Abstract
BACKGROUND AND PURPOSE: Distinguishing between toxoplasmosis and lymphoma brain lesions in AIDS patients is difficult but clinically relevant. This study was performed to test the hypothesis that diffusion-weighted (DW) imaging with apparent diffusion coefficient (ADC) maps can be used to distinguish these two lesions.
METHODS: The ADC values were calculated in regions of interest placed in 13 brain toxoplasmosis lesions and eight brain lymphoma lesions in patients with AIDS. These values were compared with those of normal white matter. ADC ratios in lymphoma lesions then were compared with those in toxoplasmosis lesions by using a two-sample two-tailed t test.
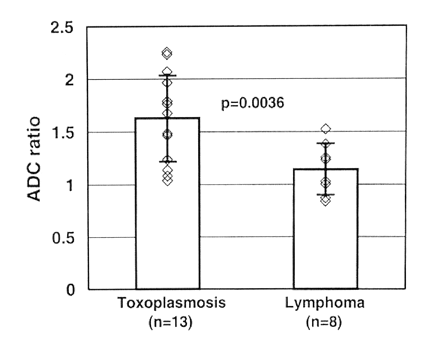
RESULTS: Toxoplasmosis lesions demonstrated significantly greater diffusion than that of lymphoma lesions (P = .004). ADC ratios were 1.63 ± 0.41 (mean ± standard deviation) in the 13 toxoplasmosis lesions and 1.14 ± 0.25 in the eight lymphoma lesions. While considerable overlap was present for ADC ratios between 1.0 and 1.6, ADC ratios greater than 1.6 were associated solely with toxoplasmosis, accounting for seven of 13 toxoplasmosis lesions.
CONCLUSION: ADC values are helpful in differentiating toxoplasmosis from lymphoma in patients with AIDS.
Accurate radiographic differentiation of focal brain masses in patients with AIDS is critical to the timely initiation of appropriate therapy. The two most common etiologies in this population, toxoplasmosis and lymphoma, frequently have similar characteristics on both CT and conventional contrast-enhanced MR images (1, 2). Some of these patients are presumptively treated for toxoplasmosis, and they may proceed to surgical biopsy if their subsequent clinical and radiologic response is unfavorable. By delaying therapy for lymphoma lesions and by exposing the patient to the potential toxicities of anti-Toxoplasma medications, the patient’s neurologic status may deteriorate (3, 4). Furthermore, the concomitant use of steroids during this therapy trial may falsely suggest that a central nervous system lymphoma has responded to Toxoplasma therapy (5).
Recently, diffusion-weighted (DW) MR imaging with apparent diffusion coefficient maps (ADC) has been used as a tool for characterizing infectious and neoplastic brain lesions. Restricted water diffusion has been demonstrated within the cores of brain abscesses (6–14). Numerous investigators have explored the diffusion properties of brain tumors, describing increased water diffusibility within necrotic tumor centers and correlating increased tumor cellularity with restricted diffusion (6, 8, 11–13,15–21). To our knowledge, no groups have reported the MR diffusion properties of infectious abscesses or neoplastic tumors in the AIDS population, whose impaired cellular immune response may confer unique diffusion properties to these lesions. The purpose of this study is to test the hypothesis that toxoplasmosis lesions and lymphoma lesions in AIDS patients can be differentiated by using DW imaging with ADC maps.
Methods
Through a computerized search of electronic medical records at our institution, we identified AIDS patients with proved or presumed (based on response to treatment) brain toxoplasmosis and patients with proved brain lymphoma who were evaluated with MR imaging during 2000–2002. Within this group of patients, initial MR imaging studies that demonstrated rim-enhancing brain lesions larger than 1 cm were included in the institutional review board–approved study. Three investigators (D.L.A.C., J.K.S., M.C.) retrospectively analyzed the conventional contrast-enhanced brain MR images and echo-planar DW images with ADC maps, and differences in opinion were solved by consensus. A diagnosis was established by means of biopsy in all patients with lymphomas and in two patients with toxoplasmosis. Positive serum antibody results and a favorable treatment response at 3 and 6 weeks were used to establish the diagnosis in five patients with toxoplasmosis. Conventional MR imaging was performed on a 1.5-T system (Vision or Sonata; Siemens, Erlangen, Germany) by using precontrast T1- (TR/TE, 500–600/12; matrix, 192 × 256; field of view, 230 × 230 mm) and T2-weighted (TR/TE, 5000–6000; matrix, 192 × 256; field of view, 230 × 230 mm) sequences in multiple planes, and postcontrast (Omniscan; Nycomed, Oslo, Norway) T1-weighted images were acquired in sagittal, axial, and coronal projections by using the same parameters as those of precontrast T1-weighted sequences. ADC maps were acquired by using echo planar diffusion-weighted sequences (TR/TE, 5100/137; matrix, 96 × 128; field of view, 230 × 230 mm) at b values of 50, 500, and 1000 s/mm2 in all patients. Diffusion gradients were applied in the z, y, and x directions.
Postprocessing of the ADC maps was performed offline on an independent computer workstation. For purposes of analysis, we chose only those lesions measuring more than 1 cm in diameter. For each lesion, regions of interest (ROIs) were manually selected in the nonenhancing central core of the lesion and in contralateral, normal-appearing white matter. ROI diameters were approximately one-half the diameter of the portion of the lesion within the enhancing rim. An ADC ratio was computed as the ratio of the mean ADC within the lesion to the mean ADC in normal white matter. Lymphoma ADC ratios were compared with toxoplasmosis ADC ratios by using a two-sample two-tailed t test, with an assumption of unequal variances. A significant difference was accepted if the P value was less than .05.
Results
We identified seven patients with toxoplasmosis (13 lesions) and four with lymphoma (eight lesions) (Table 1). The ages of the patients with toxoplasmosis (five men and two women) ranged from 26 to 46 years (mean, 35.7 years), and the ages of the lymphoma patients (three men and one woman) ranged from 36 to 51 years (mean, 40.8 years). In both the lymphoma group and the toxoplasmosis group, T2-weighted images demonstrated edema around all lesions, and gadolinium-enhanced T1-weighted images showed rim enhancement with a dark central core. Toxoplasmosis lesions demonstrated diffusion significantly greater than that of lymphoma lesions (P = .004) (Figs 1–3). ADC ratios were 1.63 ± 0.41 (mean ± SD) (range, 1.04–2.26) in the 13 toxoplasmosis lesions and 1.14 ± 0.25 (range, 0.84–1.52) in the eight lymphoma lesions. While considerable overlap was present for the ADC ratios between 1.0 and 1.6, ADC ratios greater than 1.6 were associated solely with toxoplasmosis, accounting for seven of 13 toxoplasmosis lesions. No toxoplasmosis lesion had an ADC less than that of uninvolved white matter.
ADC ratios in toxoplasmosis and lymphoma lesions. Column heights and error bars represent the mean and SD, respectively. ADC ratios in the toxoplasmosis lesions are significantly greater than those in the lymphoma lesions.
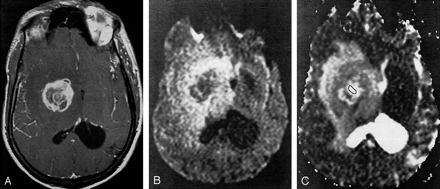
Axial images in an AIDS patient with toxoplasmosis.
A, Axial T1-weighted gadolinium-enhanced MR image. The lesion in the right basal ganglia has an irregular, enhancing rim.
B, DW image (b = 1000 s/mm2). The core of the lesion demonstrates unrestricted diffusion.
C, ADC map of a toxoplasmosis lesion in the right basal ganglia. The outline indicates the ROI within the lesion used for ADC computation. The core of the lesion has a mean ADC that is increased relative to that in normal white matter (ADC ratio, 2.23).
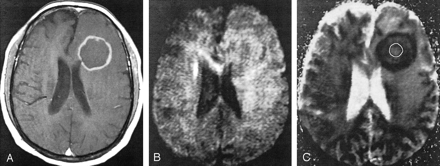
Axial images in an AIDS patient with lymphoma.
A, Axial T1-weighted gadolinium-enhanced MR image. A lesion with an enhancing rim is present in the left frontal lobe.
B, DW image (b = 1000 s/mm2). The signal intensity of the core of the lesion is similar to that of uninvolved white matter.
C, ADC map of a lymphoma lesion in the left frontal lobe. The outline indicates the ROI within the lesion used for ADC computation. The core of the lesion has a mean ADC that is similar to that of normal white matter (ADC ratio, 1.25).
Findings in seven patients with toxoplasmosis (13 lesions) and four with lymphoma (eight lesions)
Discussion
Given the difficulty in differentiating infectious lesions from malignant brain masses in AIDS patients by using conventional CT and MR imaging and given the potential morbidity associated with delayed diagnosis and surgical biopsy, additional noninvasive tools are needed to direct appropriate therapy. Numerous investigators have advocated the use of thallium-201 single photon emission CT as a means to distinguish lymphoma from nonmalignant brain lesions in AIDS patients, demonstrating sensitivities of as high as 100% (22–25). Recent studies, however, yielded less promising results, with sensitivities of 60% and 71%, respectively (26, 27). Positron emission tomography (PET) has been shown to be useful in the accurate differentiation of hypometabolic toxoplasmosis lesions (28, 29), with a reported sensitivity and specificity of more than 90% (28). In one series (27), the sensitivity of PET was lower (50%–66%), but the authors acknowledged that their small sample sizes could have influenced their results. The limited availability of PET scanners and the substantial cost of the study may preclude its widespread use in this clinical setting. Proton MR spectroscopy has also been used to study brain lymphoma and toxoplasmosis. Chang and Ernst (30) and Chang et al (31) found unique metabolite profiles in toxoplasmosis and lymphoma lesions by using MR spectroscopy. In contrast, two other studies (27, 32) demonstrated an overlap in the metabolite profiles of these lesions, and the investigators suggested that other imaging modalities may be more reliable in this setting. Ernst et al (3) used perfusion MR imaging and reported a significantly increased relative cerebral blood volume in lymphoma lesions. They recommended further studies with larger sample sizes to confirm their findings.
Contrary to many of the previously mentioned methods that require special acquisition and postprocessing, DW imaging is readily available and rapidly performed, and the images are easily postprocessed. Because of these reasons, we sought to determine if DW imaging could be used to differentiate toxoplasmosis from lymphoma in patients with AIDS. In our study, the ADC values in toxoplasmosis lesions were significantly greater than those in lymphoma lesions. This finding suggests that this method might provide valuable additional information in differentiating toxoplasmosis from lymphoma in AIDS patients.
All toxoplasmosis lesions in our patients revealed increased diffusion relative to that in white matter, in contradistinction to the restricted diffusion within brain abscesses described in numerous earlier investigations (6–14). Toxoplasmosis ADC ratios in our study were 1.63 ± 0.41, with no ADC ratio less than 1.0, whereas mean ADC ratios within brain abscesses in other studies ranged from 0.36 to 0.83 (7, 8). Chang and Ernst (30) reported an ADC ratio of 1.05 in a single toxoplasmosis lesion. Whether the ROI used for ADC computation in that last study included the contrast-enhancing rim is unclear. The restricted diffusion within a brain abscess is thought to be due to a restriction of microscopic movement of water molecules within a complex matrix of proteins, inflammatory cells, cellular debris, and bacteria in high-viscosity pus. Water molecules in abscesses are bound to carboxyl, hydroxyl, and amino groups on the surfaces of macromolecules, which further restrict their translational motion. Like other abscesses, the abscesses of toxoplasmosis consist of an outer astroglial reaction that surrounds a proliferating zone of capillaries and fibroblasts, which in turn surround a central region of dead and dying brain infiltrated by polymorphonuclear leukocytes and histiocytes (2). Increased diffusion within toxoplasmosis lesions may reflect relatively decreased viscosity within their central cores; this is perhaps due to an impaired cellular immune response in these immunocompromised patients. Further pathologic studies to quantify the viscosity and cellularity, both in toxoplasmosis abscesses and in other brain abscesses, may provide further insight into these radiologic findings.
In our patients, diffusion within lymphoma lesions was more restricted than in toxoplasmosis lesions, and this difference likely reflected the highly cellular, aggressive nature of lymphoma lesions, even within their nonenhancing central cores. Lymphoma ADC ratios in this study (1.14 ± 0.25) were similar to those reported by Guo et al (1.15 ± 0.33) (33). Several studies have demonstrated that diffusion within lymphoma lesions is typically less than that in most other brain malignancies (13, 18, 20, 35). Guo et al (33) also compared the cellularity in high-grade astrocytomas and lymphomas by histologically quantifying the nuclear-to-cytoplasmic ratio (N/C). They found that mean N/C was 1.45 in lymphomas and 0.24 in high-grade astrocytomas, while the mean ADC ratio was 1.15 in lymphomas and 1.68 in high-grade astrocytomas. They concluded that the higher cellularity of lymphoma lesions contributes to their more restricted diffusion.
The results of our study suggest that ADC values can play a role in the therapeutic algorithm for neurologically impaired AIDS patients whose brain lesions show a nonspecific appearance. If a focal brain mass is identified on initial brain MR images and if the ADC ratio in that lesion is greater than 1.6, the patient may be able to safely tolerate a trial of antitoxoplasma therapy. However, if the ADC ratio is less than 1.6, and particularly if it is less than 1.0, a lower clinical threshold should be used for advancing to other imaging modalities or early surgical biopsy. This information should be readily available in most patients, as DW imaging with ADC mapping are currently included in most routine brain MR imaging protocols.
Our study has some limitations. One is that the ADC maps were computed by using three b values (50, 500, and 1000 s/mm2). However, Burdette et al (34) reported a mean percentage difference of 0.84% between ADCs calculated by using a six-point technique (b = 1, 100, 200, 500, 800, and 1000 s/mm2) and ADCs calculated by using a two-point technique (b = 1 and 1000 s/mm2). Their results suggested that potential errors introduced by using only a three-point technique should be negligible. The method used for drawing ROIs and computing ADC ratios were standardized across all lesions and performed by a single observer. We were not, however, blinded to the diagnosis of each lesion at the time of ADC computation. Further, although the difference in mean ADC ratios in the two groups was statistically significant, the relatively small sample sizes currently preclude the prescription of a threshold ADC ratio for use in the clinical setting, especially given the substantial overlap between the groups involving ADC ratios of 1.0–1.6. Blinded, prospective studies with larger sample sizes are needed to firmly establish the sensitivity and specificity of this tool in differentiating toxoplasmosis from lymphoma in patients with AIDS.
Conclusion
Our results demonstrate that ADC values are helpful in distinguishing toxoplasmosis from lymphoma brain lesions in patients with AIDS. We found that lesion-white matter ADC ratios greater than 1.6 were seen only in toxoplasmosis. Because DW images and ADC maps are easily obtained, postprocessed, and interpreted, this technique may play an important role in the early diagnosis of these lesions, and the results may influence the care of AIDS patients for whom brain images show lesions with nonspecific appearances.
References
- Received September 23, 2002.
- Accepted after revision November 12, 2002.
- Copyright © American Society of Neuroradiology