Abstract
BACKGROUND AND PURPOSE: High signal intensity (HSI) at the pituitary stalk is reported in pituitary adenomas. Our purpose was to clarify how and when this HSI formed, its long-term fate, and its relation to the function of infundibuloneurohypophyseal (INH) system.
METHODS: Twenty-two patients with pituitary adenoma and supradiaphragmatic extension underwent 1.5-T MR imaging. Patients were assigned to two groups A (n = 18; those with stalk tip HSI) and B (n = 4; those without HSI) on postoperative T1-weighted images. Endocrine status was postoperatively evaluated and compared in both groups.
RESULTS: Group A patients did not have postoperative permanent diabetes insipidus (DI). Preoperative images in 17 patients revealed linear or ovoid HSI on the adenoma surface immediately above the diaphragma sellae. Of these, two with a poorly developed diaphragma sellae had HSI near the median eminence and inside the sella turcica. HSI was not apparent in the remaining patient with a giant, irregularly shaped adenoma. In group B, three patients had permanent DI. No patient had HSI on preoperative images.
CONCLUSION: Postoperative pituitary stalk HSI is derived from preoperative supradiaphragmatic HSI on the adenoma surface. The suspected mechanism is blockage of the hypophyseal-pituitary axis, with an accumulation of neurosecretory granules at the diaphragmatic level. Diaphragmatic shape may influence the location of HSI. The shape and location of HSI are essentially stable for years after surgery. No patients with permanent DI had HSI before or after surgery. HIS at the pituitary stalk tip is a useful landmark for predicting functional integrity of the INH system in patients with a large pituitary adenoma.
With the advent of superconductive MR systems, a more detailed depiction of normal and pathologic anatomy of the infundibuloneurohypophyseal (INH) system has become possible (1, 2). High signal intensity (HSI) in the posterior lobe is considered to represent neurosecretory granules that consist of an antidiuretic hormone (ADH)–neurophysin (carrier protein) complex packaged within a phospholipid membrane (3, 4). Patients with central diabetes insipidus (DI) have been shown to lack the HSI (1, 2, 4). Any process that disturbs the transport of ADH from the hypothalamus to the posterior lobe can result in the accumulation of HSI material in the median eminence or pituitary stalk proximal to the obstruction (1); this result has been reported in large pituitary adenomas, a most common pituitary lesion (1, 5, 6). However, to our knowledge, how and when HSI forms, its fate throughout the clinical course, and its relation to the function of the INH system have not been elucidated. The assessment of these features is not only radiologically interesting, but also clinically useful in the perioperative management of DI, because transient DI frequently occurs, and it may even become permanent after surgery for large adenomas (7, 8). This retrospective study was performed to elucidate the frequency of appearance of postoperative HSI at the pituitary stalk tip, its origin, its formation mechanism, and its clinical implications in large pituitary adenomas.
Methods
In the last 4 years, operations were performed for 22 pituitary adenomas with suprasellar extension at Kawatetsu Chiba Hospital, Japan. In three patients with prolactinoma, postoperative adjuvant bromocriptine therapy was performed. Patients included eight men and 14 women aged 17–74 years (mean age, 51.5 years). The adenoma types were nonfunctioning in 16 patients, prolactin secreting in five, and growth-hormone secreting in one. The maximum height of the ranged from 19 to 52 mm.
A three-dimensional T1-weighted MR imaging examination was performed before and after surgery with a 1.5-T MR system. A Gyroscan ACSNT 1.5-T and T1-weighted fast spin-echo method was used before and after a 10-mL intravenous injection of gadopentetate dimeglumine. Sequence parameters used were the following: TR/TE/NEX of 475/17/2 with a section thickness of 3 mm and an intersection gap of 0.5 mm. The matrix was 512 × 256, and the FOV was 20 cm.
Postoperative MR imaging was performed 2 weeks to 4 years after surgery. The presence or absence of HSI and its location, when present, were determined when compatible MR findings were obtained in three dimensions. Three physicians (N.S., S.H., I.T.) evaluated the radiologic findings, and the final decision was made by consensus. Patients were grouped into 2 subclasses based on the postoperative T1-weighted MR findings. Group A consisted of patients with HSI at the tip of the pituitary stalk; group B, patients without this HSI.
At postoperative endocrinologic evaluation, DI was considered to be absent in patients without polydipsia and polyuria and in those who did not need 1-desamino-8-d-arginine vasopressin (DDAVP, or desmopressin). Their basal levels of ADH and plasma osmolarity were incompatible with the diagnosis of central DI. A water-deprivation test and/or hypertonic saline infusion test were performed to determine the diagnosis of postoperative persistent DI in patients who needed DDAVP (8). Anterior pituitary hormones were replaced postoperatively, when necessary, as determined by the results of basal hormonal levels and triple-loading test results (ie, thyrotropin releasing hormone, leutenizing hormone releasing hormone, and insulin test results).
Results
Group A
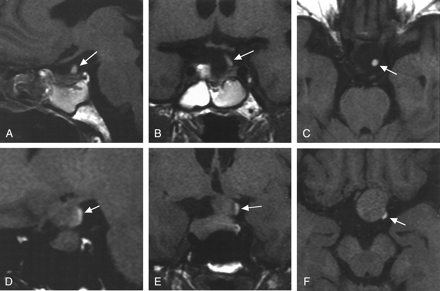
No patients in group A had permanent central DI after surgery (Table 1). Regarding the postoperative MR images, the location of the pituitary stalk became evident after surgery (Figs 1A–C, 2A and B, 3A). HSI areas were present at the tip of the pituitary stalk, where it had an ovoid or occasionally linear shape; its width ranged from 1 to 4 mm (Figs 1A–C, 2A and B, 3A). Postoperative MR studies were performed 2 weeks to 4 years after surgery. The HSI at the stalk tip remained unchanged in 10 patients who underwent repeated postoperative MR examinations for as long as 2 years after surgery (Figs 1A–C, 2A and B).
Case 5, group A. Images in a 63-year-old man with a nonfunctioning adenoma show that the HSI (arrow) was present both before and 2 weeks after surgery.
A–C, At 2 weeks after surgery, ovoid HSI is visible at the level of the diaphragm sellae on sagittal (A), coronal (B), and axial (C) sections.
D–F, Before surgery, linear HSI is visible at the posterolateral surface of adenoma, above the indentation formed by a diaphragma sellae, on the sagittal (D), coronal (E), and axial (F) sections.
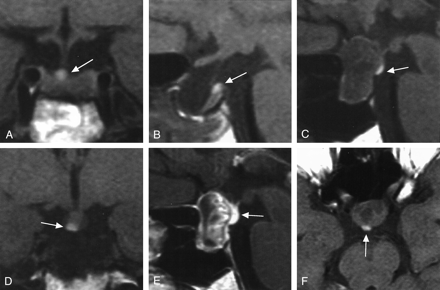
Case 16, group A. In this patient, a 35 year–old woman with a nonfunctioning adenoma, MR studies were repeated at 3, 12, and 24 months after surgery and showed essentially the same findings, including HSI (arrow).
A and B, At 2 years after surgery, HSI is visible at the tip of pituitary stalk at the level of the diaphragma sellae on the coronal (A) and sagittal (B) sections.
C–F, Before surgery, linear HSI is visible at the posterior surface of adenoma, above the indentation formed by the diaphragma sellae, on the sagittal (C), coronal (D), contrast-enhanced sagittal (E), and axial (F) sections. HSI is identifiable at the stalk stump. The enhanced image shows HSI with more remarkable enhancing effect due to normal pituitary tissue; this finding suggests its connection to the pituitary stalk.
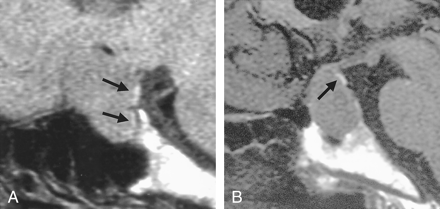
Case 17, group A. Images in a 74 year-old-woman with a growth hormone–secreting adenoma show HSI (arrow).
A, Coronal section 6 months after surgery. Postoperatively, the adenoma was debulked, and the HSI was below the chiasm and deviated to the left. It was continuous with the proximal portion of the pituitary stalk.
B, Coronal section before surgery. Preoperatively, an ovoid HSI is visible on the supradiaphragmatic level on the left side. The optic chiasm is compressed superiorly.
Clinical features of 22 patients with pituitary adenoma
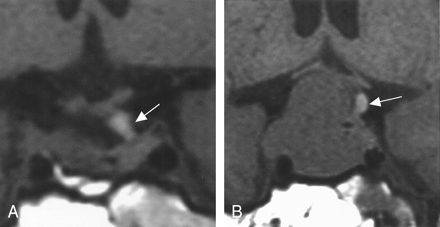
On preoperative MR images in 17 patients, a linear, round, or ovoid HSI was visible on the posterior or lateral surface of the adenoma above the sella diaphragm. The lower end of the HSI was revealed at or above the level of the indentation formed by the posterior or lateral protrusion of the diaphragm (Figs 1D and E, 2C and D, 3B). Images in one patient showed linear HSI at the posterior portion in the sella as well (case 9, Fig 4A). The other patient had the HSI near the median eminence, distant from the diaphragmatic level (case 14, Fig 4B). The indentation of the tumor due to the posterior diaphragmatic margin was less conspicuous in these patients than in the others. With the aid of contrast enhancement, the location of HSI was more clearly demonstrated at the stump of the pituitary stalk, even preoperatively (Fig 2C and E). By comparing the MR images before and after surgery in 17 patients, the postoperative HSI of the pituitary stalk seemed to be derived from the preoperative HSI on the adenoma surface (Figs 1–3).
Sagittal sections obtained before surgery show HSI (arrows).
A, Case 9, group A 39-year-old woman with nonfunctioning adenoma. HSI was noted in both supra- and intrasellar areas. The indentation at the level of the diaphragma sellae was noted but not prominent.
B, Case 14, group A. A 54-year-old woman with nonfunctioning adenoma. HSI was noted close to the median eminence. Indentation of diaphragma sellae was not evident in this case.
Images in the remaining patient (case 18) did not show such HSI on the adenoma surface. This patient had a huge prolactinoma with an irregular contour.
Group B
Group B consisted of four patients without HSI at the tip of the pituitary stalk. In three of the four patients in this group (cases 20–22), permanent DI developed, and the patients required the administration of DDAVP after surgery. Although none of the patients in group B patients had HSI after surgery, one patient (case 20) had an HSI focus within the adenoma due to pituitary apoplexy that made it difficult to determine presence or absence of HSI.
Comparison of Groups A and B
After surgery, permanent DI was present in three of the four patients of group B whose images did not show HSI, and DI was absent in all patients of group A whose images did show HSI (Tables 1 and 2). The postoperative absence of HSI was a significant indicator of permanent DI (P = .024, Fisher exact probability). Preoperative tumor size was not significantly different between group A and group B (P = 0.51, unpaired t test).
Cases of pre- and postoperative HSI and postoperative DI
Discussion
The postoperative presence of HSI within the pituitary stalk was reported previously, but its frequency or preoperative state was not described (1, 5, 6). In the present study of 1.5-T MR images, 18 of 22 patients with macroadenoma of the suprasellar extension had postoperative HSI at the pituitary stalk. In these patients, postoperative MR images in 17 showed supradiaphragmatic linear, round, or ovoid HSI at the posterior or lateral surface of the adenoma. With the use of a high-magnetic-field system and proper sequence parameters as described earlier, HSI is frequently detected in large adenomas with suprasellar extension. After the successful resection of the adenomas, the HSI focus relocates into or toward the inside of the sella. The displaced HSI within the pituitary stalk assumes a more normal position, but the position of the HSI does not change relative to the stalk on the pre- and postoperative images. Although its preoperative shape tends to be linear, HSI becomes ovoid after surgery, probably due to the elimination of compression. For the same reason, a slight increase in size is seen on postoperative images.
Postoperative surgical packing, residual tumor, or hematoma after surgical manipulation may show intrasellar HSI, particularly within 3 months after surgery (9, 10); this finding can make their differentiation from HSI difficult. In this study, such surgical artifacts were excluded in all patients with sufficient tumor removal. Accordingly, the pituitary stalk was clearly identifiable by confirming its anatomic continuity to the HSI and by comparing nonenhanced and enhanced images.
Preoperative HSI on the adenoma surface has been speculated to be due to the accumulation of neurosecretory granules that contain ADH-neurophysin complex along the pituitary stalk (1, 3, 4). However, the mechanism of its formation has not been detailed. Some have also been suggested that it is simply due to adenoma compression against pituitary stalk (5, 6). By closely evaluating our MR studies, the distal end (pituitary gland side) of the HSI was shown to be constantly located at or slightly above the indentation produced by the posterior or lateral margin of the diaphragma sellae (Figs 1–3). The width of the diaphragmatic opening ranges from 3 to 9 mm in length, and the margin of the diaphragmatic opening varies in its rigidity and flexibility (11–13). The degree of the development of the posterior or lateral margin of the diaphragma sellae is reflected by the indentation of the adenoma on sagittal and coronal MR sections
Preoperative HSI commonly accumulates at the distal end of the pituitary stalk just above the diaphragma sellae. On the other hand, poor development of the diaphragma sellae may permit posterior displacement of HSI in the sella turcica, as shown in Figure 4A, or it may result in a more proximal location close to the median eminence, as shown in case Figure 4B (6). Such cases may show that the lack of a particular blockage site results in the accumulation of neurosecretory granules at any portion along the pituitary stalk. These anatomic variations of the diaphragm affect the intensity of the interruption in the transportation of neurosecretory granules and the resultant blockage at the pituitary stalk. This and other cases reveal that the shape of the diaphragma sellae is a major factor that influences the location of HSI.
No patients in group A, whose images demonstrated HSI after surgical resection, had permanent DI, whereas three patients in group B without HSI had permanent DI. This finding indicates that the postoperative presence of HSI reflects the functional integrity of the INH system. In examining patients with polydipsia and polyuria after surgery, the observation of the pituitary stalk is useful in differentiating central DI from other causes.
In pituitary adenomas, central DI is rarely the initial sign (8). Freda et al (14) stated that, in their experience with 828 pituitary adenomas, no cases had an initial presentation of DI. Transient postoperative DI is reported to occur in 10–60% of patients (7). Permanent postoperative DI is relatively rare, with a reported incidence of 0.5–15% (7). The cause of permanent DI is considered to be primary injury or secondary ischemia caused by surgical manipulation of the posterior lobe and/or pituitary stalk; alternatively, latent DI can appear as a result of the perioperative administration of adrenocorticosteroid (7, 8). The findings of this study may suggest that permanent DI more readily occurs in patients without preoperative HSI. In our study, tumor size was not associated with the preoperative presence of HSI. The exact mechanisms that account for the absence of preoperative HSI and its possible relation to DI in some patients with a large pituitary adenomas are undefined; these need to be investigated in the future.
The size and location of the HSI were essentially stable for years, as shown on the postoperative MR images, although the location slightly changed, depending on the compaction of residual tumor or surgical packing. Patients who underwent surgery years earlier are reported (6) to have HSI at the stalk tip, similar to our findings. On the basis of such radiologic evidence, we speculate that HSI is radiologically and functionally stable and possibly functionally equivalent to an ectopic posterior lobe (4). However, this finding needs further pathologic investigation.
Our study has revealed the limitations of the MR evaluation of INH system, as well as its usefulness. One patient in group A did not have permanent DI; the preoperative images did not show HSI. This patient had a huge, irregularly shaped prolactinoma. One patient in group B, in whom permanent DI developed, had an adenoma with HSI due to pituitary apoplexy. We assume that the images in this patient did not show preoperative HSI.
In another patient in group B, one without preoperative HSI, DI did not develop. In this patient, the tumor was not large, irregularly shaped, or hyperintense. The absence of HSI in the pituitary gland is reported in 0–15.6% of healthy subjects without DI (15, 16). In the literature, the absence of HSI in the posterior lobe does not always coincide with clinically overt DI. Our findings and those of other reports show that invisibility of preoperative HSI is attributable to multiple factors (eg, configuration and complex intensities) and that the absence of pre- or postoperative HSI does not always indicate the presence of DI.
Conclusion
Preoperative supradiaphragmatic HSI on the adenoma surface and postoperative HSI reflects the interrupted transportation of ADH-neurophysin complex that is caused by compression of the diaphragma sellae. The shape and degree of the development of the diaphragma sellae, as demonstrated in sagittal and coronal sections, may determine the location of HSI. Its absence suggests impairment of the INH system. The assessment of HSI is useful in predicting the functional integrity of the INH system in patients with a large pituitary adenoma.
Although the clinical importance and usefulness of HSI evaluation in pre- and postoperative T1-weighted images have yet to be determined in pituitary adenomas, HSI should be included as one of the radiologic signs in the evaluation of the functional integrity of the INH system in patients with a large pituitary adenoma. When preoperative HSI is absent, an endocrinologic evaluation of INH system be performed.
References
- Received January 22, 2001.
- Accepted after revision December 27, 2001.
- Copyright © American Society of Neuroradiology