Abstract
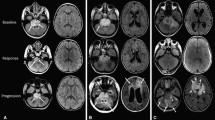
Pseudoprogression (PsP) is a treatment-related phenomenon which hinders response interpretation. Its prevalence and clinical impact have not been evaluated in children/adolescents. We assessed the characteristics, risk factors and prognosis of PsP in children/adolescents and young-adults diagnosed with non-brainstem high grade gliomas (HGG) and diffuse intrinsic pontine gliomas (DIPG). Patients aged 1–21 years diagnosed with HGG or DIPG between 1995 and 2012 who had completed radiotherapy were eligible. PsP was assessed according to study-specific criteria and correlated with first-line treatment, molecular biomarkers and survival. Ninety-one patients (47 HGG, 44 DIPG) were evaluable. Median age: 10 years (range, 2–20). Eleven episodes of PsP were observed in 10 patients (4 HGG, 6 DIPG). Rates of PsP: 8.5 % (HGG); 13.6 % (DIPG). Two episodes of PsP were based on clinical findings alone; nine episodes had concurrent radiological changes: increased size of lesions (n = 5), new focal enhancement (n = 4). Temozolomide, MGMT methylation or H3F3A mutations were not found to be associated with increased occurrence of PsP. For HGG, 1-year progression-free survival (PFS) was 41.9 % no-PsP versus 100 % PsP (p = 0.041); differences in 1-year overall survival (OS) were not significant. For DIPG, differences in 1-year PFS and OS were not statistically significant. Hazard ratio (95 %CI) of PsP for OS was 0.551 (0.168–1.803; p = 0.325) in HGG; and 0.308 (0.107–0.882; p = 0.028) in DIPG. PsP occurred in both pediatric HGG and DIPG patients at a comparable rate to adult HGG. PsP was associated with improved 1-yr PFS in HGG patients. PsP had a protective effect upon OS in DIPG patients.

Similar content being viewed by others
References
Brandsma D, Stalpers L, Taal W, Sminia P, van den Bent MJ (2008) Clinical features, mechanisms, and management of pseudoprogression in malignant gliomas. Lancet Oncol 9:453–461
Verma N, Cowperthwaite MC, Burnett MG, Markey MK (2013) Differentiating tumor recurrence from treatment necrosis: a review of neuro-oncologic imaging strategies. Neuro Oncol 15:515–534
Wen PY, Macdonald DR, Reardon DA et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28:1963–1972
Kruser TJ, Mehta MP, Robins HI (2013) Pseudoprogression after glioma therapy: a comprehensive review. Expert Rev Neurother 13:389–403
Brandes AA, Franceschi E, Tosoni A et al (2008) MGMT promoter methylation status can predict the incidence and outcome of pseudoprogression after concomitant radiochemotherapy in newly diagnosed glioblastoma patients. J Clin Oncol 26:2192–2197
Thompson EM, Guillaume DJ, Dósa E, Li X, Nazemi KJ, Gahramanov S, Hamilton BE, Neuwelt EA (2012) Dual contrast perfusion MRI in a single imaging session for assessment of pediatric brain tumors. J Neurooncol 109:105–114
Ozdemir N, Celkan T (2010) Pseudoprogression after radiotherapy with concurrent temozolomide in a child with anaplastic astrocytoma. Pediatr Hematol Oncol 27:317–319
Negretti L, Blanchard P, Couanet D, Kieffer V, Goma G, Habrand JL, Dhermain F, Valteau-Couanet D, Grill J, Dufour C (2012) Pseudoprogression after high-dose busulfan-thiotepa with autologous stem cell transplantation and radiation therapy in children with brain tumors: impact on survival. Neuro Oncol 14:1413–1421
Chassot A, Canale S, Varlet P et al (2012) Radiotherapy with concurrent and adjuvant temozolomide in children with newly diagnosed diffuse intrinsic pontine glioma. J Neurooncol 106:399–407
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW, Kleihues P (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114:97–109
Warren KE (2012) Diffuse intrinsic pontine glioma: poised for progress. Front Oncol 2:205
Gaspar N, Marshall L, Perryman L et al (2010) MGMT-independent temozolomide resistance in pediatric glioblastoma cells associated with a PI3-kinase-mediated HOX/stem cell gene signature. Cancer Res 70:9243–9252
Gessi M, Gielen GH, Hammes J, Dörner E, Zur Mühlen A, Waha A, Pietsch T (2013) H3.3 G34R mutations in pediatric primitive neuroectodermal tumors of central nervous system (CNS-PNET) and pediatric glioblastomas: possible diagnostic and therapeutic implications? J Neurooncol 112:67–72
Bechet D, Gielen GGH, Korshunov A et al (2014) Specific detection of methionine 27 mutation in histone 3 variants (H3K27M) in fixed tissue from high-grade astrocytomas. Acta Neuropathol 128:733–741
Sturm D, Witt H, Hovestadt V et al (2012) Hotspot mutations in H3F3A and IDH1 define distinct epigenetic and biological subgroups of glioblastoma. Cancer Cell 22:425–437
Bady P, Sciuscio D, Diserens A-C et al (2012) MGMT methylation analysis of glioblastoma on the Infinium methylation BeadChip identifies two distinct CpG regions associated with gene silencing and outcome, yielding a prediction model for comparisons across datasets, tumor grades, and CIMP-status. Acta Neuropathol 124:547–560
Sanghera P, Perry J, Sahgal A, Symons S, Aviv R, Morrison M, Lam K, Davey P, Tsao MN (2010) Pseudoprogression following chemoradiotherapy for glioblastoma multiforme. Can J Neurol Sci 37:36–42
de Wit MCY, de Bruin HG, Eijkenboom W, Sillevis Smitt PAE, van den Bent MJ (2004) Immediate post-radiotherapy changes in malignant glioma can mimic tumor progression. Neurology 63:535–537
Taal W, Brandsma D, de Bruin HG, Bromberg JE, Swaak-Kragten AT, Smitt PAES, van Es CA, van den Bent MJ (2008) Incidence of early pseudo-progression in a cohort of malignant glioma patients treated with chemoirradiation with temozolomide. Cancer 113:405–410
Clarke JL, Iwamoto FM, Sul J et al (2009) Randomized phase II trial of chemoradiotherapy followed by either dose-dense or metronomic temozolomide for newly diagnosed glioblastoma. J Clin Oncol 27:3861–3867
Roldán GB, Scott JN, McIntyre JB et al (2009) Population-based study of pseudoprogression after chemoradiotherapy in GBM. Can J Neurol Sci 36:617–622
Gerstner ER, McNamara MB, Norden AD, Lafrankie D, Wen PY (2009) Effect of adding temozolomide to radiation therapy on the incidence of pseudo-progression. J Neurooncol 94:97–101
Pouleau H-B, Sadeghi N, Balériaux D, Mélot C, De Witte O, Lefranc F (2012) High levels of cellular proliferation predict pseudoprogression in glioblastoma patients. Int J Oncol 40:923–928
Stuplich M, Hadizadeh DR, Kuchelmeister K et al (2012) Late and prolonged pseudoprogression in glioblastoma after treatment with lomustine and temozolomide. J Clin Oncol 30:e180–e183
Chaskis C, Neyns B, Michotte A, De Ridder M, Everaert H (2009) Pseudoprogression after radiotherapy with concurrent temozolomide for high-grade glioma: clinical observations and working recommendations. Surg Neurol 72:423–428
Radbruch A, Fladt J, Kickingereder P et al (2015) Pseudoprogression in patients with glioblastoma: clinical relevance despite low incidence. Neuro Oncol 17:151–159
Chan DTM, Ng RYT, Siu DYW et al (2012) Pseudoprogression of malignant glioma in Chinese patients receiving concomitant chemoradiotherapy. Hong Kong Med J 18:221–225
Linhares P, Carvalho B, Figueiredo R, Reis RM, Vaz R (2013) Early Pseudoprogression following Chemoradiotherapy in Glioblastoma Patients: the Value of RANO Evaluation. J Oncol 2013:690585
Chang JH, Kim C-Y, Choi BS, Kim YJ, Kim JS, Kim IA (2014) Pseudoprogression and pseudoresponse in the management of high-grade glioma: optimal decision timing according to the response assessment of the neuro-oncology working group. J Korean Neurosurg Soc 55:5–11
Hygino da Cruz LC, Rodriguez I, Domingues RC, Gasparetto EL, Sorensen AG (2011) Pseudoprogression and pseudoresponse: imaging challenges in the assessment of posttreatment glioma. AJNR Am J Neuroradiol 32:1978–1985
Warren KE, Poussaint TY, Vezina G et al (2013) Challenges with defining response to antitumor agents in pediatric neuro-oncology: a report from the response assessment in pediatric neuro-oncology (RAPNO) working group. Pediatr Blood Cancer 60:1397–1401
Gunjur A, Lau E, Taouk Y, Ryan G (2011) Early post-treatment pseudo-progression amongst glioblastoma multiforme patients treated with radiotherapy and temozolomide: a retrospective analysis. J Med Imaging Radiat Oncol 55:603–610
Young RJ, Gupta A, Shah AD, Graber JJ, Zhang Z, Shi W, Holodny AI, Omuro AMP (2011) Potential utility of conventional MRI signs in diagnosing pseudoprogression in glioblastoma. Neurology 76:1918–1924
Chamberlain MC, Glantz MJ, Chalmers L, Van Horn A, Sloan AE (2007) Early necrosis following concurrent Temodar and radiotherapy in patients with glioblastoma. J Neurooncol 82:81–83
Tsien C, Galbán CJ, Chenevert TL et al (2010) Parametric response map as an imaging biomarker to distinguish progression from pseudoprogression in high-grade glioma. J Clin Oncol 28:2293–2299
Yaman E, Buyukberber S, Benekli M, Oner Y, Coskun U, Akmansu M, Ozturk B, Kaya AO, Uncu D, Yildiz R (2010) Radiation induced early necrosis in patients with malignant gliomas receiving temozolomide. Clin Neurol Neurosurg 112:662–667
Peca C, Pacelli R, Elefante A, De Caro MD, Vergara P, Mariniello G, Giamundo A, Maiuri F (2009) Early clinical and neuroradiological worsening after radiotherapy and concomitant temozolomide in patients with glioblastoma: tumour progression or radionecrosis? Clin Neurol Neurosurg 111:331–334
Topkan E, Topuk S, Oymak E, Parlak C, Pehlivan B (2012) Pseudoprogression in patients with glioblastoma multiforme after concurrent radiotherapy and temozolomide. Am J Clin Oncol 35:284–289
Kang H-C, Kim C-Y, Han JH, Choe GY, Kim JH, Kim JH, Kim IA (2011) Pseudoprogression in patients with malignant gliomas treated with concurrent temozolomide and radiotherapy: potential role of p53. J Neurooncol 102:157–162
Ceschin R, Kurland BF, Abberbock SR, Ellingson BM, Okada H, Jakacki RI, Pollack IF, Panigrahy A (2015) Parametric response mapping of apparent diffusion coefficient as an imaging biomarker to distinguish pseudoprogression from true tumor progression in peptide-based vaccine therapy for pediatric diffuse intrinsic pontine glioma. AJNR Am J Neuroradiol 36:2170–2176
Nasseri M, Gahramanov S, Netto JP, Fu R, Muldoon LL, Varallyay C, Hamilton BE, Neuwelt EA (2014) Evaluation of pseudoprogression in patients with glioblastoma multiforme using dynamic magnetic resonance imaging with ferumoxytol calls RANO criteria into question. Neuro Oncol 16:1146–1154
Fabi A, Russillo M, Metro G, Vidiri A, Di Giovanni S, Cognetti F (2009) Pseudoprogression and MGMT status in glioblastoma patients: implications in clinical practice. Anticancer Res 29:2607–2610
Kong D-S, Kim ST, Kim E-H, Lim DH, Kim WS, Suh Y-L, Lee J-I, Park K, Kim JH, Nam D-H (2011) Diagnostic dilemma of pseudoprogression in the treatment of newly diagnosed glioblastomas: the role of assessing relative cerebral blood flow volume and oxygen-6-methylguanine-DNA methyltransferase promoter methylation status. AJNR Am J Neuroradiol 32:382–387
Park C-K, Kim J, Yim SY et al (2011) Usefulness of MS-MLPA for detection of MGMT promoter methylation in the evaluation of pseudoprogression in glioblastoma patients. Neuro Oncol 13:195–202
Wu G, Broniscer A, McEachron TA et al (2012) Somatic histone H3 alterations in pediatric diffuse intrinsic pontine gliomas and non-brainstem glioblastomas. Nat Genet 44:251–253
Korshunov A, Ryzhova M, Hovestadt V et al (2015) Integrated analysis of pediatric glioblastoma reveals a subset of biologically favorable tumors with associated molecular prognostic markers. Acta Neuropathol 129:669–678
Prager AJ, Martinez N, Beal K, Omuro A, Zhang Z, Young RJ (2015) Diffusion and perfusion MRI to differentiate treatment-related changes including pseudoprogression from recurrent tumors in high-grade gliomas with histopathologic evidence. AJNR Am J Neuroradiol 36:877–885
Matsusue E, Fink JR, Rockhill JK, Ogawa T, Maravilla KR (2010) Distinction between glioma progression and post-radiation change by combined physiologic MR imaging. Neuroradiology 52:297–306
Hatzoglou V, Ulaner GA, Zhang Z, Beal K, Holodny AI, Young RJ (2013) Comparison of the effectiveness of MRI perfusion and fluorine-18 FDG PET-CT for differentiating radiation injury from viable brain tumor: a preliminary retrospective analysis with pathologic correlation in all patients. Clin Imaging 37:451–457
Acknowledgments
Ms Helen Underwood, Royal Marsden Registry department. Mr Karl Spolander, PACS manager. Ms Julie Mycroft, Royal Marsden Pharmacy. We acknowledge NHS funding to the NIHR Biomedical Research Centre (BRC) and the NIHR Clinical Research Facility at The Royal Marsden and the Institute of Cancer Research, as well as Experimental Cancer Medicines Centre (ECMC) funding. Additionally, F.C. holds a senior fellowship funded by the BRC and the Sohn Foundation. L.F., L.M. and L.V.M. are funded by the Oak Foundation (Grant OCay-04-169). A.D.J.P. is supported by Cancer Research UK (CRUK): programme grant C1178/A10294 - Chair in Paediatric Oncology. M.O.L. is an NIHR Senior Investigator. CRUK and Engineering and Physical Sciences Research Council (EPSRC) funding to the pediatric programme in functional imaging (C7809/A10342).
Funding
CRUK and EPSRC funding to the CRUK Centre for Cancer Imaging (C1060/A16464, C1060/A10334).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Carceller, F., Fowkes, L.A., Khabra, K. et al. Pseudoprogression in children, adolescents and young adults with non-brainstem high grade glioma and diffuse intrinsic pontine glioma. J Neurooncol 129, 109–121 (2016). https://doi.org/10.1007/s11060-016-2151-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-016-2151-8