Abstract
Summary: Gamma knife radiosurgery is an effective technique for treating arteriovenous malformations. However, treatment failures can occur as a result of incomplete visualization of the arteriovenous malformation nidus. We describe the use of bilateral femoral artery catheterization and simultaneous dual vessel cerebral angiography to facilitate treatment planning during gamma knife treatment.
Stereotactic radiosurgery is a relatively safe and effective means for treating arteriovenous malformations (AVMs) (1). However, incomplete obliteration occurs in 25% to 36% of patients (1–5). Analysis reveals that 26% to 60% of treatment failures result from failure to visualize the entire nidus (1–3). Stereotactic radiosurgery for vascular malformations involves the translation of 2D angiographic data into 3D space. Many larger AVMs supplied by multiple feeding vessels require separate angiograms to completely visualize the entire nidus, thus increasing the complexity of treatment planning. We describe a bilateral femoral artery catheterization technique with the simultaneous injection of two cerebral vessels, which produces a single biplanar image of the entire AVM nidus. This technique facilitates dose planning and may thereby improve the efficacy of treatment.
Methods
Patient Population
To date, seven patients have undergone bilateral femoral artery catheterization to allow for simultaneous dual vessel angiography at our institution. The decision to perform bilateral femoral catheterizations was made by the attending neurosurgeon and neurointerventionalist in cases in which simultaneous delineation of the dual vascular supply provided the most accurate representation of the AVM.
Of the seven treated patients, two were men and five were women. The ages at the time of treatment ranged from 26 to 66 years (Table 1). The mean total AVM volume was 14.6 mL, and the mean marginal dose per treatment was 15.9 Gy. Five patients had initially presented with hemorrhage, and two presented with seizures.
Angiographic characteristics and anatomic locations of the seven treated AVMs
Technique
A stereotactic head frame was placed on the patient’s head the morning of the procedure. After head frame application, high resolution contrast-enhanced MR imaging and then cerebral angiography were performed. For all seven patients, angiography was accomplished with the use of bilateral femoral artery catheterization with two 4-French catheters. Each catheter was placed in the appropriate cerebral vessels. The vessels were first studied independently by biplanar digital subtraction angiography and finally by simultaneous two-vessel angiography in digital subtraction angiography and cut film modalities.
Depending on the size of the AVM, parameters for the injection of contrast material varied from 8 to 10 mL administered at 7 to 9 mL/s for the internal carotid artery and 8 to 10 mL administered at 4 to 5 mL/s for the vertebral arteries. Continuous heparin flush was used between angiographic runs to prevent thromboembolic phenomena.
Radiosurgery was performed with the Leksell gamma knife (Elekta Instruments, Atlanta, GA). The treatment plan was devised jointly by the neurosurgeon and the radiation oncologist.
Results
Seven patients underwent bilateral femoral artery catheterizations for dual vessel simultaneous angiography. Angiograms from three representative cases are shown (Figs 1–3). Dose planning was performed by using the Leksell GammaPlan software provided by Elekta. Nidus volume was determined based on the MR imaging data. The angiographic data were scanned into the software, and the outline of the angiographic nidus was traced on the computer on the anteroposterior and lateral films. These margins were then projected onto the MR imaging data. In each case, the technique was helpful for accurate dose planning. No angiographic complications occurred in any of the patients.
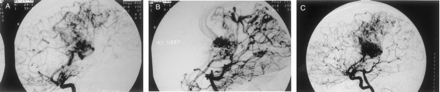
Patient 1.
A, Lateral projection arterial phase image from digital subtraction angiogram of the right internal carotid artery.
B, Lateral projection arterial phase image from digital subtraction angiogram of the right vertebral artery.
C, Dual vessel injections provide better representation of the AVM nidus for radiosurgical planning. The right M1 vessel in this patient was stenotic, resulting in extensive leptomeningeal collaterals.
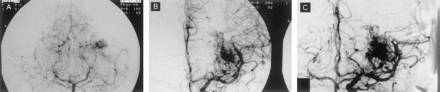
Patient 6.
A, Anteroposterior view angiogram of the left vertebral artery.
B, Anteroposterior view angiogram of the left internal carotid artery.
C, Dual vessel injections provide better representation of the entire nidus in a single biplanar image.
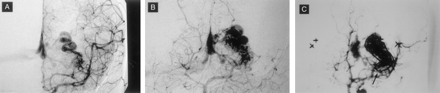
Patient 3.
A, Anteroposterior view angiogram of the left internal carotid artery.
B, Anteroposterior view angiogram of the left vertebral artery.
C, Dual vessel injections show an inferomedial component of the nidus (arrow) that is poorly visualized with the vertebral injection alone (see B).
Discussion
Microsurgical resection remains the most effective treatment for AVMs of the brain. However, depending on the size and location of the lesion, surgery can carry an unacceptable risk of serious neurologic sequela. During the past several years, the development of endovascular and radiosurgical techniques has improved our ability to treat lesions that were previously considered untreatable.
Radiosurgery is effective in treating a wide variety of vascular lesions, yet treatment failures remain a significant problem. Recent studies have reported failure rates that range from 25% to 36%. Factors associated with successful obliteration of AVMs with stereotactic radiosurgery include AVM volume, number of draining veins, patient age, previous embolization, and AVM location (1). After analyzing specific cases, failure of obliteration has been attributed to numerous factors, such as AVM size, intranidal fistula, suboptimal dosing, failure to visualize the entire AVM on the planning images, improper assessment of AVM shape, recanalization of the nidus after embolization, reappearance of AVM vessels that had been compressed by hemorrhage, and radiobiologic resistance (1, 5).
Despite continued advances in radiographic imaging techniques, visualization and appreciation of the complete AVM nidus remains as much an art as it is a science. Among treatment failures, the residual nidus was not included in the original radiation field in 26% to 60% of cases (2–5). Three-dimensional imaging modalities such as MR imaging and MR angiography have improved the accuracy of stereotactic radiation (6, 7). However, conventional 2D angiography remains an integral part of planning. Larger and more complex lesions may derive their blood supply from multiple vessels. In these cases, multiple angiograms are required to visualize each component of the AVM nidus. Dose planning based on multiple angiograms requires mental integration of the images when setting the targets. In one reported case, the difficulties inherent in integrating multiple angiographic studies during treatment planning resulted in failure to incorporate the entire nidus in the treatment field (5).
The difficulty in treatment resides in determining the “true” margin of the nidus. We are looking for a correlation between what appears to be the nidus based on the MR imaging data with the margins as defined by the angiographic data. The problem occurs when there is a disparity between the two sets of data. Often, this occurs in larger, more diffuse lesions with which the actual nidal margin is open to interpretation. The use of multiple angiographic studies results in several sets of projected lines across the MR imaging data, increasing the complexity of the data set. The dual vessel technique allows us to essentially “fuse” two data sets into a single piece of data. Overlay of this data onto the MR data renders a single projection of the summed volume rather than multiple projections composed of overlapping volumes. This simplifies the treatment planning, and it is hoped that it reduces possible errors.
Success rates related to radiosurgery are a function of both the volume and topography of the nidus. Treatment volume has a direct impact on treatment efficacy as a function of radiation dose. That is, smaller volumes can receive higher doses of radiation, thus increasing the likelihood of occlusion. Conversely, larger volumes require lower doses of radiation to keep treatment morbidity low. The tradeoff is a lower likelihood of occlusion.
Within a given volume, a percentage of treatment failure occurs from failure to appreciate the true topography of the lesion. Failure to include small areas of the AVM nidus in the treatment volume may result in a small, persistent residual. It is hoped that the dual vessel technique reduces this specific error by improving our appreciation of the topography of these lesions. Because these volumes are usually only a small percent of the overall treatment volume, they do not result in a change of the treatment dose.
Obvious limitations are inherent in the use of anteroposterior and lateral view angiographic films to evaluate 3D volumes. A recent technique developed by Imbesi et al (8) uses a 2D technique rather than linear 1D measurements to improve nidus measurement. Currently, the use of angiographic data with the MR imaging data set remains complementary. Ultimately, the best evaluation of AVM nidus size and topography would occur with volumetric measurements. As imaging techniques improve, the use of 3D angiographic data derived from either MR angiography or CT angiography might eventually replace the use of conventional angiography.
Recent advances in endovascular techniques have included the simultaneous use of multiple catheters to treat increasingly complex lesions. Bilateral femoral artery catheterizations are used routinely by many busy endovascular services, and our current technique is a natural extension of this experience. The use of simultaneous dual vessel angiography allows us to visualize the entire nidus on a single biplanar study. We think that the use of this technique allows a more accurate representation of the true nidus during gamma knife treatment planning, particularly for lesions crossing watershed vascular zones and situated in periventricular locations where dual choroidal (anterior and posterior) supply is likely.
The overall risks associated with dual femoral puncture are probably small. A recent review of our experience with stereotactic cerebral angiography reveals a total complication rate of approximately 1/1000 (Joseffer et al., unpublished data). Because many of these patients undergo numerous angiograms during their lives, an additional groin puncture in this context should not add a significant amount of additional risk.
Conclusion
Gamma knife radiosurgery can be extremely effective in treating AVMs. However, inaccurate dose planning can contribute to a significant number of treatment failures. Simultaneous dual vessel cerebral angiography may play an important role in improving the efficacy of treatment. Obviously, additional patients and longer follow-up are needed to evaluate the overall impact of this technique in reducing the number of treatment failures.
- Received June 6, 2002.
- Accepted after revision July 18, 2002.
- Copyright © American Society of Neuroradiology