Abstract
BACKGROUND AND PURPOSE: DTI data of the normal healthy spinal cord in children are limited compared with adults and are typically focused on the cervical spinal cord. The purpose of this study was the following: to investigate the feasibility of obtaining repeatable DTI parameters along the entire cervical and thoracic spinal cord as a function of age in typically developing pediatric subjects; to analyze the DTI parameters among different transverse levels of the cervical and thoracic spinal cord; and to examine the sex differences in DTI parameters along the cervical and thoracic spinal cord.
MATERIALS AND METHODS: Twenty-two subjects underwent 2 identical scans by using a 3T MR imaging scanner. Axial diffusion tensor images were acquired by using 2 overlapping slabs to cover the cervical and thoracic spinal cord. After postprocessing, DTI parameters were calculated by using ROIs drawn on the whole cord along the entire spinal cord for both scans.
RESULTS: An increase in fractional anisotropy and a decrease in mean diffusivity, axial diffusivity, and radial diffusivity were observed with age along the entire spinal cord. Significantly lower fractional anisotropy and higher mean diffusivity values were observed in the lower cervical cord compared with the upper cervical cord. Axial diffusivity values in the cervical cord were higher compared with the thoracic cord. No statistically significant sex differences were observed for all DTI parameters. There was a moderate-to-strong repeatability for all DTI parameters.
CONCLUSIONS: This study provides an initial understanding of DTI values of the spinal cord relevant to age and sex and shows that obtaining repeatable DTI values of the entire cord in children is feasible.
ABBREVIATIONS:
- AD
- axial diffusivity
- FA
- fractional anisotropy
- MD
- mean diffusivity
- RD
- radial diffusivity
- SC
- spinal cord
DTI is a valuable technique that measures the diffusivity of water molecules in tissues and provides in vivo information about tissue integrity and structure that is not apparent with conventional MR imaging. The commonly used DTI measures are fractional anisotropy (FA), which describes the degree of directional dependence; mean diffusivity (MD), which expresses the magnitude of the diffusion; axial diffusivity (AD), which represents water diffusivity parallel to the axonal fibers; and radial diffusivity (RD), which represents water diffusivity perpendicular to the axonal fibers.1 DTI studies have investigated the cervical spinal cord (SC) in healthy adult subjects and have shown that DTI parameters are sensitive to age-related microstructural changes.2,3 In contrast to the studies in adults, 1 study has evaluated the age-dependent evolution of FA and ADC values of the pediatric cervical SC.4 There are no studies, to date to our knowledge, describing the age-related changes in the DTI parameters along the entire cervical and thoracic SC in pediatric subjects.
The usefulness of DTI has been shown for examining the cervical SC in pediatric subjects.5⇓–7 However, a comprehensive study examining the diffusion characteristics and repeatability of DTI along the cervical and thoracic SC in pediatric subjects has not been reported. On the basis of the promising outcomes of previous pediatric studies and owing to the growing practice of obtaining DTI in clinical patients,7,8 it is essential to establish normative pediatric DTI values along the cervical and thoracic SC to aid in the clinical interpretation of the injured SC.
The purpose of this study was 3-fold: to investigate the feasibility of obtaining repeatable DTI parameters along the entire cervical and thoracic SC as a function of age in typically developing pediatric subjects by using an inner FOV DTI sequence; to analyze the DTI parameters among different levels of the cervical and thoracic SC; and to examine the sex differences in the DTI parameters along the cervical and thoracic SC.
Materials and Methods
Study Design
This initial study was part of a larger research effort to develop and validate DTI as a method for assessing the SC in children, similar to others that are working to validate DTI of the brain. The validation requires examining a population of subjects that have core variables such as age, sex, race, ethnicity, handedness, and education levels for any normative data base.9 Twenty-two typically developing subjects (mean age, 11.44 ± 3.07 years) were recruited. There were 8 males (mean age, 10.51 ± 3.31 years) and 14 females (mean age, 11.97 ± 2.92 years). These subjects were recruited mostly through our colleagues or family members. A few typically developing children were recruited as siblings of some of our spinal cord injury population. Thirteen subjects were excluded during the recruitment for the following reasons: Three declined, 7 had scheduling conflicts, and 3 had metal piercings or permanent retainers. Subjects were included in the study if they had no evidence of spinal cord injury or pathology, as assessed by performing a neurologic screen (a questionnaire about their medical history associated with neurologic injuries and a brief assessment of motor and sensory function and reflexes). Subjects were excluded from the study if they were unable to tolerate MR imaging without sedation or had infantile, juvenile, or adolescent idiopathic scoliosis or any abnormality of the nervous and/or musculoskeletal system. No subjects with orthodontic hardware were included in this study. Written informed assent (child) and consent (parent) were obtained under the protocol approved by the Temple University institutional review board.
MR Imaging Protocol
The scans were performed by using a 3T Verio MR imaging scanner (Siemens, Erlangen, Germany) with a 4-channel neck matrix and an 8-channel spine matrix coil. The protocol consisted of an initial gradient-echo T2WI scout sagittal scan of the cervical and thoracic SC. The sagittal images were used to prescribe axial sections of the cervical and thoracic SC. Next, axial gradient-echo T2WI; a sagittal T2WI 3D SPACE sequence (sampling perfection with application-optimized contrasts by using different flip angle evolution; Siemens, Erlangen, Germany); sagittal TSE T1WI; sagittal TSE T2WI; and axial DTI scans were obtained.
DTI was performed by using an inner FOV sequence. The inner FOV sequence was based on a single-shot echo-planar imaging sequence for DWI with 2D-selective radiofrequency excitations, which improve the SNR and reduce geometric distortions in SC imaging, described in detail elsewhere.5,10 Manual shim volume adjustments were also performed before data acquisition to restrict the adjustment volume to the anatomy of interest as much as possible.
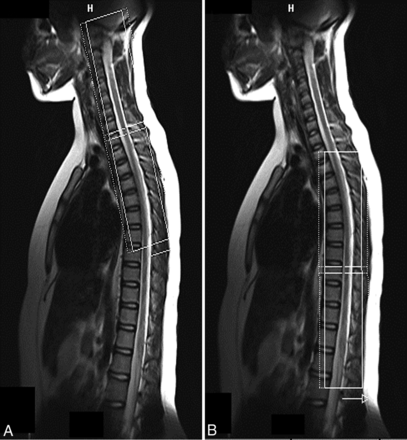
Diffusion tensor images were acquired axially by using 2 overlapping slabs, in the same anatomic location prescribed for the T2 gradient-echo images, to cover the cervical (C1-upper thoracic region) and thoracic (upper thoracic-L1) SC (Fig 1). The imaging parameters for each slab of DTI acquisition included the following: FOV = 164 mm, phase FOV = 28.4% (47 mm), 3 averages of 20 diffusion directions, 6 B0 acquisitions, b=800 s/mm2, voxel size = 0.8 × 0.8 × 6 mm3, axial sections = 40, TR = 7900 ms, TE = 110 ms, acquisition time = 8:49 minutes. To test the repeatability of DTI, we imaged all subjects twice by using the same protocol within 2 hours following the initial scan. Anesthesia was not administered. Cardiac and respiratory gating was not used, to keep scan times as short as possible.
Localization image from which the axial cervical (A) and thoracic (B) SC sections are prescribed. The inner solid white rectangular line represents the inner FOV, which was oversampled (dotted white line) to avoid aliasing.
Image Preprocessing and Analysis
Initially, a central mask was applied to the raw DTI to eliminate the anatomy outside the SC. A mean B0 image was calculated, generated from the coregistration of all 6 B0 acquisitions. The diffusion-weighted images were corrected for motion by using a rigid-body-correction algorithm.11 After motion correction, tensor estimation was done on a voxel-by-voxel basis from the axial DTI. All processing was performed by using in-house software developed in Matlab (MathWorks, Natick, Massachusetts). For robust diffusion tensor estimation, a nonlinear fitting algorithm implemented in RESTORE technique was used.12
Each ROI was manually drawn on the whole cord on gray-scale FA maps at every axial section, on the basis of the methodology from the previous article,5 along the cervical and thoracic SC for both scans. For drawing the ROI, sections corresponding to the anatomic regions with accurate vertebral levels were defined for the cervical and thoracic SC by a board-certified pediatric neuroradiologist. The placement of ROIs was performed by a neuroscientist (S.S.) and confirmed by a neuroradiologist (S.H.F). There was a consistent sparing of the outer margin of the cervical and thoracic SC of approximately 1-voxel width to minimize volume averaging with the CSF.
In this study, the 22 subjects were divided into the following age groups: 6–11 years (n = 12) and 12–16 years (n = 10) based on similar standards and age definitions used for reporting spinal cord injury in the pediatric population.13 DTI parameters were quantified at each intervertebral disc level and at the mid-vertebral body level of the cervical and thoracic SC in all subjects. DTI parameters were compared for the cervical and thoracic SC as a function of age and among different levels of the cervical and thoracic SC. To study the changes in diffusion parameters among different levels of the cervical and thoracic SC, we averaged DTI values across the following levels per subject: upper cervical cord (C1, mid-dens, base dens, mid-C2, C2–C3, mid-C3), middle cervical cord (C3–C4, mid-C4, C4–C5, mid-C5), and lower cervical cord (C5–C6, mid-C6, C6–C7, mid-C7, C7–T1), upper thoracic cord (mid-T1, T1–T2, mid-T2, T2–T3, mid-T3, T3–T4, mid-T4, T4–T5), middle thoracic cord (mid-T5, T5–T6, mid-T6, T6–T7, mid-T7, T7–T8, mid-T8, T8–T9), and lower thoracic cord (mid-T9, T9–T10, mid-T10, T10–T11, mid-T11, T11–T12, mid-T12, T12–L1, mid-L1). The mean values were used for comparison among the upper, middle, and lower cervical and thoracic SC across all subjects.
Statistical Analysis
Means with SD were calculated for DTI parameters according to age groups, cord levels, and sex. The coefficient of variation was calculated as an additional measure of variability. Because the DTI parameters failed the Shapiro-Wilk test for normality, the data were transformed into “normalized ranks” to allow parametric testing.14 The ANOVA for repeated measures and linear regression was used to compare scans, cord levels, and age. A P value of .05 was used throughout to determine statistical significance. Test-retest repeatability was calculated by using the intraclass correlation coefficient according to the method of Shrout and Fleiss.15 All statistical analyses were performed with SAS, Version 9.4 (SAS Institute, Cary, North Carolina).
Results
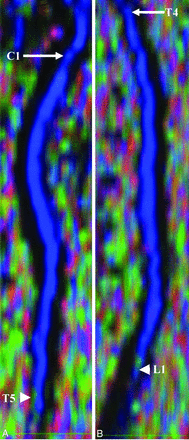
All 22 subjects tolerated 2 repeat imaging sessions. Each session was 40–45 minutes in duration with a 2-hour delay between sessions. Imaging with the inner FOV sequence with 2D-selective radiofrequency excitations resulted in DTI with high spatial resolution. Better distinction between WM and GM structures was observed throughout the cord; however, in general, the GM-WM differentiation was better in the cervical and upper thoracic cord (Fig 2). In Fig 2, the green anisotropy in the fourth row might represent some spurious voxels; and in the eighth row, the conus is present in the imaging volume and the artifacts seen are largely due to partial volume averaging and complex CSF flow. Figure 3 shows the sagittal reconstruction of FA color maps of 2 overlapping slabs of the same subject in Fig 2. Figure 3A shows the cervical and upper thoracic region (C1 to T5), and Fig 3B shows the upper thoracic through the conus region (T4 to L1).
Axial FA color maps of each intervertebral disc level and within the mid-vertebral body level along the entire cervical and thoracic SC in a typically developing subject (C1 to T12–L1 level). An example of GM at the mid-C5 level is shown with an arrow, and WM, with an arrowhead.
Sagittal reconstruction of FA color maps of 2 overlapping slabs of the same subject as shown in Fig 2. A, The cervical and upper thoracic regions (C1, arrow, to T5, arrowhead). B, The upper thoracic-through-conus regions (T4, arrow, to L1, arrowhead).
DTI Parameters along the Cervical and Thoracic SC According to Age
Table 1 summarizes the DTI values for 2 age groups. FA values were low in subjects 6–11 years of age, while they were high in subjects 12–16 years of age along the cervical and thoracic SC. MD, AD, and RD values followed an opposite trend and were high in subjects 6–11 years of age and low in subjects 12–16 years of age. The increase in FA values from age groups 6–11 years to 12–16 years was statistically significant (P = .0193).
Means of FA, MD, AD, and RD along the cervical and thoracic SC according to age groups
As shown in Table 1, MD decreased significantly from age groups 6–11 years to 12–16 years (P < .0001). There was a significant decrease in AD from age groups 6–11 years to 12–16 years (P < .0001). Similarly, RD decreased significantly from age groups 6–11 years to 12–16 years (P < .0001) (Table 1).
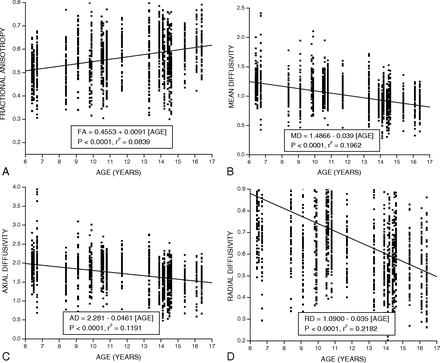
A simple linear regression analysis was used to measure the effect of age on DTI parameters. As seen from Fig 4, an increase in FA was observed; however, MD, AD, and RD showed a decrease along the cervical and thoracic SC with age.
Linear fit plots showing the relationship among FA (A), MD (B), AD (C), and RD (D) and age along the cervical and thoracic SC (C1 to mid-L1) in typically developing pediatric subjects.
DTI Parameters According to Cord Levels
Table 2 shows that FA values in the upper cervical cord and middle cervical cord were significantly higher compared with the lower cervical cord (P < .0001). No statistically significant difference in FA values was observed in the upper cervical cord compared with the middle cervical cord (P = .3732). In the upper thoracic cord, FA values were significantly lower compared with the middle thoracic cord (P < .0001), while they were significantly higher compared with the lower thoracic cord (P < .0001). FA values in the middle thoracic cord were significantly higher compared with the lower thoracic cord (P < .0001) (Table 2).
Means of FA, MD, AD, and RD according to different levels of the cervical and thoracic SC
A significant decrease in MD values was observed in the upper cervical cord and middle cervical cord compared with the lower cervical cord (P < .0001, P = .0265). No statistically significant difference in MD values was observed in the upper cervical cord compared with the middle cervical cord (P = .1357). In the middle thoracic cord, a significant increase in MD values was observed compared with the upper thoracic cord and lower thoracic cord (P < .0001). No statistically significant difference was observed in the upper thoracic cord compared with lower thoracic cord (P = .2665) (Table 2).
AD values in the upper cervical cord were significantly higher compared with the upper thoracic cord (P < .0001) and lower thoracic cord (P < .0001). No significant difference was observed in the upper cervical cord compared with middle thoracic cord (P = .1485). In the middle cervical cord, AD values were significantly higher compared with the upper thoracic cord (P < .0001), middle thoracic cord (P = .0033), and lower thoracic cord (P < .0001). AD values in the lower cervical cord were significantly higher compared with the upper thoracic cord (P < .0001), middle thoracic cord (P = .0158), and lower thoracic cord (P < .0001) (Table 2).
RD values in the upper cervical cord were not statistically significant compared with the upper thoracic cord (P = .2707), middle thoracic cord (P = .1044), and lower thoracic cord (P = .9320). Significant differences in RD values were observed in the middle cervical cord compared with the upper thoracic cord (P = .0198) and middle thoracic cord (P = .0048). No statistically significant difference was observed in the middle cervical cord compared with lower thoracic cord (P = .1774). A significant increase in RD values was observed in the lower cervical cord compared with the upper (P < .0001), middle (P < .0001), and lower thoracic cord (P < .0001) (Table 2).
Coefficient of Variation
The coefficient of variation represents the SD as a percentage of the mean and is an additional indication of the variability of the DTI measurements. The coefficient of variation showed that the variability of the DTI parameters was very good for both age groups and similar to other reports of DTI measurement variability.16 Overall, FA showed the least variability (6–11 years, 16.66%; 12–16 years, 17.24%).
DTI Parameters and Sex
DTI parameters did not show any statistically significant difference between males (FA: 0.56 ± 0.09; MD: 1.07 ± 0.29; AD: 1.80 ± 0.43; RD: 0.71 ± 0.25) and females (FA: 0.56 ± 0.10; MD: 1.01 ± 0.25; AD: 1.71 ± 0.39; RD: 0.66 ± 0.22) along the cervical and thoracic SC.
Test-Retest Repeatability
Table 3 shows the intraclass correlation coefficient and 95% CIs for test-retest repeatability of DTI values for the cervical and thoracic SC. There was a moderate-to-strong repeatability for DTI values for the cervical and thoracic SC (Table 3).
Test-retest repeatability for each DTI parameter per the cervical and thoracic SC
Discussion
In this study, the entire SC of 22 typically developing children was successfully scanned to obtain DTI parameters (FA, MD, AD, and RD) that revealed location and age- and sex-related trends and moderate-to-strong repeatability. To the best of our knowledge, this study is the first to examine developmental and sex differences in DTI parameters of the entire pediatric SC. One previous study did examine FA and ADC changes in the pediatric cervical spine,4 with findings similar to those in our study.
The results of this study showed that with increasing age, FA increased along the cervical and thoracic SC. Studies in the brain have reported age-related changes in the DTI parameters to describe the normal developmental characteristics from infancy to adulthood.17 In addition, it is proposed that changes in DTI parameters in the brain as a function of age may result from decreasing water content, myelination of fiber tracts, and the thickening diameter of fiber tracts.18 The SC is an extension of the brain, transmitting messages from the brain to the periphery and vice versa; and similar physiologic processes may occur in the SC. It has been reported that myelination in the SC starts during the second half of fetal life, peaks during the first year postnatally, and continues until 20 years of age.19 In our study, an increase in FA with age in the SC was observed, similar to the FA findings in the brain, which suggests the ongoing process of myelination.
In our study, MD, AD, and RD decreased with age along the cervical and thoracic SC. MD is the average of all 3 eigenvalues and represents a variety of physiologic processes that include continuing myelination, increased organization, and compactness of the axonal bundles. It has been demonstrated that there is a strong correlation between AD/RD and histology in animal studies.20 AD in the WM tracts correlates well with the integrity of axons in these tracts, while RD in the WM tracts correlates with the amount of myelin.20 Decreased AD with age may result from increased axonal density or axonal caliber, leading to an addition of diffusion barriers parallel to the axon due to reduced interaxonal space. Decreased RD with age may be due to continuing myelination because thickening of the myelin sheath will reduce perpendicular water diffusion.
In addition to age-related changes, our study demonstrated that diffusion characteristics are not consistent throughout the SC and are dependent on the cord level. The SC is greater in width and diameter in the cervical and lumbosacral regions, forming the cervical plexus and the lower thoracic lumbar–sacral plexus, the site of neurons that primarily supply the extremities.21 These neurons represent GM that is higher in concentration compared with the remainder of the cord.21 In the SC, the GM is a central butterfly-shaped structure surrounded by the WM. Additionally, the volume of WM and GM is different depending on the location within the SC. Goto and Otsuka22 reported that the GM volume, on average, represents 18% of the SC at the anatomic level at the origin of the C2-through-C8 nerve root (the cervical region). The volume of GM represents 13.2% of the SC at the anatomic level at the origin of the T1-through-T12 nerve root (the thoracic region). The amount of WM decreases gradually in the caudal direction because the long ascending and descending pathways contain fewer axons at successively more caudal levels of the SC.21
When comparing DTI parameters at different locations of the cervical and lower thoracic regions, our study demonstrated significantly lower FA and higher MD values in the lower cervical cord compared with the upper cervical cord. Another study demonstrated that at the upper cervical cord, the right-to-left diameter of the SC and the area of the central GM are smaller compared with the lower cervical cord.23 The relative lower cervical cord enlargement, which corresponds to the cervical plexus, is due to an increase in the GM/WM ratio. It has been reported that in adults, the central GM of the SC has lower FA and higher MD compared with the WM.24 In our study, the larger central GM area in the lower cervical cord may be responsible for decreased FA and increased MD compared with the upper cervical cord. Decreased FA values in the lower thoracic cord compared with the upper thoracic cord may be due to the variations in the percentage of GM within different levels of the thoracic cord.
Diffusion in the healthy SC is highly anisotropic with transverse ADC values less than longitudinal ADC values.25 Anisotropy in the SC is due to diffusion barriers encountered as water moves perpendicular to the fibers. These barriers, such as the cell membrane and myelin sheath, result in low transverse ADC values. As water diffuses longitudinally in the SC, these diffusion barriers are not encountered and hence longitudinal ADC is higher.25 Our study also demonstrated higher AD values in the cervical and thoracic cord compared with RD values. It has been reported that the density and integrity of cytoskeletal proteins (microtubules and neurofilaments) affect longitudinal water diffusion.26 Schwartz et al27 demonstrated a positive correlation of longitudinal ADC with axon diameter in the normal rat cervical cord and explained that this was due to the inverse relationship between axon caliber and cytoskeletal protein density. These findings may explain our results of increased AD values in the upper, middle, and lower cervical cord, which may contain a higher percentage of large-diameter axons compared with the upper, middle, and lower thoracic cord. Because large axons have a decreased density of microtubules and neurofilaments, this may result in increased longitudinal water diffusion in the cervical cord. It has been observed in ex vivo studies that increased axonal diameter and decreased axonal density correlate with increased transverse diffusion in SC tracts.28 In our study, larger axons in the lower cervical cord may explain the increased RD values compared with the thoracic cord.
DTI has proved to be a useful tool for examining sex differences in the brain WM during childhood and adolescence29; however, in the SC, these are not well-studied. We found no significant sex differences, when comparing males and females, along the cervical and thoracic SC. Our results are consistent with the findings of previous DTI studies of the maturing cervical SC4 and brain maturation,30 which also did not show any sex differences. These sex-related DTI findings in the SC need to be further investigated to determine whether they hold up with larger sample sizes and, if so, to produce normative data for males and females of different ages.
As we have shown previously in the cervical SC, there was good-to-strong repeatability in DTI values.5,8 It has also been reported that in adult cervical SC DTI, there is good intra- and interobserver variability.16 In our current study, we have shown that within the cervical and thoracic SC, DTI values showed moderate-to-strong repeatability. These findings add to the existing evidence that DTI values for the normal SC are repeatable.
One of the limitations of the study was the use of manual ROI selection, which may have introduced partial volume contamination from the CSF surrounding the cord. The ROIs drawn were conservative, to minimize this volume-averaging effect. In our study, because the DTI measurements were performed on the whole cord, the inclusion of GM and WM in the analysis might have affected the DTI values.31 Currently, no automatic methods exist for delineating WM and GM from DTI SC images, and manual segmentation is cumbersome and time-consuming. Therefore, for future analysis, an automated or semiautomated segmentation method is required for accurately delineating the GM and WM in the SC. Another limitation of this study is the low number of subjects within different age groups and sex. Future work with more subjects is needed in these subgroups to establish normative DTI data of the pediatric cord. There was also heterogeneity of the signal in many subjects at the edge of the coil and in the lower thoracic region, which potentially can be reduced with improved imaging techniques. In addition, we chose a section thickness of 6 mm to allow a maximal SNR of the imaging voxel while balancing the in-plane resolution and the number of sections needed to scan the subject within a clinically acceptable time of acquisition per slab. This limitation could be overcome in the future by imaging the SC at a higher field strength, with improved radiofrequency coils and multiband DTI techniques, which will allow imaging small voxels while still maintaining a relatively short imaging time.
Conclusions
This study provides an initial understanding of DTI values of the SC relevant to age and sex and shows that obtaining repeatable DTI values of the entire cervical and thoracic cord in children is feasible.
Acknowledgments
We thank Mahdi Alizadeh for helping with the manuscript preparation.
Footnotes
Disclosures: Scott H. Faro—RELATED: Grant: National Institutes of Health–National Institute of Neurological Disorders and Stroke.* John Gaughan—RELATED: Consulting Fee or Honorarium: Temple University, National Institutes of Health R01 NS079635 (statistical consulting). Mary Jane Mulcahey—RELATED: Grant: National Institute of Neurological Disorders and Stroke *; Support for Travel to Meetings for the Study or Other Purposes: National Institute of Neurological Disorders and Stroke *; UNRELATED: Board Membership: President, American Spinal Injury Association, no payment; Vice President, Pediatric Spine Foundation, no payment; Consultancy: Topics in Spinal Cord Rehabilitation, Comments: Associate Editor; Grants/Grants Pending: Community Health Network Foundation,* Department of Defense,* Shriners Hospitals*; Royalties: Mac Keith Press (editor, book); Travel/Accommodations/Meeting Expenses Unrelated to Activities Listed: Boston Rehabilitation Outcomes Center (Visiting Scientist, 2015–2016). Feroze B. Mohamed—RELATED: Grant: National Institutes of Health–National Institute of Neurological Disorders and Stroke.* *Money paid to the institution.
This work was supported by National Institute of Neurological Disorders of the National Institutes of Health under award No. R01NS079635 (F.B. Mohamed and M.J. Mulcahey, Principal Investigator).
Indicates open access to non-subscribers at www.ajnr.org
References
- Received February 11, 2016.
- Accepted after revision May 23, 2016.
- © 2016 by American Journal of Neuroradiology