Abstract
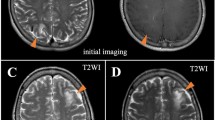
Tacrolimus is a potent immunosuppressive drug widely used to prevent and treat graft-versus-host disease (GVHD) in stem cell transplantation (SCT). Among 49 patients receiving tacrolimus who underwent SCT from January 2000 to July 2003, 10 patients (20%) developed encephalopathy. The commonly observed symptoms were convulsions and drowsiness, and most patients complained of signal symptoms such as headache, nausea, and cortical blindness before onset. The most common abnormality on neuroimages was high-intensity lesions in white matter on magnetic resonance imaging T2-weighted or fluid-attenuated inversion recovery images. At onset, all patients were receiving treatment for acute GVHD (grade II/III) or extensive chronic GVHD and demonstrated an abrupt increase in blood pressure from baseline levels. The serum tacrolimus concentration was generally within acceptable levels at onset. Symptoms gradually improved in all patients when the blood pressure was lowered with antihypertensive medication, regardless of continued tacrolimus administration following a short-term suspension. The pathogenesis of tacrolimus-related encephalopathy is multifactorial, although refractory GVHD and a sudden increase in blood pressure seem to be major predisposing factors. Because the withdrawal of tacrolimus or switching to less potent anti-GVHD agents usually worsens the GVHD, the administration of tacrolimus should be managed by closely monitoring serum levels and controlling blood pressure.
Article PDF
Similar content being viewed by others
References
Yanik G, Levine JE, Ratanatharathorn V, Dunn R, Ferrara J, Hutchinson RJ. Tacrolimus (FK506) and methotrexate as prophylaxis for acute graft-versus-host disease in pediatric allogeneic stem cell transplantation. Bone Marrow Transplant. 2000;26:161–167.
Tamaki H, Kawakami M, Ikegame K, et al. Successful treatment of tacrolimus (FK506)-related leukoencephalopathy with cerebral hemorrhage in a patient who underwent nonmyeloablative stem cell transplantation. Int J Hematol. 2004;80:291–294.
Devine SM, Newman NJ, Siegel JL, et al. Tacrolimus (FK506)- induced cerebral blindness following bone marrow transplantation. Bone Marrow Transplant. 1996;18:569–572.
Misawa A,Takeuchi Y, Hibi S, Todo S, Imashuku S, Sawada T. FK506-induced intractable leukoencephalopathy following allogeneic bone marrow transplantation. Bone Marrow Transplant. 2000;25:331–334.
Grimbert P, Azema C, Pastural M, et al. Tacrolimus (FK506)- induced severe and late encephalopathy in a renal transplant recipient. Nephrol Dial Transplant. 1999;14:2489–2491.
Mori A, Tanaka J, Kobayashi S, et al. Fatal cerebral hemorrhage associated with cyclosporin-A/FK506-related encephalopathy after allogeneic bone marrow transplantation. Ann Hematol. 2000;79:588–592.
Furukawa M, Terae S, Chu BC, Kaneko K, Kamada H, Miyasaka K. MRI in seven cases of tacrolimus (FK-506) encephalopathy: utility of FLAIR and diffusion-weighted imaging. Neuroradiology. 2001; 43:615–621.
Teive HA, Brandi IV, Camargo CH, et al. Reversible posterior leucoencephalopathy syndrome associated with bone marrow transplantation. Arq Neuropsiquiatr. 2001;59:784–789.
Antonini G, Ceschin V, Morino S, et al. Early neurologic complications following allogeneic bone marrow transplant for leukemia: a prospective study. Neurology. 1998;50:1441–1445.
Faraci M, Lanino E, Dini G, et al. Severe neurologic complications after hematopoietic stem cell transplantation in children. Neurology. 2002;59:1895–1904.
Eidelman BH, Abu-Elmagd K, Wilson J, et al. Neurologic complications of FK 506. Transplant Proc. 1991;23:3175–3178.
de Brabander C, Cornelissen J, Smitt PA, Vecht CJ, van den Bent MJ. Increased incidence of neurological complications in patients receiving an allogenic bone marrow transplantation from alternative donors. J Neurol Neurosurg Psychiatry. 2000;68:36–40.
Kochi S, Takanaga H, Matsuo H, et al. Induction of apoptosis in mouse brain capillary endothelial cells by cyclosporin A and tacrolimus. Life Sci. 2000;66:2255–2260.
Bleyer WA. Neurologic sequelae of methotrexate and ionizing radiation: a new classification. Cancer Treat Rep. 1981;65(suppl 1):89–98.
Yeager AM, Wagner JE Jr, Graham ML, Jones RJ, Santos GW, Grochow LB. Optimization of busulfan dosage in children undergoing bone marrow transplantation: a pharmacokinetic study of dose escalation. Blood. 1992;80:2425–2428.
Zabernigg A, Maier H, Thaler J, Gattringer C. Late-onset fatal neurological toxicity of fludarabine. Lancet. 1994;344:1780.
Reece DE, Frei-Lahr DA, Shepherd JD, et al. Neurologic complications in allogeneic bone marrow transplant patients receiving cyclosporin. Bone Marrow Transplant. 1991;8:393–401.
Matsuda Y, Hara J, Miyoshi H, et al. Thrombotic microangiopathy associated with reactivation of human herpesvirus-6 following high- dose chemotherapy with autologous bone marrow transplantation in young children. Bone Marrow Transplant. 1999;24:919–923.
Maslo C, Peraldi MN, Desenclos JC, et al. Thrombotic microangiopathy and cytomegalovirus disease in patients infected with human immunodeficiency virus. Clin Infect Dis. 1997;24:350–355.
Hinchey J, Chaves C, Appignani B, et al. A reversible posterior leukoencephalopathy syndrome. N Engl J Med. 1996;334:494–500.
Kaczmarek I, Groetzner J, Meiser B, et al. Impairment of the blood-brain barrier can result in tacrolimus-induced reversible leucoencephalopathy following heart transplantation. Clin Transplant. 2003;17:469–472.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kanekiyo, T., fnHara, J., Matsuda-Hashii, Y. et al. Tacrolimus-Related Encephalopathy following Allogeneic Stem Cell Transplantation in Children. Int J Hematol 81, 264–268 (2005). https://doi.org/10.1532/IJH97.04162
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1532/IJH97.04162