Abstract
BACKGROUND AND PURPOSE: While functional MR imaging and other techniques have contributed to our knowledge of functional brain localization, these methods have not been extensively applied to the complex and incompletely understood task of swallowing. We used functional MR imaging to investigate motor cortex activity during swallowing in healthy human adults.
METHODS: Eight subjects were imaged on a 1.5-T MR system using blood oxygen level–dependent contrast mechanisms. Subjects performed three swallowing tasks and a finger-tapping task. Areas of activation in the cortex and subcortical areas were tabulated, and a laterality index, defined as LI = [Σs left − Σs right]/[Σs left + Σs right] × 100, was computed for the three tasks.
RESULTS: Activation was observed in the primary motor and sensory cortices, motor processing and association areas, and subcortical sites. This activity was dominant for one hemisphere with left hemispheric dominance more prevalent among the subjects. Right hemispheric dominance, however, showed stronger lateralization than the left hemisphere.
CONCLUSION: Our data indicate that specific sites in the motor cortex and other cortical and subcortical areas are activated with swallowing tasks and that hemispheric dominance is a feature of swallowing under these conditions. In addition, we demonstrate the utility of functional MR imaging in the study of the cortical representation of swallowing and suggest a role for functional MR imaging in the diagnosis of dysphagia of cerebral origin.
Swallowing is a complex physiologic function, requiring control or regulation at the cortical, brain stem, and peripheral levels. It follows that an understanding of the cortical control mechanisms in swallowing would facilitate the ability to predict dysfunction following cerebral injury or disease. Nevertheless, efforts to correlate vascular territory or size (large or small vessels) of cerebrovascular accidents with specific oral, pharyngeal, or laryngeal deficits have been disappointing (1–4). This inability to correlate cerebral vascular disease with peripheral dysfunction may relate to a fundamental lack of understanding of cortical function in human swallowing.
Some animal studies have shown cortical activity in the inferolateral precentral gyrus with swallowing (5, 6), whereas others have shown swallowing behavior evoked by stimulation of the lateral pericentral and insular cortex as well as the precentral cortex (7). In humans, most of the available data on cortical contributions to swallowing have come from reports on the peripheral functional outcome of cerebral vascular injury (8, 9). Some information has also come from invasive cortical stimulation or recording from patients during neurosurgical procedures (10). Both types of data provide only limited information on the cortical contributions to swallowing. Nevertheless, recent investigations have implicated a more significant role for the motor cortex in the motor control of swallowing (7, 11). These studies suggest that the motor cortex may be one of several sites that integrate initiation and execution of the swallowing motor program, and even of cortical reorganization in response to changing environmental demands (11–13).
The advent of functional MR imaging techniques has permitted measurements of cortical activity during the performance of specific motor tasks (14). Functional MR imaging has been used to study simple movements, such as single-plane finger movements, and even more complex functions, including the motor processing of speech (15). The purpose of this study was to use functional MR imaging techniques to investigate the motor control of swallowing in a preliminary effort to gain insight into the control scheme of this complex physiologic function.
Methods
Eight healthy adult subjects (four men and four women; average age, 34 years; seven right-handed, one left-handed) were imaged in the axial and coronal planes using standard functional MR imaging blood oxygen level–dependent (BOLD) techniques. The subjects were imaged on a 1.5-T MR system using gradient-echo echo-planar sequences with the following acquisition parameters: 2000/60 (TR/TE), 64 × 64 matrix, 24-cm field of view, 5-mm-thick contiguous sections, and a 90° flip angle. A total of 140 images at each of 14 section locations were obtained using a standard quadrature bird cage head coil. Spin-echo (450/14) high-resolution anatomic images were acquired in the axial and coronal planes in the same section locations during the same imaging session. Subjects' heads were immobilized with foam and taped to the head holder to prevent motion. Institutionally approved written informed consent was obtained for all subjects.
Task Paradigms
To stimulate cortical activation associated with swallowing, the following paradigm was employed: subjects performed four repetitions of swallowing for 10 or 15 seconds alternating with rest for 30 seconds. Subjects first performed repeated dry swallows (swallowing their own saliva) for 10 seconds, then repeatedly swallowed a small bolus (approximately 3 mL) of self-administered room-temperature sterile water supplied through a plastic catheter for 10 seconds. Hand movement was restricted during self-administration of the water bolus to avoid contamination of the signal by motion artifacts. Last, subjects performed repeated dry swallows for 15 seconds. For each swallow task, subjects were instructed to begin swallowing on verbal command and to swallow as fast as possible until instructed to stop. The 10- and 15-second activation cycles were chosen to permit adequate sampling of motor activity during swallowing, given the considerable intrasubject variability in peripheral motor output for swallowing (11).
As a positive control for motor cortex activation, subjects additionally performed a self-paced finger-tapping task as a standard measure of motor cortical activation on functional MR imaging examinations (16). Subjects tapped the fingers against the opposing thumb on both hands simultaneously, at maximum speed for 30 seconds, with a rest period of 30 seconds, repeated twice.
Data Analysis
Image reconstruction and analysis were performed using software routines written in Interactive Data Language (Research Systems, Inc, Boulder, CO). Motion correction was executed with an in-plane least-squares motion correction algorithm. Spurious signals resulting from motion or cardiac or respiratory cycles were removed via a section-wise method that removes spatially coherent signal changes via the application of a partial correlation method to each section in time (17). A cross-correlational analysis was applied to create statistical maps of activation associated with each motor task, and the resultant images were overlaid on the spin-echo anatomic images (18). Statistical maps of swallowing and finger-tapping were generated using P < .03, r = 0.60. Localization of activated pixels for comparison of different tasks within subjects was performed relative to the central sulcus. The central sulcus was identified according to the method of Meyer et al (19).
To verify that the subjects swallowed during the activation periods and remained motionless during rest periods, MR-compatible surface electrodes were placed over the thyrohyoid muscle and thyroid cartilage to measure movement of the hyoid/laryngeal complex. Signals were recorded on a monitor (Model 3100, In Vivo Research Systems, Orlando, FL). Timing of the electrode recordings was synchronized to the swallowing task paradigm.
The location of activated areas for each task in each subject was determined by identifying the location of activation in Brodmann's areas using established neuroanatomic landmarks (20, 21).
To differentiate areas of activation associated with swallowing from those associated with finger tapping within subjects, a χ2 analysis was performed comparing site (location of activated pixels relative to the central sulcus and intrahemispheric fissure in the axial plane, and the sylvian fissure and intrahemispheric fissure in the coronal plane), location (section number), volume, and percentage of activation.
Swallowing movements are thought to produce motion artifacts resulting in spurious activation. To ensure elimination of data compromised by motion, echo-planar acquisitions were inspected visually in cine mode. No gross head motion occurred for the data sets used in this investigation. In addition, the data sets were analyzed for translational and rotational movement in each plane using SPM96 (Wellcome Department of Cognitive Neurology). Mean translation movement in the z, y, and x planes for swallowing were 0.28 mm (± 0.6 mm SD), −0.28 mm (± 0.7 mm SD), and −0.05 mm (± 0.2 mm SD), respectively. Mean translation in the z, y, and x planes for finger-tapping were 0.3 mm (±0.3 mm SD), 0.26 mm (±0.4 mm SD), and 0.18 mm (± 0.3 mm SD), respectively. Mean rotational movements for swallowing were as follows: pitch (−0.12° ± 1.0° SD), yaw (0.23° ± 0.4° SD), and roll (−0.13° ± 0.7° SD). Mean rotational movements for finger-tapping were as follows: pitch (0.12° ± 0.3° SD), yaw (0.11° ± 0.13° SD), and roll (0.06° ± 0.04° SD). An unpaired Student's t-test was performed (CI = 95; Bonferroni-Dunn correction) to determine differences in the amount of movement between the swallowing and finger-tapping tasks. No significant differences between the tasks were found in the three planes for translation movements (z, t = 0.87, P = .45; y, t = −1.91, P = .15; x, t = −1.62, P = .20) or in the three directions for rotational movements (pitch, t = −1.09, P = .36; yaw, t = −1.22, P = .31; roll, y = −1.54, P = .22).
Results
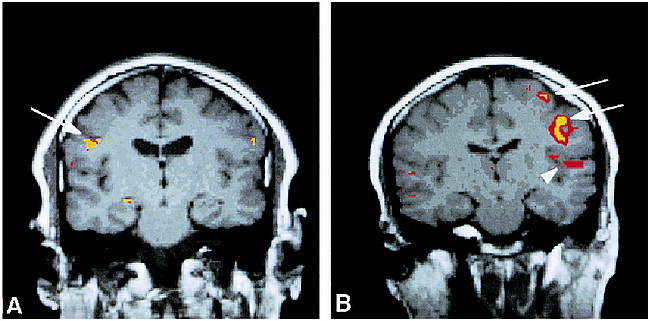
Swallowing during the 10- and 15-second paradigms resulted in bilateral activation in the mid-lateral precentral gyrus (primary motor cortex) in all subjects. Figure 1 shows coronal images from subjects 4 and 6 with activation of the right or left motor cortex, respectively, during a 10-second dry swallow task. Activation of the mid-inferior portion of the primary motor cortex (Brodmann's area 4) for all subjects during swallowing was consistently located at least one section (5 mm) anterior and inferior to Brodmann's area 4 activated by finger-tapping (χ2 test, P = .22, not significant), indicating different sites of activation within the primary motor cortex.
A and B, Coronal functional MR images overlaid to T1-weighted (450/14/0.75) anatomic MR images show activation of the primary motor cortex (arrows) and superior temporal gyrus (arrowhead) during a 10-second dry swallow task for subjects 4 (A) and 6 (B)
Table 1 shows the distribution of activation in cortical and subcortical sites (designated in Brodmann's areas) during the swallowing paradigms. The activation recorded for these three tasks, for most of the subjects, was generally diffuse, occurring in several locations in the frontal, parietal, and temporal lobes. Specifically, Table 1 shows activation occurring predominately in Brodmann's area 4 (the primary motor cortex), areas 3, 2, and 1 (the primary somatosensory cortex), area 6 (the supplementary motor cortex), areas 9 and 10 (the prefrontal cortex), area 42 (the transverse temporal gyrus), areas 24 and 31 (the cingulate gyrus, insular cortex, and internal capsule), areas 44 and 45 (the speech areas), as well as in areas 39 and 40 (other association areas), area 22 (the superior temporal gyrus), and areas 5 and 7 (the sensory-motor integration areas).
Distribution of activation during swallowing tasks in Brodmann's areas (area number designation)
We observed that the relative distribution of activation appears to change with different tasks, as measured by the number of areas with activation identified (see Table 1). For example, greater activation of the right motor cortex as compared with the left motor cortex was observed for the 10-second dry task in all subjects except case 6. The 10-second wet and 15-second tasks overall were more evenly distributed, with three subjects having greater activity on the right (cases 2, 4, and 7: 10-second wet task; cases 1, 3, and 4: 15-second tasks), three subjects with greater activity on the left (cases 3, 5, and 6: 10-second wet task; cases 5, 6, and 7: 15-second tasks), and two subjects with bilateral activation (cases 1 and 8: 10-second wet task; cases 2 and 8: 15-second tasks).
This apparent predominance of activity on one side of the motor cortex within subjects during swallowing tasks suggests that the motor control of swallowing may be characterized by hemispheric dominance, as has been documented for speech (10, 22). Hemispheric dominance has been quantified in other studies using a laterality index (LI), defined by the ratio 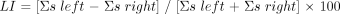 where s = (percentage of activation) × (number of activated pixels). A positive LI indicates left hemispheric dominance while a negative LI indicates right hemispheric dominance. Ratios at or close to 0 are thought to represent an indeterminate dominance (15, 23).
where s = (percentage of activation) × (number of activated pixels). A positive LI indicates left hemispheric dominance while a negative LI indicates right hemispheric dominance. Ratios at or close to 0 are thought to represent an indeterminate dominance (15, 23).
Table 2 shows the LI for each of our eight subjects during the 10-seconds dry, 10-seconds wet, and 15-seconds dry swallowing tasks. Five of the eight subjects (cases 3, 5, 6, 7, and 8) showed left hemispheric dominance during the 10-second dry swallow task; two (cases 1 and 4) were right dominant, and one (case 2) was indeterminate or slightly right dominant. For the 15-second task, five subjects (cases 2, 4, 5, 6, and 7) were left dominant and three (cases 1, 3, and 8) were right dominant. Five of the subjects (cases 2, 3, 4, 7, and 8) were left dominant during the 10-second wet task, whereas subjects 1 and 5 were right dominant; subject 6 was indeterminate. Examination of the data for the three swallowing tasks individually, as well as for the three tasks pooled, consistently showed that five (63%) of the eight subjects were left dominant. Within subjects, however, left or right hemispheric dominance was not consistent through the three tasks for six of the eight subjects. Thus, the LI changed for one out of the three tasks in 75% of the subjects.
Laterality index (LI) for different swallowing tasks
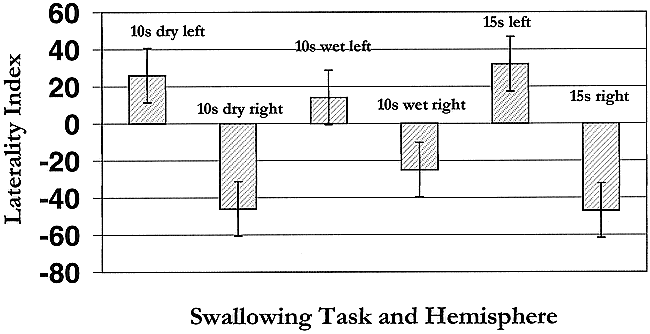
In addition, all subjects showed a change in the degree of laterality among the three tasks; that is, the value of the LI increased or decreased within each subject over the three tasks. Moreover, the relative degree of the LI appeared to be greater for the right hemisphere relative to the left hemisphere. An ANOVA (StatView 4.5, Abacus Concepts, Berkeley, CA) was performed to examine the interactions between the three tasks (to look for effects of task sequence on hemispheric dominance), interactions for right and left hemispheric dominance, and the LI within and among subjects. The only variable with a significant effect was right versus left hemispheric dominance, P < .001. Thus, task sequence, within or between subject variability, does not explain the observed changes in the LI across tasks. There was, however, a significant effect for hemispheric dominance in that when subjects were right-hemisphere dominant for a particular swallowing task, they showed stronger dominance than when they were left-hemisphere dominant. This tendency is illustrated graphically in Figure 2, which shows the mean LI for the three swallowing tasks (all subjects pooled) in the right and left hemispheres.
Graph of the mean LI for three swallowing tasks in the right and left hemispheres. Each bar represents pooled data from all eight subjects. Positive values = left hemisphere; negative values = right hemisphere. Length of the bar represents the degree of laterality for that hemisphere
Last, the relationship between handedness and hemispheric dominance for swallowing tasks was examined with a Pearson correlation analysis. There was no significant relationship between handedness and hemispheric dominance for swallowing in this study (r = −0.12, 95% confidence interval), consistent with findings from other studies (24).
Discussion
The purpose of this investigation was to identify sites of cortical activation during swallowing by using functional MR imaging techniques. Swallowing in healthy adult subjects resulted in activation of the primary motor cortex, with activation also identified in the primary somatosensory cortex, the supplementary motor cortex, the prefrontal cortex, the transverse temporal gyrus, the insular cortex, the internal capsule, the cingulate gyrus, the speech areas, as well as other association areas, the auditory association area, and the sensory-motor integration areas.
The activation identified in this study corresponds to sites of cortical stimulation or evoked responses reported in other studies (7, 13, 24) as well as to sites not identified with traditional cortical stimulation techniques. Multiple sites of activation, including those associated with motor processing, suggest that the motor control of swallowing may involve several cortical sites to initiate, process, and execute the necessary output for swallowing. Activation of other cortical sites, such as the supplementary motor area, represented in the superior and middle frontal gyri, is believed to be associated with motor planning and, in particular, with planning of sequential movements, as occurs with swallowing (25). Moreover, activation in the various association areas, including the speech areas and the sensory-motor integration areas, is consistent with earlier studies reporting swallowing responses evoked via stimulation of these areas and activity of prefrontal and parietal association areas (7, 10).
In subcortical areas, activation of the posterior limb of the internal capsule, the insular cortex, and the thalamus was observed in the subjects during swallowing tasks. Activation of the internal capsule is an expected and important functional feature in swallowing, as the internal capsule serves to functionally connect cortical and brain stem nuclei via the cortical bulbar tracts. Vascular injury to these white matter tracts has been associated with oropharyngeal deficits in swallowing (26). Activation in the white matter, however, raises the issue of the BOLD effect in the absence of synaptic activity, since the white matter consists of axons. It is well established that neuronal activity is accompanied by increasing metabolic activity. BOLD techniques do not directly measure metabolic activity; rather BOLD contrast is a reflection of the hemodynamic response associated with increases in metabolic activity (27, 28). Both synaptic activity and axonal conduction require the active maintenance of ionic (Na+ − K+) gradients. The metabolic processes that drive these gradients involve the same substrates (eg, glutamate transport) in both the synaptic cleft and in the axon (29–31). Thus, synaptic activity and axonal activity rely on fundamentally similar metabolic processes. Aside from metabolic requirements, other explanations for the white matter activation observed in these experiments may include spatial averaging from the basal ganglia in some cases. Regardless of the specific source(s) of the white matter signals, further investigation into the relationship between white matter function and hemodynamic response is warranted.
In other areas, the insular cortex is thought to serve a variety of functions, including sensory and motor integration between primary cortex and other subcortical (thalamic) nuclei or limbic areas. Other functions include a role in gustatory sensation, visceral motor activity, and motor association, as well as the processing of “routine” or “overlearned” motor associated tasks. Thus, activation of the insular cortex during swallowing tasks may be linked to its visceral motor and integrative functions (32).
The nuclei of the thalamus are heterogeneous in their functional roles, with some nuclei serving as relays for cortical areas and others generally serving as association areas (33, 34). The globus pallidus projects to the ventral anterior group of the thalamus, which then projects to the primary motor and supplementary motor cortices. The ventral posterior group is primarily involved with somatosensory integration, receiving input from the spinothalamic and trigeminal tracts and projecting to the primary somatosensory cortex. The pulvinar, as an association nucleus, maintains reciprocal connections to the temporal, parietal, and occipital lobes for integration of sensory information and cognitive and visual association functions. Activation of thalamic nuclei during swallowing tasks indicates the necessary role of sensory and motor input processing via thalamocortical or thalamostriatal pathways in swallowing.
For the majority of the tasks in this experiment, subjects showed bilateral activation during swallowing (Table 1), although with evidence of hemispheric dominance, most commonly on the left. Previous investigations have shown a prevalence of left hemispheric dominance in speech motor control (35, 36). Likewise, some areas of activation identified during swallowing were similar to those reported during articulation in speech motor control studies; specifically, areas in the superior temporal gyrus and middle frontal cortex (15). Thus, the motor control of swallowing appears to be characterized by cortical processes that are similar to those observed for speech: hemispheric lateralization in cortical function and multiple sites of activation. While this study did not measure cortical activation during speech, future investigations will compare cortical control processes for speech and swallowing in the same cohort of subjects.
Hemispheric dominance, however, appeared to be variable in some subjects depending on the swallowing task. As shown in Table 2, six of the eight subjects alternated hemispheric dominance during the three swallowing tasks. For example, subject 3 was left-hemisphere dominant during the 10-second dry and 10-second wet swallow tasks, while during the 15-second task, the subject showed right hemispheric dominance. This tendency toward alternate hemispheric dominance during different swallowing tasks could be attributed to a variable attention state. Other investigations have found differential activation in the motor cortex during motor activity with different attention states (37). Alternatively, previous studies have shown that subjects use highly variable neuromuscular strategies in different swallowing sensory and motor tasks (11, 38). Hamdy et al (24) used transcranial magnetic stimulation in a group of 20 healthy subjects to show that evoked responses from the pharynx and esophagus were lateralized to either the right or left hemisphere, but there was no consistent pattern of lateralization from these sites. Thus, alternate hemispheric dominance may reflect a cortical organization scheme for swallowing that facilitates the diverse neuromuscular demands of different swallowing tasks.
In addition, as Figure 2 shows, lateralization in the right hemisphere tended to be greater than in the left hemisphere, although, as discussed previously, the majority of subjects were left-hemisphere dominant for swallowing tasks. The average LI (Table 2) for any of the swallowing tasks, however, was less than 5 or close to 0. These results suggest that the hemispheric control of swallowing, when averaged across subjects and tasks, is indeterminate or bilateral. Why the right hemisphere shows stronger lateralization is unclear and remains to be determined; however, recent studies have provided insight into the developmental dichotomy of the two hemispheres. Swallowing is a phylogenetically old physiologic function and in an investigation of functional brain asymmetry during childhood, Chiron and colleagues (39) showed that human infants are right-hemisphere dominant up to the age of 3, at which point asymmetric shifts to the left occur. One possibility is that stronger lateralization in the right hemisphere during swallowing tasks may represent a primal cortical organizational structure.
Nonetheless, there are experimental considerations and limitations to this investigation. First, the activation observed during these tasks could represent the cortical activity associated with swallow initiation (the oral phase of swallowing, thought to be under voluntary control) and not activation associated with motor control of the reflexive or automatic phases of swallowing: the pharyngeal and esophageal phases (40). This was addressed during the 10-second wet task by requiring the subjects to swallow a small bolus. Delivery of a bolus to the pharyngeal inlet (tonsillar pillars) is believed to stimulate the pharyngeal phase of swallowing (40). Apart from differences in laterality measures, sites of activation during the 10-second wet task were very similar to the 10- or 15-second dry tasks for each subject. Thus, the cortical activation observed in this investigation likely represents the cortical activity associated with all phases of swallowing.
Furthermore, subjects were required to swallow repeatedly while lying supine over the course of several seconds. Although this does not represent “normal” swallowing conditions, subjects completed swallows of normal duration for all swallowing tasks, as measured by the number of swallows in each activation period. Cortical activation observed under these conditions appears to be associated with swallows of normal duration (mean, 1.8 seconds).
Last, the subjects' attention state was not corrected for in any of the swallowing tasks. Statistical analysis, however, showed no effect of task sequence, which would indicate that differing cognitive attention states due to task sequence did not significantly contribute to the observed variability in hemispheric dominance. Rushworth and colleagues (41) examined cued sequential hand movements and proposed that the left parietal cortex may play a role in motor attention, specifically, the shift from one movement to the next in sequential motor movements. Future studies are indicated to determine the hemispheric role of motor attention states for complex movements such as swallowing.
Conclusion
The results of this investigation confirm earlier studies of cortical activity in swallowing and contribute additional information on the cortical organization for swallowing. Although we found hemispheric dominance for different swallowing tasks under the conditions of this investigation, examination of the role of hemispheric dominance in the cortical control of swallowing and the implications for normal and abnormal swallowing is necessary.
Finally, this study demonstrates the utility of functional MR imaging in investigating the cortical control of swallowing. While this study examined healthy subjects, our results suggest a role for functional MR imaging in examining cortical and subcortical function in abnormal swallowing. Clearly, a database of cortical and subcortical function in healthy subjects during swallowing tasks is an essential prelude to the examination of abnormal swallowing. Having established a normal database, functional MR imaging may prove an adjunctive technique to conventional MR imaging in the investigation of dysphagia following cerebral injury, insult, or disease.
Acknowledgments
We thank Andrew Kalnin for his helpful comments on the manuscript.
Footnotes
1 Supported by Foundation of UMDNJ Grant #15–97.
2 Presented in part at the fifth annual meeting of the International Society for Magnetic Resonance in Medicine, Vancouver, April 1997, and at the annual meeting of the American Society of Neuroradiology, Toronto, May 1997.
3 Address reprint requests to Kristine M. Mosier, DMD, PhD, Department of Oral Pathology Biology and Diagnostic Sciences, Division of Oral & Maxillofacial Radiology, Room C-829, New Jersey Dental School, University of Medicine and Dentistry of New Jersey, 110 Bergen St, Newark, NJ 07103.
References
- Received February 27, 1998.
- Accepted after revision March 29, 1999.
- Copyright © American Society of Neuroradiology