Abstract
BACKGROUND AND PURPOSE: Conventional ultrasonography techniques do not allow visualization of the distal cervical segment of the internal carotid artery (ICA). In a study of patients with severe ICA stenosis, we performed transoral carotid ultrasonography (TOCU) to assess its ability to image this segment of the artery.
METHODS: The study participants consisted of 20 consecutive patients who had severe carotid stenosis and who underwent carotid endarterectomy between 1999 and 2000. TOCU, conventional carotid ultrasonography, and cerebral angiography were prospectively performed before and after carotid endarterectomy.
RESULTS: In all patients, the distal portion of the ICA could be clearly detected by B mode using TOCU and no plaque was observed. The diameter of the distal portion of the ICA significantly increased after carotid endarterectomy (3.9 ± 0.5 mm [mean ± SD]), compared with before (3.5 ± 0.8 mm), when it was estimated by TOCU (P < .01). In seven patients, the postoperative diameter of the distal ICA increased >10%. The mean increase in the postoperative diameter was estimated to be 15.0 ± 23.0% by TOCU, which significantly correlated with the findings (23.9 ± 33.7%) based on cerebral angiography (P < .01). The diameter increased >10% postoperatively in 71% of the patients with the degree of cross-sectional stenosis >95% as shown by carotid ultrasonography and in 86% of the patients whose preoperative diameter was <3.0 mm.
CONCLUSION: TOCU provides additional information regarding the characteristics of the distal ICA that can be obtained neither by conventional carotid ultrasonography nor by angiography.
Multicenter randomized trials revealed evidence that stroke risk is reduced by carotid endarterectomy (CEA) in patients with severe carotid stenosis (1–4). Extracranial carotid lesions are less frequent among Japanese than among North Americans and Europeans. However, a recent report stated that extracranial carotid lesions are increasing, even among Japanese (5). Extracranial atherosclerotic carotid lesions are located within 2 cm from the origin of the internal carotid artery (ICA) in most patients (6). Therefore, conventional carotid ultrasonography is able to depict the carotid lesions in most of the patients with carotid stenosis (7, 8). However, duplex scanning may be difficult when the bifurcation is very high and when the carotid plaque is heavily calcified, thus preventing an adequate insonation of the bulb. In such cases, only a limited area of ICA can be observed by conventional carotid ultrasonography. MR angiography is another method for evaluating the extracranial ICA. However, it often overestimates the grade of carotid stenosis, which can thus lead to an inaccurate interpretation of the results (9).
At present, angiography is the most reliable method for evaluating carotid stenosis. However, using angiography alone, it is also impossible to distinguish whether the distal ICA is narrow as a result of pathologic lesions such as atherosclerosis or as a result of other causes such as hypoplasia. Thus, caution is still required when interpreting the findings.
Recently, a new technique, transoral carotid ultrasonography (TOCU), has been developed to examine the distal extracranial ICA (cervical portion). Yasaka et al (10) reported that TOCU enables us to evaluate the distal ICA noninvasively at the patient’s bedside. This technique might overcome the defects associated with conventional methods, including carotid ultrasonography, MR angiography, and angiography. In the present study, we investigated the vessel diameter and various parameters about the flow velocity, as well as the B-mode appearance of the distal ICA, in patients with high grade carotid stenosis by using TOCU.
Methods
Patients
The study participants consisted of 20 consecutive patients who underwent CEA at our hospital between 1999 and 2000. Therefore, the entry criteria were identical to those for CEA (11). The demographic and clinical characteristics of the patients are summarized in Table 1. TOCU, carotid ultrasonography by external approach, and cerebral angiography were prospectively performed before and after CEA.
Demographic and clinical characteristics
TOCU
ATL HDI 5000 (Hitachi Co.) was used for color Doppler flow imaging. The 12- to 5-MHz linear array transducer was used for conventional carotid ultrasonography. The 9- to 5-MHz convex array transducer, which was originally designed for transrectal probe, was used for TOCU. The methods for TOCU were similar to those described by Yasaka et al (10). The probe tip was painted with echo jelly and was covered with a clean cover and gently touched to the pharyngeal posterolateral wall. The ICA, external carotid artery, and jugular vein could be identified using the B mode, color flow image, and Doppler flow pattern. The appearance of the arterial wall was observed by B mode with TOCU. The diameter and flow velocity of the distal ICA were measured by TOCU before and after CEA. For the present study, we defined the cervical portion of the ICA as the distal ICA. Using TOCU, extracranial ICA was visualized as a vertical linear vessel bent slightly backward. We measured the diameter of the ICA at the nearest point from the surface of the posterior pharyngeal wall (at a depth of approximately 2 cm). Measurement of the diameter was performed three times, and mean value was adopted. However, the obtained values were identical to each other in most cases. TOCU was performed by a neurologist (K.K.) who had performed the examination in approximately 200 patients. Conventional carotid ultrasonography was also performed by the same examiner. These ultrasonography examinations were performed before angiography, and the examiner was thus not aware of the results of the measurements obtained with the other modality (digital subtraction angiography). We examined the relationship between the grade of carotid stenosis and the postoperative dilatation of the distal ICA. The degree of ICA stenosis was calculated by cross-sectional area obtained using conventional carotid ultrasonography and the percent increase in the diameter of the distal ICA was estimated by TOCU.
Operation Procedure
Endarterectomy was performed by a neurosurgeon (T.I.) using a microscope. Anesthesia was introduced by fentanyl citrate, thiamylal sodium, and propofol. The blood pressure, heart rate, blood gas analysis, and various Doppler flow parameters in the carotid artery were monitored, and an electroencephalogram was continuously monitored throughout the procedure. A shunt tube was inserted into both ends of the carotid artery to preserve the bypass flow to the intracranial distal arteries in all except one case. In one case, the distal ICA was clamped during the procedure, because its diameter was too thin for insertion of the tube.
Statistical Analysis
The changes in the parameters before and after CEA were analyzed using the Wilcoxon’s signed-rank-sum test. A regression analysis was used to analyze the relationship between the percent increase in the diameter of the distal ICA estimated by both TOCU and that by cerebral angiography. P < .05 was considered to be significant.
Results
Patient Demographics
Table 1 summarizes the patient characteristics. The patients consisted of 17 men and three women with a mean age of 67.6 ± 6.6 years (mean ± SD). The grade of carotid stenosis was estimated by angiography, using the North American Symptomatic Carotid Endarterectomy Trial method (1) and ranged between 44% and 99%. The average carotid stenosis was 74.9 ± 14.6%. Nineteen of 20 patients had symptomatic carotid lesions. The patients had risk factors that included hypertension (80%), smoking (75%), hyperlipidemia (55%), and diabetes mellitus (35%). They had systemic vascular complications, such as ischemic heart disease (35%) and arteriosclerosis obliterans (10%).
B-Mode Appearance of the Distal ICA
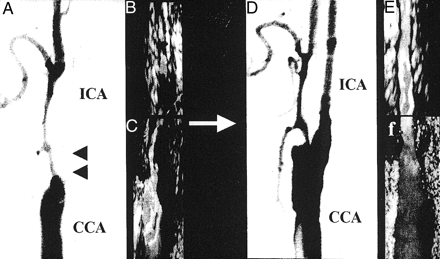
For all patients, the distal ICA could be detected by B mode on TOCU and no plaque was observed. Figure 1 shows collapse of the distal ICA due to severe stenosis located in the origin of the ICA. TOCU clearly discriminates the vessel properties of the distal ICA.
Angiographic and ultrasonographic findings of carotid arteries before (left panels) and after (right panels) CEA. Arrowheads indicate severe stenosis in the origin of the ICA. Note that the distal ICA dilated after CEA.
A, Angiographic findings before CEA.
B, TOCU findings before CEA.
C, Conventional carotid ultrasonographic findings before CEA.
D, Angiographic findings after CEA.
E, Transoral carotic ultrasonographic findings after CEA.
F, Conventional carotd ultrasonographic findings after CEA.
Changes in Flow Velocity in the Distal ICA
The mean flow velocity of the distal ICA showed a slight increase postoperatively (40.7 ± 11.8 cm/s) compared with preoperatively (38.8 ± 12.2 cm/s), although the difference was not statistically significant (P = .5) (Table 2). Neither the peak systolic (69.2 ± 25.6, preoperatively; 71.5 ± 20.9, postoperatively) (P = .3) nor the end diastolic (23.8 ± 7.1, preoperatively; 24.1 ± 7.1, postoperatively) (P = .8) flow velocities were significantly changed after CEA.
Change in parameters before and after carotid endarterectomy
Dilatation of Distal ICA after CEA
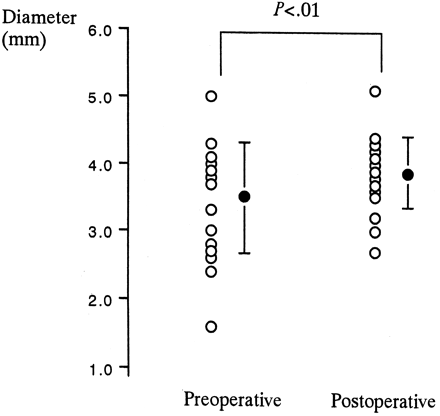
The diameter significantly increased from 3.5 ± 0.8 to 3.9 ± 0.5 mm after endarterectomy (P < .01) (Fig 2). The ratio of the diameter of the ICA to the diameter of the common carotid artery, obtained at an identical level and based on the findings of cerebral angiography, significantly increased after CEA (0.59 ± 0.14) compared with before (0.51 ± 0.26; P < .01). The percent increases in the diameter of the distal ICA estimated by both TOCU and cerebral angiography were 15.0 ± 23.0% and 23.9 ± 33.7%, respectively. The postoperative increase in the distal ICA diameter estimated by two different methods correlated significantly (P < .01) (Fig 3). In seven (35%) of 20 patients, the distal ICA was dilated >10% when estimated by TOCU. In three patients, the postoperative increase in the distal ICA diameter as estimated by cerebral angiography was more remarkable than that by TOCU (Fig 3). The mean flow velocity of the distal ICA in these three patients was significantly lower (25.4 ± 1.4 cm/s) than that in the other patients (41.2 ± 11.7 cm/s) when the Mann-Whitney U test (P < .05) was used.
Diameter of the distal ICA before and after CEA is plotted. Diameter was measured intraorally at the level of the post-pharyngeal portion by using TOCU. Closed circles and error bars indicate mean ± SD.
Comparison of the percent increase in the diameter of the distal ICA as estimated by TOCU and cerebral angiography.
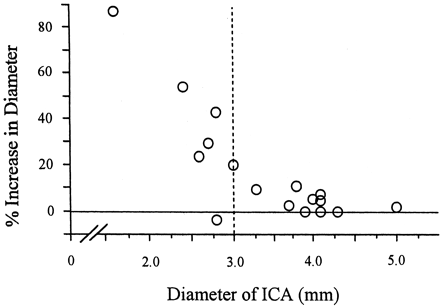
Because the grade of carotid stenosis was higher, the ICA dilated to a much greater extent. The increase in postoperative dilatation of the distal ICA was >10% in five (71%) of seven patients who were shown to have >95% stenosis when carotid ultrasonography was used (Fig 4). In seven patients with an ICA diameter of <3.0 mm before CEA, six patients showed dilatation of the distal ICA of >10% postoperatively (Fig 5).
Relationship between degree of carotid stenosis and postoperative dilatation of the ICA. Percent increase in the diameter of the distal ICA was plotted against the grade of carotid stenosis. Grade of carotid stenosis in a cross-sectional area was estimated by conventional carotid ultrasonography. Diameter of the distal ICA was estimated by TOCU. Dotted line indicates 95% stenosis.
Relationship between preoperative diameter and postoperative dilatation of the ICA. Percent increase in the diameter of the distal ICA was plotted against the preoperative diameter of the distal ICA. Diameter of the distal ICA was estimated by TOCU. Dotted line indicates diameter of 3.0 mm.
Discussion
Randomized clinical studies showed that CEA could reduce the risk of subsequent ipsilateral cerebral ischemia in patients with symptoms of cerebral or retinal ischemia in the distribution of high grade carotid stenosis. An angiographic assessment is indispensable in selecting those patients for whom CEA will be highly beneficial, because the degree of carotid stenosis is estimated by the findings of cerebral angiography. Angiography is essential for visualizing the distribution of atherosclerotic changes in cerebral arteries. However, angiography has some defects: 1) angiography can show the inner rim of the vascular lumen but not provide information regarding the outer diameter; 2) neither the characteristics of the vascular wall nor the degree of stenosis in a cross-sectional area can be evaluated; 3) when the stenosis is extremely severe, the lumen cannot be filled with contrast medium (laminar flow) (12); and 4) cerebral angiography is invasive and not suitable for repetitive examinations.
Conventional carotid ultrasonography is useful for evaluating the common carotid artery or the origin of the ICA and external carotid artery, and it can be performed noninvasively at the patient’s bedside. However, it is still difficult to distinguish severe stenosis or occlusion in the presence of calcificated plaque or in patients with high bifurcation. Moreover, Hass et al (13) reported that frequency distribution of stenosis or occlusion in the cervical portion of the ICA was approximately 9% of 4748 patients with the signs and symptoms of ischemic cerebrovascular disease. Therefore, possible arterial lesions in the distal ICA must be evaluated in patients with carotid stenosis. No detailed information regarding the distal ICA can be obtained by using only conventional carotid ultrasonography. MR angiography is another tool with which to noninvasively evaluate carotid lesions. However, the resolution does not provide sufficiently detailed information regarding the vascular lumen and MR angiography often overestimates the grade of carotid stenosis (9).
When the distal ICA appears to be extensively narrow based on the angiographic findings, the following possibilities exist: 1) the vessel lumen is actually narrow over long distances by atherosclerosis or other organic lesions; 2) the lumen may collapse due to reduced blood flow; 3) the vessel is hypoplastic; or 4) the lumen is not fully filled with contrast medium because of severe stenosis. A differential diagnosis of these conditions is impossible by using only carotid ultrasonography or only angiography. Recently, Yasaka et al (10) reported that the distal ICA can be noninvasively observed by TOCU. It enables us to measure the diameter, cross-sectional area, and flow velocity in the distal ICA. The present results clearly showed that the properties of the distal ICA could be evaluated by TOCU in all cases. TOCU provides more detailed information regarding the ICA in the cervical portion in patients with stenosis of the origin of the ICA.
In the present study, we investigated the characteristics of the distal ICA and its change after endarterectomy. The diameter of the distal ICA significantly increased from 3.5 ± 0.8 to 3.9 ± 0.5 mm after CEA (P < .01). This indicates that the post-stenotic portion of the ICA collapsed because of low blood flow preoperatively and dilated after the removal of stenosis. We next compared the distensibility of ICA after CEA as estimated by TOCU and angiography. In the present study, the percent increase in the diameter of the distal ICA as calculated by angiography was much higher than that calculated by TOCU in three of 20 patients. In these patients, the flow velocity of the distal ICA as detected by TOCU was significantly lower (25.4 ± 1.4 cm/s) than that in other patients (41.2 ± 11.7 cm/s; P < .05). When the lumen is not sufficiently filled up with contrast medium because of laminar flow, the apparent diameter evaluated by angiography might become smaller than that evaluated by TOCU. Therefore, the present results are consistent with the idea that the preoperative diameter of the distal ICA was underestimated by angiography not only because of collapse but also because of laminar flow. As a result, judging the indications for CEA in such patients may be inaccurate.
We also investigated the relationships between the degree of stenosis and postoperative dilatation. As the stenosis became more severe, postoperative dilatation became more prominent. When the cross-sectional area of the lumen is <5%, the distal ICA tends to show remarkable dilatation after CEA (Fig 4). The ICA was dilated after endarterectomy to a great extent, when its diameter was below 3.0 mm (Fig 5). As a result, cross-sectional stenosis >95% in the origin of the ICA or a preoperative diameter of < 3.0 mm in the distal ICA might thus suggest postoperative dilatation. Thus, the preoperative diameter of the ICA as well as the grade of carotid stenosis might predict the distensibility of the distal ICA.
This study is the first report to show dilatation in the post-stenotic portion of the distal ICA after removal of stenosis. In these patients, the grade of carotid stenosis may be underestimated if it is calculated by the North American Symptomatic Carotid Endarterectomy Trial method. Additional studies are needed to elucidate the efficacy of CEA in patients with high grade carotid stenosis and narrow distal ICA. Previous randomized trials evaluated severe carotid stenosis by using only angiography. However, angiography alone cannot provide sufficient information regarding the distal ICA in patients with severe carotid stenosis. It is possible that such patients may not be indicated for CEA if the apparent diameter of the distal ICA is narrow because of collapse and/or laminar flow. In some cases, angiographic findings may lead to inaccurate indications for CEA.
Conclusion
TOCU is a method for providing additional information regarding the distal ICA. In cases in which the preoperative diameter of the distal ICA is <3.0 mm or the degree of cross-sectional stenosis is >95%, a possible collapse of the distal ICA must be considered during differential diagnosis.
Acknowledgments
The authors are grateful to Drs. K. Yasumori, S. Arakawa, and T. Katsuta for useful suggestions.
Footnotes
Supported by Research Grants for Cardiovascular Diseases (12A-2 and 12C-10) from the Ministry of Health and Welfare of Japan.
References
- Received August 27, 2001.
- Accepted after revision February 18, 2002.
- Copyright © American Society of Neuroradiology