The location or topography of hypoperfusion is likely a critical determinant of outcome in acute ischemic stroke. The spatial distribution of a perfusion defect is directly influenced by the site of vascular occlusion and corresponding collateral flow. Such vascular localization is readily apparent on angiography and may also be evident on MR imaging as early vessel signs (EVS) or intravascular signal intensity derangements, including fluid-attenuated inversion recovery (FLAIR) vascular hyperintensity (FVH), and gradient-echo susceptibility (GRE SVS). Although similar in appearance, proximal and distal EVS may represent different pathophysiologic substrates.
Schellinger et al (1) provide an important analysis on the diagnostic and prognostic value of such relatively subtle EVS in hyperacute stroke cases treated with intravenous thrombolysis. They explicitly note that their study is unique, because “no clinical study to date has taken thrombus composition as being reflected by GRE (and FLAIR) images and its role for rtPA [recombinant tissue plasminogen activator] response into account” (p. 623). Similar to prior reports of EVS, the authors combine proximal and distal EVS as a single entity. Proximal and distal EVS are both associated with perfusion abnormalities induced by proximal arterial occlusion or thrombosis, yet distal EVS may not be due to the MR imaging signal intensity characteristics of thrombus.
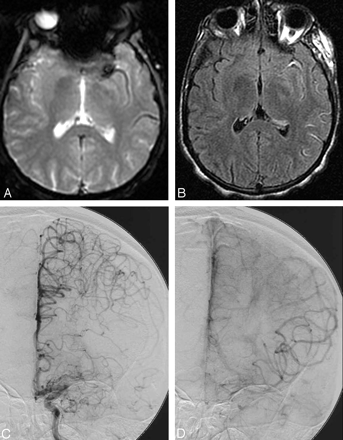
Distal EVS may be associated with retrograde collateral flow distal to thromboembolic occlusion of a proximal artery (Fig 1). Distal EVS are typically manifest in middle cerebral artery occlusion as serpiginous structures in the sylvian fissure extending distally through the convoluted architecture of the cerebral sulci. It remains highly unlikely and unproven that such intravascular signal intensity derangements are due to thrombus extending along the entire course of the middle cerebral artery. Contemporaneous angiography may reveal patency of these vascular segments filled by slow, retrograde collateral perfusion via leptomenigeal anastomoses (Fig 1). FVH likely represents exceedingly slow collateral flow induced by retrograde perfusion of an arterial tree with diminished intravascular pressure. Distal EVS on gradient echo, or hypointensity in distal segments, may be associated with intravascular deoxygenation of collateral blood due to precapillary oxygen loss (2). The appearance of such distal EVS on gradient echo is dissimilar to GRE SVS described in the report by Schellinger et al (Fig 1A). Intravascular gradient-echo hypointensity in distal segments rarely demonstrates the prominent, and frequently coexistent, blooming artifact apparent as GRE SVS due to proximal thrombosis.
The authors also explore the enticing hypothesis that clot burden evident on MR imaging as manifest by EVS may be associated with thrombolytic efficacy. In contrast to endovascular reperfusion techniques using mechanical clot disruption, intravenous thrombolysis is dependent on the extent of clot surface that is exposed to the lytic agent at only the proximal and distal ends of the clot. Clot location, not necessarily clot burden, may influence thrombolytic exposure to the distal clot surface via collaterals. Conversely, proximal diversion of blood flow to collateral routes may also diminish forward pressure on the clot, hindering recanalization of thromboembolic occlusion. Clot location and corresponding collateral routes may therefore be critical variables in assessing not just tissue perfusion, but also thrombolytic efficacy in acute stroke.
EVS on MR imaging may provide valuable information regarding collaterals, not just thrombosis. Conventional perfusion MR imaging and MR angiography may be used for detection of proximal arterial occlusion, yet selective techniques such as regional perfusion MR imaging may more reliably delineate collateral flow (3). Until such noninvasive techniques are refined, angiography will remain the gold standard for defining the location of proximal occlusion and corresponding collaterals.
The location and not the mere presence of EVS on MR imaging may disclose distinct vascular correlates. In acute ischemic stroke, it is imperative to consider the location of proximal flow cessation, the location of thrombus, and the location of compensatory collateral flow.
Contemporaneous MR imaging and angiography in acute left middle cerebral artery stroke. EVSs due to proximal thrombosis and distal collateral flow are evident on gradient-echo (A) and FLAIR (B). Angiography demonstrates proximal arterial occlusion (C) and slow, retrograde collateral flow (D).
- Copyright © American Society of Neuroradiology