Abstract
Summary: Wegener granulomatosis is a multisystemic disorder characterized by necrotizing vasculitis that primarily involves the respiratory tract. The orbits, heart, skin, joints, and nervous system are frequently involved. We describe the MR imaging findings of Wegener granulomatosis in the cervical spine and correlate them with the histopathologic features. MR imaging showed epidural liquid masses surrounded by granulomatous masses that compressed the cervical myelin from behind.
Wegener granulomatosis (WG) is a rare multisystemic necrotizing granulomatous vasculitis that characteristically involves the respiratory tracts and kidneys. The clinical manifestations and symptoms are nonspecific and include systemic findings such as fever, malaise, weight loss, arthralgia, and myalgia. Although most patients initially present with upper airway illness, ophthalmic and neurologic involvement is common (22–54%). The latter manifests as a peripheral or cranial neuropathy (11–21%) or mononeuritis multiplex (1, 2). Meningeal and cerebral involvement (3) is rare. We present a case of WG in a patient with cervical myelopathy caused by involvement of the cervical spine. The patient presented with epidural masses that were detected with MR imaging.
Case Report
WG was diagnosed in a 59-year-old woman who had recurrent episodes of fever. The diagnosis was based on the presence of circulating anti-neutrophil cytoplasmic antibodies (c-ANCA) and the histologic features of a needle biopsy sample from a thoracic lesion, which showed signs of necrotizing vasculitis.
In 1999, the patient reported epistaxis. In December 1999, she developed ulcers of the lower leg. The histologically proved diagnosis of pyoderma gangrenosum was made in March 2000. In July 2000, the patient was admitted to the hospital with fever and severe headache. Chest radiography demonstrated severe pulmonary infiltrates and nodules (Fig 1). The patient reported a neck ache as well as reduced mobility of the cervical spine. The histologic findings in a sample obtained at CT-guided needle biopsy of a pulmonary lesion revealed a necrotizing vasculitis. Pathologically altered laboratory data were as follows: hemoglobin, 6.8 g/100 mL (normal range for women, 13–18 g/100 mL); hematocrit, 0.34 (normal range for women: 0.40–0.52); C-reactive protein, 149.8 mg/100 mL (normal range, <8 mg/100 mL); and c-ANCA titer, 128 relative units per milliliter (normal range, <20 relative units per milliliter of antibodies against proteinase 3). In the urine, the count for protein, leukocytes, and erythrocytes was moderate. Treatment was started with azathioprine (50 mg/d) and prednisone (150 mg/d). The patient then developed signs of a cervical myelopathy with paralysis. MR imaging of the cervical spine was performed by using a head-neck coil and T2-weighted and pre- and postcontrast T1-weighted sequences. Images were acquired in the axial and sagittal planes. Because of pronounced movement artifacts, the image quality was reduced. The T2-weighted images showed epidural hyperintense liquid enclosed by hypointense masses overlying the cervical spinal cord. The myelin was compressed from behind. The process was complicated by herniation of the nucleus pulposus in the C6-C7 segment. On the T1-weighted images, the overlying process was slightly hyperintense relative to CSF, with a hypointense area therein. The masses enhanced after the administration of contrast medium. The inner zone showed no enhancing effect. The meninges showed only a moderate enhancement (Fig 2). We diagnosed the epidural process as inflammatory masses caused by WG. The patient underwent laminectomy for decompression of the spinal cord. During surgery, the dura mater looked thickened. Yellowish necrotic fluid was drained from the epidural space. Histologically, typical features of necrotizing vasculitis were seen. The depicted vessels showed endothelial necrosis, fibrin deposition, and dense granulation tissue with an inflammatory infiltrate. In the surrounding tissue, a chronic inflammatory infiltrate was evident as well (Fig 3). Because of severe headaches, MR imaging of the brain was performed to exclude cerebral involvement. The standard head coil and standard sequences were used: T1-weighted, T2-weighted spin-echo, fluid-attenuated inversion recovery, MR angiographic, and contrast-enhanced sequences. Only a few nonspecific white matter lesions could be detected. No areas of abnormal contrast enhancement were present. The meninges showed no pathologic enhancement. The treatment included cyclophosphamide (2 × 50 mg/d), the administration of azathioprine was ended after the laminectomy, and the prednisone dose was reduced (40 mg/d). The number of pulmonary nodules diminished and the ulcer of the lower leg healed.
Chest radiograph demonstrates pulmonary nodules and infiltrates
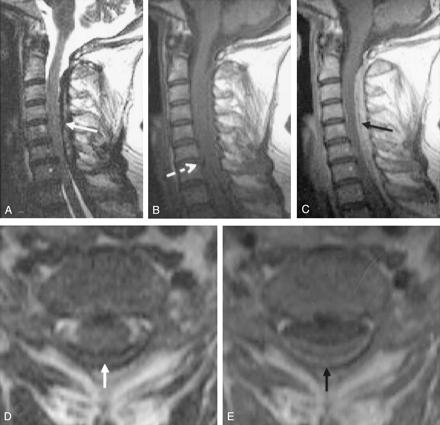
MR images of the cervical spine.
A, T2-weighted sagittal image shows hyperintense fluid (arrow) enclosed by hypointense masses overlying the spinal cord.
B, Sagittal T1-weighted image without contrast enhancement. Arrow indicates a prolapse of the nucleus pulposus.
C, Sagittal T1-weighted image obtained after the administration of gadopentetate dimeglumine demonstrates enhancing granulomatous masses (arrow) and central necrotic hypointense fluid.
D, Axial T2-weighted image shows a hyperintense liquid epidural mass (arrow).
E, Axial T1-weighted contrast-enhanced image shows hyperintense granulomatous tissue surrounding the liquid necrotic mass (arrow).
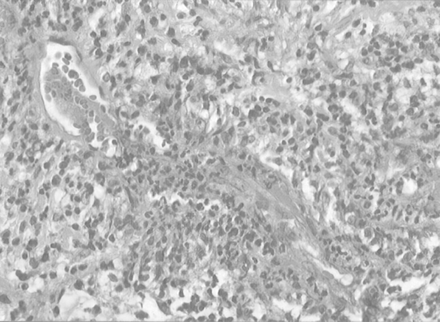
Histologic findings in the surrounding soft tissue with signs of a granulomatous inflammatory process due to WG. The vessels show endothelial necrosis, fibrin deposition, and dense granulocytic tissue with an inflammatory infiltrate.
Discussion
Necrotizing granulomatous lesions of the upper or lower respiratory tracts, a generalized necrotizing vasculitis of arteries and veins, and glomerulonephritis were described as a clinical entity in 1936 (4). Now referred to as WG, the disease involves the paranasal sinuses, lungs, kidneys, eyes, skin, joints, muscles, cardiac system, and nervous system. Circulating c-ANCA is a diagnostic marker in WG (5). ANCAs are directed at proteinase three (PR 3), a serine proteinase (6). Antibodies such as ANCA may augment neutrophilic activation and degranulation, resulting in increased endothelial cell injury. In the follow-up of patients in remission, the c-ANCA titer should be determined, because an increase in the antibody titer is usually associated with clinical exacerbation (7). Moderately elevated c-ANCA titers have also been shown in association with other systemic immune-mediated diseases such as polyarteritis nodosa and systemic lupus erythematosus, as well as in Takayasu disease and Churg-Strauss syndrome (5). These entities can usually be differentiated from Wegener disease on the basis of clinical and serologic findings. The diagnosis of WG requires the presence of a histologically proved granulomatous inflammatory process, focal necrosis, fibrinoid degeneration, and giant cells. The nasal mucosa is an easily accessible biopsy site. In our patient, a CT-guided needle biopsy was performed in an intrapulmonary lesion.
Neurologic involvement in WG primarily involves the peripheral nervous system. Cranial neuropathy is the most common CNS manifestation as a result of a small-vessel vasculitis. In 1963, Drachman (8) proposed several mechanisms for nervous system involvement, as follows: 1) granulomatous involvement by continuous invasion from sites close to the CNS (eg, extension of nasal, paranasal, or orbital granulomata) involving the optic nerve, chiasm, pituitary gland, nasal vestibule, base of the brain, and meninges (26%); 2) CNS granulomatous vasculitis producing mononeuritis multiplex, polyneuritis, myopathy, subarachnoid and intracerebral hemorrhage, and cerebral arterial or venous thrombosis (28%); and 3) granulomatous lesions remote from nasal granulomas and involving the meninges, cranial nerves, and brain and parietal bone (4%). An analysis by Hoffman et al (9) confirmed that mononeuritis multiplex as a typical peripheral manifestation developed in 24 (15%) of 158 patients, and that CNS involvement occurred in 8%. CNS involvement is rarely the initial disease manifestation (2, 3, 5). When stroke does occur in patients with WG, it may be related to the primary manifestations of the disease (eg, vasculitis or occlusion of arterial vessels by granulomatous masses), to the secondary effects (eg, endocarditis), or to causes unrelated to WG (1).
As in other vasculitides, findings at cerebral angiography can be negative in suspected cases of cerebral vasculitis related to Wegener disease because of the involvement of small vessels (50–300 μm). The spectrum of CT and MR findings was demonstrated in an article by Provenzale and Allen in 1996 (1). These findings included dural thickening and enhancement, cerebral and brain stem infarction, and signal intensity abnormalities in the white matter and brain stem (10). The MR imaging appearance in a series of 18 patients by Murphy et al (11) included diffuse linear dural thickening and enhancement (n = 6); focal dural thickening and enhancement contiguous with orbital, nasal, or paranasal disease (n = 5); nonspecific white matter lesions (n = 10); enlarged pituitary gland (n = 2); cerebellar lesions; cerebral atrophy (n = 8); and cerebellar atrophy (n = 2). The authors recommend the use of standard T2- and T1-weighted sequences, as well as of fat-suppressed T1-weighted MR imaging with gadolinium enhancement, to differentiate the thickened dura from the CSF and fat. Granulomatous lesions in the brain parenchyma can appear as homogeneously enhancing or ring-enhancing masses on T1-weighted images and as regions of hyperintense signal intensity on T2-weighted images, typically in the white matter (12, 13). In the report by Murphy et al (11), one patient had diffuse dural thickening and enhancement, both intracranially and overlying the thoracic cord. In our case, MR images of the brain demonstrated only few nonspecific white matter lesions but no meningeal enhancement. An epidural liquid mass overlying the cervical spinal cord was observed; this resulted in a compression of the cord with clinical signs of cervical myelopathy. In our patient, the histologically proved diagnosis of the MR findings was a granulomatous inflammatory process consistent with WG. To our knowledge, one report about a case of WG describes dural thickening in the thoracic cord that causes subacute myelopathy (2). The differential diagnosis for linear meningeal thickening is broad and includes infectious meningitis (including tuberculosis), hypertrophic pachymeningitis; neurosyphilis; neurosarcoid; and dural tumors such as lymphoma, meningioma, and metastases. In rare instances, fibromas can cause dural thickening (1). In most cases, WG can be correctly diagnosed on the basis of associated clinical, laboratory, and radiologic findings.
References
- Received May 29, 2002.
- Accepted after revision July 9, 2002.
- Copyright © American Society of Neuroradiology