Abstract
BACKGROUND AND PURPOSE: A malformed corpus callosum carries a risk for abnormal neurodevelopment. The advent of high-frequency transducers offers the opportunity to assess corpus callosum development in early pregnancy. The aim of the study was to construct a reference chart of the fetal corpus callosum length on ultrasound between 13 and 19 weeks of gestation and to prospectively examine growth patterns in pathologic cases.
MATERIALS AND METHODS: We performed a prospective cross-sectional study between 2020 and 2022 in well-dated, low-risk, singleton pregnancies between 13 and 19 weeks of gestation. A standardized image was obtained in the midsagittal plane. Imaging criteria were used as a confirmation of the early corpus callosum. Measurements were taken by 4 trained sonographers. Intra- and interobserver variability was assessed. Corpus callosum length in centiles were calculated for each gestational week.
RESULTS: One hundred eighty-seven fetuses were included in the study. All cases met inclusion criteria. At 13 weeks of gestation, the margins of the early corpus callosum were sufficiently clear to be measured in 80% (20/25) of fetuses. A cubic polynomial regression model best described the correlation between corpus length and gestational age. The correlation coefficient (r2) was 0.929 (P < .001). Intra- and interobserver variability had high interclass correlation coefficients (>0.99). Presented is the earliest published case of agenesis of corpus callosum and a case of dysgenetic corpus callosum in Rubinstein-Taybi syndrome.
CONCLUSIONS: Provided is a nomogram of the early fetal corpus callosum. Applying imaging criteria helped to identify a case of complete agenesis of the corpus callosum as early as 14 weeks.
ABBREVIATIONS:
- CC
- corpus callosum
- GA
- gestational age
The corpus callosum (CC) is a commissural structure composed of neuronal fibers crossing the midline, connecting both hemispheres.1⇓-3 Agenesis and dysgenesis of the corpus callosum carry risk for abnormal neurodevelopment.4⇓-6 A short CC may be the first clue of abnormal formation. Normal CC growth charts are available from 16 weeks of gestation.7⇓⇓⇓⇓⇓⇓-14 None are provided for earlier weeks. The advent of high-frequency transducers offers the opportunity to bring forward anomaly screening to the first trimester.15⇓⇓⇓⇓-20 A recent publication depicted the early development of the CC on sonography, between 14 and 17 weeks;21 however, whether at 13 weeks of gestation the commissural fibers of the CC have developed and can be demonstrated is still debated.3,21,22 The aims of the study were the following: 1) to construct a normal modeled reference chart of the length of the fetal CC on 2D ultrasound between 13 and 19 weeks of gestation, 2) to propose imaging criteria to support identification of the early CC, and 3) to prospectively examine the growth of the CC in suspected or confirmed pathologic cases.
MATERIALS AND METHODS
A prospective cross-sectional study was conducted between 13 and 19 weeks of gestation at a single tertiary care center (Chaim Sheba Medical Center). Inclusion criteria were the following: 1) singleton pregnancies, 2) accurate pregnancy dating using first trimester crown-rump length or a history of regular menses, 3) absence of known major fetal malformations or genetic aberrations, and 4) absence of conditions known to affect fetal growth.
Measurements were taken by 4 examiners, Eran Kassif, T.W, A.M., and E.H. using a Voluson E10 ultrasound machine (GE Healthcare) with an abdominal RM6C 2–6 MHz convex probe or a vaginal RIC 6–12 MHz probe (both probes, GE Healthcare) as appropriate. A transvaginal approach was used up to 17 weeks of gestation, and a transabdominal approach, from 18 weeks in a nonvertex presentation.
To acquire a standardized image, we obtained a midsagittal plane of the fetal brain through the anterior fontanelle. The image was magnified so that the fetal head would occupy 70% of the screen. The probe was angled until the CC was horizontal with clear edges. The anterior-posterior length of the CC was measured.
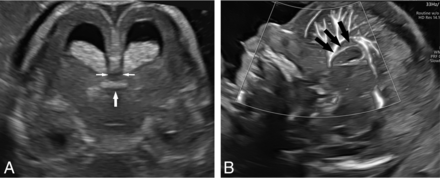
The identification of the early CC was supported by 3 imaging criteria: 1) the appearance of a hypoechogenic structure, 2) crossing the midline of the brain, and 3) located beneath the pericallosal artery and above the echogenic tela choroidea (Fig 1 and Online Videos 1 and 2). The pericallosal artery was demonstrated using color Doppler sonography. Slow Flow Doppler was used when the pericallosal artery was not clear or continuous on color Doppler.
Imaging criteria for early CC identification. A, A hypoechogenic structure (double arrows) crossing the midline of the brain above the echogenic tela choroidea (single arrow), located beneath the pericallosal artery (black arrows, B).
We performed an additional pilot study assessing the reproducibility of CC measurements. Fifty-nine fetuses were assessed for intraobserver variability, and 37, for interobserver variability. For intraobserver variability, 2 measurements were taken by the same operator on 2 different images. For interobserver variability, a second operator measured the CC length on a newly acquired image.
Postnatally, we contacted patients in whom the fetal corpus measurement was found to be ≤5th centile. This was identified in retrospect after the normal corpus growth chart was constructed. The patients were queried about the details of their pregnancy, delivery, postnatal period, and the development of the children, focusing on the Maternal Child Health Care Clinic visits.
Maternal Child Health Care Clinic Developmental Assessment
The Maternal Child Health Care Clinic is a national health care service in Israel that serves children from birth to 6 years of age, providing vaccines and conducting routine growth and developmental examinations. The Maternal Child Health Care Clinic assessments are performed regularly during scheduled visits at 1, 2, 4, 6, 9, 12, 18, 24, 36, 48, and 60 months of age. A structured age-based developmental assessment is performed using a standardized protocol addressing 4 developmental domains: gross motor, fine motor, language, and personal-social.23 This protocol was validated on a large Israeli pediatric population in a recent study.24
The study protocol was approved by Sheba Medical Center ethics committee (5344–18-SMC). All participating patients were informed and consented.
Statistical Analysis
Statistical analyses were performed using SPSS Statistics, Version 20 (IBM), and Excel 2016 software (Microsoft). All tests were 2-tailed, and a P value of <.05 was considered statistically significant. The intra- and interobserver variability was assessed using the Bland-Altman plot limits of agreement (SD, 1.96).25 The statistical analysis used to calculate the modeled centiles was based on a method previously described.26⇓-28
After assessment of normal distribution using the Shapiro-Wilk test, the fitted mean and SD were calculated using the regression models as a function of gestational age (GA) of the raw data (y = a + b × GA + b1 × GA2 + b2 ×GA3). The r2 statistic was studied to assess the best quality of fit.
The modeled centiles for a given GA were calculated as follows: centile GA = mean GA + K × SDGA, where K is the corresponding centile of the Gaussian distribution (for example, determination of the 10th and 90th centiles requires K = ±1.28; determination of the fifth and 95th centiles requires K = ±1.645, and so forth). An additional z score for assessing model fit was measured by the following formula: z score= (YGA – MGA) / SDGA. Normal distribution of the z scores using the Shapiro–Wilk W test was calculated as well.
RESULTS
One hundred eighty-seven fetuses of low-risk pregnancies were included. All cases met our strict inclusion criteria. Maternal characteristics are summarized in Table 1. The Pearson correlation coefficient showed a positive and significant correlation between CC length and GA (Table 2).
Maternal characteristicsa
CC length—correlation with fetal characteristics
Intra- and Interobserver Variability
A high interclass correlation was achieved for both intra- and interobserver variability: 0.997 (0.995–0.998, 95% CI) and 0.998 (0.996–0.999, 95% CI), respectively, indicating an excellent level of agreement.
The Nomograms
The raw data were best correlated with GA by a cubic polynomial regression formula. The correlation coefficient (r2) was 0.929 (P < .001). After we aggregated the mean and SD for each week, CC length was best fitted according to a cubic polynomial regression formula for the mean CC for GA in weeks.
Table 3 provides the predicted CC length in centiles for each GA. The mean absolute z score between measured and predicted values was 0.127 of 0. The Shapiro-Wilk test indicated a normal distribution with P values of .313.
Corpus callosum length (mm)a
Corpus Callosum during the 13th Week of Gestation
The appearance of a developing CC was sought in 25 fetuses at 13 weeks of gestation. A biometry concordant for 13 weeks was confirmed in all cases. Of these, in 80% (20/25), the early CC was clearly demonstrated and measured. In 5 cases, the margins of the CC were not clear enough for a precise measurement.
Demonstration of CC Growth
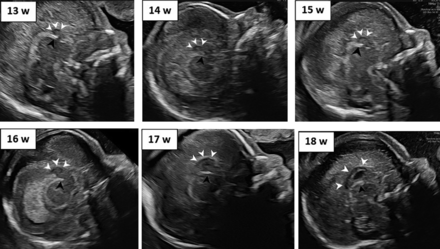
Figure 2 shows the gradual growth of the CC between 13 and 18 weeks. In each image, the CC and tela choroidea of the third ventricle are marked.
The growing early CC between 13 and 18 weeks. Marked in each image are the tela choroidea (black arrowhead) and CC (white arrowheads). W indicates weeks.
Short Early CC
We evaluated the outcome of 6 cases that were found to have a short CC (≤5th centile). In 2 of the cases, subsequent measurements of the CC during the second and third trimesters were consistently ≤5th centile as an isolated finding. Both patients refused further investigation due to personal religious beliefs. All patients were contacted postnatally, and they reported having had an uneventful pregnancy and delivery. Currently, these infants are between 7 and 15 months of age. Five infants are developing appropriately, as per the Maternal Child Health Care Clinic developmental assessment. A sixth infant, 7 months of age, was reported as healthy and normally developing by his parents and has never visited the Maternal Child Health Care Clinic, according to the parents’ preference. The medical record of this patient was reviewed, confirming normal physical and neurologic development by his pediatrician and also by attending physicians during a hospital admission for a viral infection.
Pathologic Cases.
By applying the imaging criteria, we identified 2 pathologic cases. We evaluated the sonographic appearance of the early fetal brains in these cases.
Absent Early CC
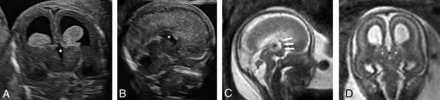
The patient was referred for an early anomaly scan at 14.4 weeks. The lateral ventricles appeared prominent, and the choroid plexuses were disproportionately small. On the coronal plane, the midline structure of the tela choroidea was not apparent, resulting in an unroofed third ventricle (Fig 3A). On the midsagittal plane, the hypoechoic structure of the early CC could not be demonstrated (Fig 3B). At 16 weeks, a fetal brain MR imaging confirmed the diagnosis of an absent CC (Fig 3C, -D). Repeat neurosonogram findings at 17 and 21 weeks remained consistent with the diagnosis. Chromosomal microarray and whole exome sequencing results were normal.
Absent early CC. A, In the coronal plane of the third ventricle and thalami, structures crossing the midline are not apparent, resulting in an unroofed third ventricle (star). B, In the midsagittal plane, a prominent unroofed third ventricle (star) is observed in the designated location of the absent CC. C, Fetal MR imaging at 16 + 6 gestational weeks in the sagittal plane demonstrating the absence of the CC, resulting in an unroofed third ventricle (star) with the partial volume effect of the interthalamic adhesion and the anterior portion of the postcommissural fornix (white arrows). D, MR imaging in the coronal plane shows the unroofed third ventricle (black star) and the adjacent thalami (white stars).
Short and Dysgenetic Early CC
The patient was referred for a targeted scan at 19 weeks of gestation due to mild ventriculomegaly and mega cisterna magna. The CC appeared dysgenetic (Fig 4) and measured below the first centile of the new reference chart. On follow-up at 22.4 weeks, the dysgenetic CC again measured below the first centile.12 Additional findings included delayed sulcation, a falciform sinus, posterior fossa malformation, an enlarged gallbladder, and rotated, low-set ears. Microarray findings were normal; however, whole exome sequencing revealed a mutation in the CREBBP gene, corresponding to Rubinstein-Taybi syndrome.
A, Short and dysgenetic CC in Rubinstein-Taybi syndrome at 19.3 weeks. The rostrum is absent, the genu is underdeveloped (single arrow), and the splenium is short (double arrow), terminating prematurely at the anterior aspect (vertical line) of the massa intermedia (star). B, A normal CC at 19 weeks for comparison. The rostrum and genu (double arrow) are apparent and splenium (single arrow) is observed to extend beyond the posterior border (vertical line) of massa intermedia (star).
DISCUSSION
Malformation of the CC may result in neurocognitive impairment of various degrees.4⇓-6,29 The availability of high-resolution ultrasound machines provides an opportunity for an early diagnosis. The present study provides a nomogram of the early fetal CC between 13 and 19 weeks, displaying linear growth with advancing gestation. The findings of the study support the novel observation that the CC can be demonstrated and measured on prenatal ultrasound as early as 13 weeks. Presented are sonograms and MR images of the earliest prenatally diagnosed case of CC agenesis published so far.
Previous prenatal studies have established nomograms of the fetal CC8,12⇓-14,30 but none included the range of 13–15 weeks of gestation and only 2 included 16–17 gestational weeks.7,30 One had a small sample size and was performed >20 years ago using sonography machines of lower resolution compared with contemporary machines.7 The second study used a transabdominal approach,30 with an inherent limited resolution.
The current study offers imaging criteria to support early CC identification (Fig 1). In the earlier weeks, before the development of the cavum septum pellucidum and callosal sulcus, the small hypoechogenic CC may be difficult to discern from its surrounding. The pericallosal artery provides a supportive landmark of the early CC. The use of the pericallosal artery at early gestation as an indirect marker of normal CC development has been previously studied and validated.31⇓-33 By 18 weeks of gestation, the cavum septum pellucidum and callosal sulcus have developed, facilitating the visualization of the CC (Fig 2).34
A recent pioneering publication by Birnbaum et al21 depicted the development of the early CC from 14 weeks using a 3D multiplanar technique. This study showed the evolution of the developing CC between 14 and 17 weeks of gestation, showing a bidirectional growth of the CC. Similar to the current study, Birnbaum et al recognized the early CC as a small hypoechogenic midline structure in the anterior part of the fetal brain, bounded by the pericallosal artery superiorly and the tela choroidea inferoposteriorly.
The current study is the second to address the sonographic demonstration of the developing CC. Furthermore, it extended the lower limit of in vivo sonographic CC demonstration to 13 weeks. This observation is supported by embryologic studies showing the crossing of decussating cingulate, neocortical, and callosal fibers across the midline at 13 weeks.2,35 The decussation of callosal fibers is guided by specialized glial cells that form the “glial sling,” a temporary structure serving as a bridge across the midline.34 In the absence of the glial sling, callosal fibers do not cross over, traveling, instead, parallel to the midline.35
Short Early CC
A short early CC under the fifth centile was observed in 3% (6/198) of cases. In 2 cases, the CC was observed to be consistently short yet well-formed throughout pregnancy, supporting the validity of the early CC growth chart.
All patients reported an uneventful pregnancy and delivery of an apparently healthy offspring. Moreover, 5/6 cases were reported to achieve expected milestones at the Maternal Child Health Care Clinic assessment, implying that a short early CC may be a normal variant. This observation is supported by previous studies demonstrating that children with a short but completely formed CC may have normal neurodevelopment.5,6
Early Detection of Pathologic Cases
Our study presents 2 early pathologic cases: complete agenesis (Fig 3) and a short dysgenetic CC (Fig 4). These cases suggest that some callosal pathologies may be apparent in early gestation, contrary to claims in the current literature of a later detection of callosal anomalies.4,19,32,36 The availability of informative images of these pathologic cases enhances our understanding of normal-versus-abnormal early brain development. Additional cases are needed to confirm these preliminary observations.
An in-depth review article by Raybaud35 addressing the normal and abnormal development of the forebrain commissures considered commissural agenesis a constellation of features, of which the most prominent is the absence of an apparent interhemispheric connection, as seen in the current agenesis of the CC case. In his review, Raybaud suggested that the term “CC agenesis” was inaccurate because histologic evidence shows that the CC fibers have, in fact, developed but are heterotopic. Unable to cross the midline, the CC fibers reroute parasagittally in a bundle commonly known as Probst bundles, named after Moriz Probst, an Austrian neuroanatomist who first described them in 1901.35 Previous neuropathologic studies of CC agenesis included cases from the midtrimester, reporting late-appearing signs such as Probst bundles, widely spaced lateral ventricles, and the absence of the cavum septum pellucidum.33,35,36 Although the current study details only a single case of CC agenesis at 14 weeks, the early signs described herein may aid in raising clinicians’ awareness of signs on the early anomaly scan in future cases.
Strengths and Limitations
There are several limitations to address. First, as an in vivo study of viable fetuses, it lacks histologic correlation to support our observation of an early CC. This is partially overcome by the sequential demonstration of the growing CC, week by week, from a minute curved structure to the characteristic shape of the CC bridging the cavum septum pellucidum, confirming its identity. A second limitation of the study is the absence of follow-up measurements later in pregnancy in all short early CC cases. This was discovered in hindsight after the construction of the reference chart.
The strengths of the study are its novelty in measuring the growth of the CC during early gestation and studying its gradual appearance from the earliest week assessed so far. Moreover, presented is the earliest prenatally diagnosed case of agenesis of the CC, providing an in vivo demonstration of the appearance of the fetal brain in this condition both on ultrasound and MR imaging (Fig 3). Additional strengths of the growth chart include prospective recruitment, strict inclusion criteria, reproducibility, and a well-sized cohort of weekly measurements.
CONCLUSIONS
The developing CC can be demonstrated and measured as early as 13 weeks of gestation using high-resolution sonography. Some major callosal pathologies can be identified at this stage. In an era striving for early detection of fetal anomalies, it is imperative to maximize the potential of early screening and to challenge the limits of existing diagnostic tools. The nomogram of the early fetal corpus callosum and the pathologic cases presented in this study may enhance our understanding of normal and abnormal patterns of callosal development.
Acknowledgments
The authors are grateful to Professor Shali Mazaki Tovi for providing insight and guidance, supporting the study. We would also like to thank Mr Andrey Greenfield for skillfully editing and illustrating Videos 1 and 2.
Footnotes
Disclosure forms provided by the authors are available with the full text and PDF of this article at www.ajnr.org.
References
- Received September 14, 2022.
- Accepted after revision December 10, 2022.
- © 2023 by American Journal of Neuroradiology