spectroscopy (1H-MRS) in vitro. Acta Neurochir Suppl 76:17–20
Isobe T et al (2002) Quantification of cerebral metabolites in glioma patients with proton MR spectroscopy using T2 relaxation time correction. Magn Reson Imaging 20(4):343–349
Tedeschi G et al (1997) Increased choline signal coinciding with malignant degeneration of cerebral gliomas: a serial proton magnetic resonance spectroscopy imaging study. J Neurosurg 87(4):516–524
Piepmeier JM (2001) Cerebral gliomas: how tumor biology affects management. Clin Neurosurg 48:3–9
Pallud J et al (2006) Prognostic value of initial magnetic resonance imaging growth rates for World Health Organization grade II gliomas. Ann Neurol 60(3):380–383
Piepmeier J et al (1996) Variations in the natural history and survival of patients with supratentorial low-grade astrocytomas. Neurosurgery 38(5):872–878 (discussion 878–879)
Louis DN et al (1991) Proliferating cell nuclear antigen and Ki-67 immunohistochemistry in brain tumors: a comparative study. Acta Neuropathol (Berl) 81(6):675–679
Neder L et al (2004) MIB-1 labeling index in astrocytic tumors–a clinicopathologic study. Clin Neuropathol 23(6):262–270
Wakimoto H et al (1996) Prognostic significance of Ki-67 labeling indices obtained using MIB-1 monoclonal antibody in patients with supratentorial astrocytomas. Cancer 77(2):373–380
Daumas-Duport C et al (1988) Grading of astrocytomas. A simple and reproducible method. Cancer 62(10):2152–2165
Kondziolka D, Lunsford LD, Martinez AJ (1993) Unreliability of contemporary neurodiagnostic imaging in evaluating suspected adult supratentorial (low-grade) astrocytoma. J Neurosurg 79(4):533–536
Cao Y et al (2006) Physiologic and metabolic magnetic resonance imaging in gliomas. J Clin Oncol 24(8):1228–1235
Julia-Sape M et al (2006) Comparison between neuroimaging classifications and histopathological diagnoses using an international multicenter brain tumor magnetic resonance imaging database. J Neurosurg 105(1):6–14
Lev MH et al (2004) Glial tumor grading and outcome prediction using dynamic spin-echo MR susceptibility mapping compared with conventional contrast-enhanced MR: confounding effect of elevated rCBV of oligodendrogliomas [corrected]. Am J Neuroradiol 25(2):214–221
Rees J (2003) Advances in magnetic resonance imaging of brain tumours. Curr Opin Neurol 16(6):643–650
Onda K et al (1994) Correlation between the bromodeoxyuridine labeling index and the MIB-1 and Ki-67 proliferating cell indices in cerebral gliomas. Cancer 74(7):1921–1926
Matsumura A et al (2005) Correlation between choline and MIB-1 index in human gliomas. A quantitative in proton MR spectroscopy study. J Clin Neurosci 12(4):416–420
Shimizu H et al (2000) Correlation between choline level measured by proton MR spectroscopy and Ki-67 labeling index in gliomas. Am J Neuroradiol 21(4):659–665
Law M et al (2006) Perfusion magnetic resonance imaging predicts patient outcome as an adjunct to histopathology: a second reference standard in the surgical and nonsurgical treatment of low-grade gliomas. Neurosurgery 58(6):1099–1107 (discussion 1099–1107)
Cha S et al (2003) Dynamic, contrast-enhanced perfusion MRI in mouse gliomas: correlation with histopathology. Magn Reson Med 49(5):848–855
Shin JH et al (2002) Using relative cerebral blood flow and volume to evaluate the histopathologic grade of cerebral gliomas: preliminary results. Am J Roentgenol 179(3):783–789
Sugahara T et al (1998) Correlation of MR imaging-determined cerebral blood volume maps with histologic and angiographic determination of vascularity of gliomas. Am J Roentgenol 171(6):1479–1486
Castillo M, Kwock L, Mukherji SK (1996) Clinical applications of proton MR spectroscopy. Am J Neuroradiol 17(1):1–15
Danielsen ER, Ross B (1999) Magnetic resonance spectroscopy diagnosis of neurological diseases (Book). Marcel Dekker, INC. Editor, New York, Basel
Leclerc X, Huisman TA, Sorensen AG (2002) The potential of proton magnetic resonance spectroscopy ((1)H-MRS) in the diagnosis and management of patients with brain tumors. Curr Opin Oncol 14(3):292–298
Nafe R et al (2003) Preoperative proton-MR spectroscopy of gliomas—correlation with quantitative nuclear morphology in surgical specimen. J Neurooncol 63(3):233–245
Law M et al (2003) Glioma grading: sensitivity, specificity, and predictive values of perfusion MR imaging and proton MR spectroscopic imaging compared with conventional MR imaging. Am J Neuroradiol 24(10):1989–1998
Reijneveld JC et al (2005) Proton MRS imaging in the follow-up of patients with suspected low-grade gliomas. Neuroradiology 47(12):887–891
Duffau H et al (2005) Contribution of intraoperative electrical stimulations in surgery of low grade gliomas: a comparative study between two series without (1985–96) and with (1996–2003) functional mapping in the same institution. J Neurol Neurosurg Psychiatry 76(6):845–851
Hoang-Xuan K et al (2004) Temozolomide as initial treatment for adults with low-grade oligodendrogliomas or oligoastrocytomas and correlation with chromosome 1p deletions. J Clin Oncol 22(15):3133–3138
Van den Bent MJ et al (2003) Phase II study of first-line chemotherapy with temozolomide in recurrent oligodendroglial tumors: the European Organization for Research and Treatment of Cancer Brain Tumor Group Study 26971. J Clin Oncol 21(13):2525–2528
Van den Bent MJ (2003) Can chemotherapy replace radiotherapy in low-grade gliomas? Time for randomized studies. Semin Oncol 30(6 suppl 19):39–44
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guillevin, R., Menuel, C., Duffau, H. et al. Proton magnetic resonance spectroscopy predicts proliferative activity in diffuse low-grade gliomas. J Neurooncol 87, 181–187 (2008). https://doi.org/10.1007/s11060-007-9508-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-007-9508-y