Abstract
Summary: We report on MR studies of a 15-year-old girl with moyamoya disease in whom diffuse leptomeningeal enhancement (“ivy sign”) was revealed by fluid-attenuated inversion-recovery (FLAIR) and contrast-enhanced imaging. We speculate that the mechanism behind this enhancement is caused by a retrograde slow flow of engorged pial vasculature via leptomeningeal anastomosis. Nevertheless, it remains unknown whether the precise source of a high signal on FLAIR images is attributable to pial vessels themselves or congested thickening of the leptomeninges or both.
The “ivy sign” refers to diffuse leptomeningeal enhancement found on postcontrast MR images of patients with moyamoya disease in childhood (1). It is considered to represent the diffuse engorgement of the pial vasculature (1). In this study, we found that the ivy sign is demonstrated on fluid-attenuated inversion-recovery (FLAIR) MR images as well as on postcontrast MR images in a child with moyamoya disease.
Case Report
A 15-year-old girl experienced transient motor weakness of the upper and lower left extremities, particularly when she played the trumpet at school. Physical examination revealed no abnormal neurologic findings.
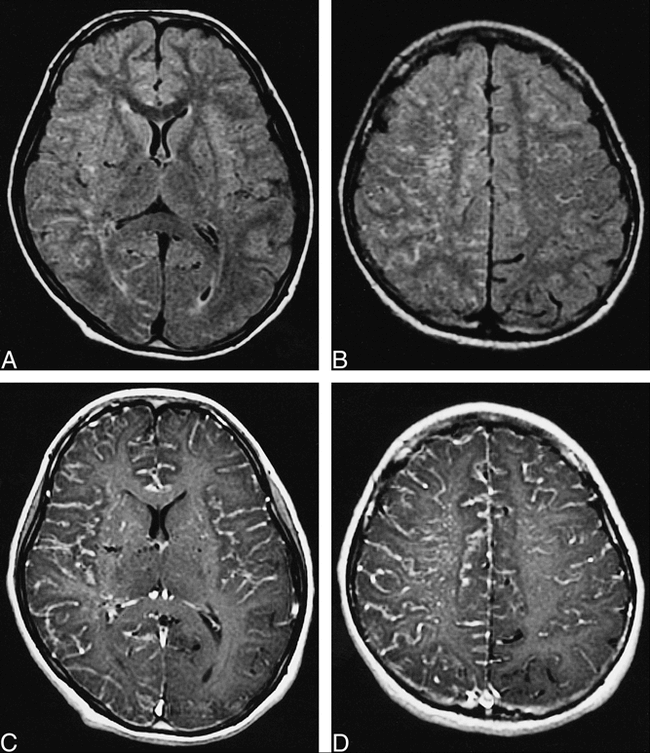
The precontrast T1-weighted images showed dilatation of perforating arteries in the bilateral basal ganglias. On T2-weighted images, no flow voids of middle cerebral arteries (MCA) were seen in bilateral sylvian fissures, and prominent enlargement of perivascular space was present, particularly in the deep white matter on the right side. FLAIR MR images revealed extensive areas of increased signal intensity, bilaterally, within the subarachnoid spaces and perivascular spaces (Fig 1A–B). Contrast-enhanced MR images showed diffuse leptomeningeal enhancement along the cortical sulci and strong enhancement of perforating arteries in the basal ganglia and deep white matter (Fig 1C–D). The right cerebral hemisphere displayed contrast enhancement more prominently than the left cerebral hemisphere. The areas of enhancement were consistent overall with those of abnormal high signal intensity on FLAIR images, although they were more extensive on contrast-enhanced MR images than on FLAIR images. A right internal carotid angiogram revealed narrowing of the terminal portion of the internal carotid artery, basal moyamoya in evolution, and poor opacification of distal territories of the anterior cerebral artery (ACA) and the middle cerebral artery (MCA). Analogous findings were seen in the left internal carotid angiogram. A right vertebral angiogram showed a substantially intact left posterior cerebral artery (PCA) and narrowing of the right PCA. It also showed marked retrograde filling in the area of bilateral ACAs and MCAs via leptomeningeal anastomosis. Cortical branches of the ACAs and MCAs were depicted poorly, especially on the right side. The angiographic stage was determined to be stage 3 according to the Suzuki and Takaku classification (2). The patient was diagnosed with moyamoya disease and was conservatively followed up after discharge.
Fifteen-year-old girl with transient motor weakness of upper and lower left extremities.
A and B, FLAIR images (10000/126/2, TI = 2200) show extensive areas of increased signal intensity in leptomeninges and perivascular spaces, predominantly in right hemisphere.
C and D, Contrast-enhanced T1-weighted images (333/14/2, no use of flow compensation) show diffuse leptomeningeal enhancement and strong enhancement of perforating arteries in basal ganglias and deep white matter. Areas of left PCA and some areas of left MCA are spared leptomeningeal enhancement.
Discussion
Leptomeningeal enhancement on postcontrast T1-weighted images has been reported in association with meningitis (3), meningeal carcinomatosis (4), and angitis (5). Common among these diseases is pathologic inflammation or carcinomatous infiltration of the leptomeninges, with or without involvement of the small leptomeningeal vessels (5).
Recently, Ohta et al (1) have reported that diffuse leptomeningeal enhancement was seen on postcontrast T1-weighted images of children with moyamoya disease. The authors named this finding on postcontrast MR images the “ivy sign,” because it resembles ivy creeping on stones. Because a markedly developed diffuse pial arterial network was observed over the cortex when a superficial temporal artery MCA anastomosis was performed, they concluded that the leptomeningeal enhancement represents fine neovascularization over the cerebral cortex. Collateral circulation via leptomeningeal anastomosis plays an important role at some stages of moyamoya disease. Because this collateral flow is supposed to be very slow due to its retrograde direction via leptomeningeal anastomosis, it is likely that such an engorged pial network would show enhancement after injection of contrast media. Essig et al (6) have reported that vascular enhancement at the cortex was seen also in patients with stenosis or occlusion of the internal carotid artery without infarction; among these patients, slow retrograde flow via leptomeningeal collateral arteries was the most important pathophysiologic correlate.
FLAIR MR imaging has become widely used in the clinical setting for cerebral lesions. In our hospital, FLAIR is used routinely for brain MR examinations. Recently, Noguchi et al (7) have reported that the distal portion of the cerebral artery was visible as an area of high signal intensity on FLAIR images in acute cerebral infarction. In two of their nine patients, contrast-enhanced T1-weighted imaging was performed also, and the arterial high signal on FLAIR images was consistent overall with the area of arterial enhancement. They speculated that markedly slow retrograde flow via leptomeningeal anastomoses is the primary source for the arterial high signal seen on FLAIR MR images (7). In our patient, angiography showed retrograde filling via leptomeningeal anastomosis in ACAs and MCAs—findings that are in accord with those of Noguchi et al. The left PCA was substantially intact on angiography, and contrast-enhanced and FLAIR images showed that in the area of the left PCA, and some areas of the left MCA proximal to those of the left PCA, the ivy sign was absent or less conspicuous. These findings suggest that a high signal on FLAIR in leptomeningeal areas reflects a retrograde slow flow of engorged pial arteries via leptomeningeal anastomosis. Although the mechanism of the ivy sign is likely to flow slowly in collateral vessels, the precise source of a high signal on FLAIR images may be complex. It seems reasonable that engorged pial vessels themselves are the source of a high signal. Congested thickening of the leptomeninges, however, potentially could be another source of a high signal.
It remains unknown whether the ivy sign is demonstrated in every case of childhood moyamoya disease. In our case, the angiographic stage was determined to be stage 3 according to the Suzuki and Takaku classification. Hemodynamics of moyamoya disease vary according to the stages of disease development. Therefore, it is suggested that the ivy sign may be seen in limited stages of moyamoya disease. Further investigation of a larger number of clinical cases of moyamoya disease is necessary.
Diffuse leptomeningeal enhancement on contrast-enhanced MR images can disappear or decrease after successful bypass surgery and be accompanied by an increase in the number of flow voids (1). These findings suggest that the ivy sign in moyamoya disease represents cortical insufficiency rather than reactive hyperemia. In our case, the patient was followed up clinically, although no surgical procedure was performed; therefore, it remains unknown whether leptomeningeal hyperintensity on FLAIR images is reversible after bypass surgery.
Conclusion
The ivy sign was seen in a child with moyamoya disease on FLAIR as well as on contrast-enhanced images. We speculate that the mechanism arises from a retrograde slow flow of engorged pial vasculature via leptomeningeal anastomosis, although it remains unknown whether the precise source of a high signal on FLAIR images is caused by pial vessels themselves or congested thickening of the leptomeninges or both.
Footnotes
↵1 Address reprint requests to Masayuki Maeda, MD, Department of Radiology, Maizuru Kyosai Hospital, Azahama, Maizuru City, Kyoto 625-8585, Japan.
References
- Received January 21, 1999.
- Copyright © American Society of Neuroradiology