Abstract
Summary: We present the case of a 14-month-old male infant who underwent diffusion tensor imaging within 24 hours of traumatic brain injury. Although conventional MR findings that included those of diffusion-weighted imaging were unremarkable, full-tensor diffusion imaging revealed striking abnormalities in regions of brain that subsequently developed overt swelling. This case engenders important hypotheses regarding mechanisms of traumatic brain swelling and anisotropic diffusion of water in pathologic tissues.
Early and accurate diagnosis of traumatic brain injury in the pediatric population remains an important component in the initial care of the patient with head trauma. Modern neuroimaging plays a primary role in this assessment. New techniques, particularly in MR imaging, may extend the role of the neuroradiologist in evaluating the earliest effects of head trauma. We present the case of a 14-month-old male infant with head trauma who showed unexpected and previously unreported findings on diffusion tensor images but no apparent abnormalities on routine MR images or on standard (not full tensor) diffusion-weighted images.
Case Report
A 14-month-old male infant with an unremarkable birth history and a medical history significant only for asthma presented to an outside hospital after a “fall.” The mother reported falling on top of the child and said that, although the child initially cried, he became unarousable after a few minutes. Physical examination revealed decreased level of consciousness with dilated, unreactive pupils, horizontal nystagmus to the right, and bitemporal ecchymoses. The child’s oxygenation saturation dropped into the 80% range, prompting intubation and helicopter transfer to our institution.
Cranial CT performed on admission (not shown) revealed a minimally depressed right occipital fracture; a thin (2–3 mm) subdural hematoma around much of the right cerebral hemisphere, along the interhemispheric fissure and along the tentorium; and an approximately 5-mm right-to-left midline shift. No evidence of intraparenchymal hemorrhage or contusion was seen.
MR imaging was performed 18 hours after admission (within 24 hours of the initial injury) on a 1.5-T GE CVi Signa system (GE Medical Systems, Milwaukee, WI) with a standard birdcage head coil. In addition to conventional sequences (T1-weighted 3D spoiled gradient-echo, T2-weighted fast spin-echo, T2*-weighted magnetization-prepared gradient-echo, and T2-weighted fluid-attenuated inversion recovery, diffusion-weighted imaging was performed by using both a standard clinical protocol (spin-echo echo-planar imaging with diffusion encoding in three orthogonal directions, b = 0 and 1000 s/mm2) and a full-tensor protocol (23 encoding directions, same b values as that of the standard clinical protocol). Maps of mean diffusivity (directionally averaged apparent diffusion coefficient [ADC]) were generated from both sets of diffusion-weighted data, and maps of fractional anisotropy (FA) (1) were generated from the full-tensor data set.
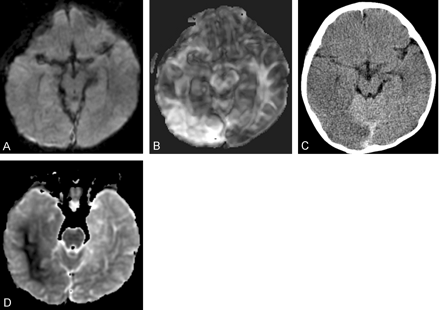
Conventional MR images (not shown) revealed no change in size of the right hemispheric subdural hematoma and no evidence of parenchymal injury. Diffusion-weighted images (Fig 1A) and ADC maps (not shown) obtained by using the standard clinical protocol also revealed no evidence of parenchymal injury. However, FA maps obtained by using the full-tensor protocol (Fig 1B) revealed striking abnormalities in relatively large areas of cortical and subcortical right hemisphere. Specifically, markedly increased anisotropy and mildly increased mean diffusivity in portions of right frontal, temporal, parietal, and occipital lobes existed. No evidence of hydrocephalus, venous sinus thrombosis, or vascular injury was found.
Images obtained in a 14-month-old male infant with traumatic brain swelling.
A, Conventional diffusion-weighted image (three encoding directions) obtained 18 hours after admission shows no evidence of parenchymal injury. The accompanying ADC map as well as conventional T2-weighted fast spin-echo and T2-weighted fluid-attenuated inversion recovery images (not shown) were equally unremarkable.
B, Fractional anisotropy map from full-tensor acquisition obtained 18 hours after admission (concurrent with A) shows readily apparent parenchymal abnormality characterized by increased FA in right occipital and posterior temporal lobes. An accompanying full-tensor ADC map (not shown) revealed minimally increased diffusivity in the affected region.
C, Follow-up CT scan obtained 93 hours after admission shows region of parenchymal edema characterized by ill-defined hypoattenuation and poor gray matter–white matter discrimination corresponding to earlier diffusion tensor abnormality (compare with B).
D, Conventional ADC map obtained 135 hours after admission shows a large area of reduced diffusivity in the affected region of the right hemisphere, primarily in white matter but also involving some cortex. Conventional T2-weighted fluid-attenuated inversion recovery images (not shown) revealed diffuse cortical swelling in affected areas.
The child was admitted to the pediatric intensive care unit. On day 3 of his hospitalization, he developed focal seizures and weakness involving his left side. Follow-up CT scans obtained at 69 and 93 hours after admission (Fig 1C) showed parenchymal edema manifesting as ill-defined hypoattenuation and poor gray-white differentiation throughout regions of the right hemisphere corresponding to the distribution of abnormalities revealed by diffusion-tensor imaging 2 days earlier. The subdural hematoma had decreased in size. Electroencephalography revealed asymmetric background slowing, maximal in the posterior quadrant of the right hemisphere, but no epileptiform activity. The patient was started on Dilantin and phenobarbital, and seizure activity did not recur.
Follow-up MR imaging obtained 135 hours after admission showed diffuse swelling of cerebral cortex throughout much of the right cerebral hemisphere. Diffusion-weighted imaging and ADC maps (Fig 1D) revealed markedly restricted diffusion throughout most of the right hemispheric white matter and some involvement of cortex. The FA map (not shown) had essentially normalized, with the exception of subtle, residually increased anisotropy in the right frontal lobe. Over the next few days, the child showed steady improvement in mobility but continued to favor his right side. A whole-body bone scan obtained 10 days after admission revealed multiple fractures, and ophthalmologic examination revealed retinal hemorrhages. He was discharged to foster parents in another city and was lost to follow-up.
Discussion
Various CT and MR imaging patterns of injury in pediatric head trauma have been described (2–4). In an evaluation of routine CT and MR imaging studies of children with a history of traumatic brain injury, Ewing-Cobbs et al (5) demonstrated a correlation between inflicted brain injury and subdural interhemispheric and convexity hemorrhages as well as signs of preexisting brain abnormality, including cerebral atrophy, subdural hygroma, and ex vacuo ventriculomegaly. Others have sought to affirm the role of MR imaging in the evaluation of pediatric head trauma by describing patterns of abnormalities more readily identified by MR than CT imaging (6, 7).
Conventional diffusion-weighted imaging is well established as an exquisitely sensitive MR technique in the detection of acute brain ischemia and is now a routine part of many MR imaging studies. Recent studies have suggested that diffusion-weighted imaging may be a sensitive indicator of blunt head trauma in the pediatric population and one that may be helpful in the early detection of infarction in children with nonaccidental head trauma. Suh et al (8) retrospectively evaluated 18 children with presumed nonaccidental head trauma who underwent diffusion-weighted imaging within 5 days of presentation. Sixteen (89%) showed abnormalities on diffusion-weighted images and ADC maps, and in 13 (81%) of the positive cases, diffusion-weighted imaging revealed more extensive brain injury than was demonstrated by conventional MR imaging or showed injuries not observed on conventional MR images. The authors suggested that diffusion-weighted imaging might have broad applications in the early detection of infarction in children with nonaccidental head injury.
Whereas in conventional diffusion-weighted imaging the diffusivity of water is typically measured in three orthogonal directions and these values averaged, a more complete and accurate description of the diffusion properties of tissue is obtained by estimating the full diffusion tensor model, which requires measurements in at least six noncollinear directions (9). From the diffusion tensor, maps of diffusion anisotropy and direction of maximum diffusivity can be generated; such maps have been shown to reflect properties of tissue microstructure (1). Clinical applications for such maps are being identified with increasing frequency, but little is known about diffusion tensor findings in the setting of blunt head trauma.
In five adult patients with traumatic focal contusions or hematomas who underwent diffusion tensor imaging, Jones et al (10) described a reduction in mean diffusivity in both gray and white matter without an associated increase in T2-weighted signal intensity. The authors suggested that this change might represent either a partial redistribution of water from the extra- to intracellular compartment or a reduction in the diffusivity of water in the cytosolic environment. In two recent studies of diffusion tensor imaging in blunt head trauma, diffusion anisotropy was found to be decreased in normal-appearing cerebral white matter on conventional MR images and to have either normal (11) or increased (12) mean diffusivity. The authors postulated several possible mechanisms, including axoplasmic transport impairment or axolemmal disruption, misalignment, or increased permeability.
In the present case, we observed transiently increased mean diffusivity and diffusion anisotropy in gray matter preceding the development of overt cortical swelling. We are aware of no previous reports describing increased diffusion anisotropy in traumatic brain injury; indeed, there are few published reports of pathologic anisotropy elevations of any cause. There are several reports in the literature of transiently increased diffusion anisotropy in acute cerebral ischemia or infarction (13–16), but all of these cases were associated with reduced mean diffusivity, in contrast to the present case, in which the anisotropy elevation was associated with increased mean diffusivity. Examination of diffusion tensor eigenvalues in the affected regions revealed that the observed ADC and FA changes resulted from an increase in the largest eigenvalue (λ1) coupled with a proportionally smaller decrease in the smallest eigenvalue (λ3).
Before postulating potential mechanisms for the present findings, it is important to confidently exclude the possibility that the findings are artifactual or the result of something other than intrinsic brain tissue disease (such as the overlying subdural hematoma). The correlative CT (Fig 1C) and follow-up MR findings lend much support to the validity of the initial diffusion tensor findings, which we were unable to explain on the basis of susceptibility effects, eddy current distortions, motion effects, or any other known sources of error. The electroencephalographic findings also supported the diagnosis of focal right hemispheric injury. We considered the possibility that locally anisotropic background gradients induced by the subdural hematoma or occult subarachnoid hemorrhage might have caused falsely elevated anisotropy measurements, but this seems highly unlikely in light of the results of Beaulieu and Allen (17). To further exclude an artifactual cause of the findings, we calculated the mean and variance of signal intensity for the 23 sets of diffusion-weighted images, discarded those sets for which the signal intensity was more than 1 SD away from the mean (three sets met this criterion), and recalculated the diffusion tensors from the remaining 20 sets of diffusion-weighted data. This reduced the conspicuity of the abnormalities (as would be expected even for truly anisotropic tensors), but they remained clearly visible (data not shown).
We can only speculate as to the pathophysiologic mechanism behind the diffusion tensor findings. The sources of diffusion anisotropy in biologic tissue, even in its normal state, are only beginning to be elucidated. Initially it might seem reasonable to exclude cytotoxic edema as an important mechanism in this case, because numerous studies have shown this to cause reduced mean diffusivity, possibly as a result of the shrinking and increased tortuosity of the extracellular space engendered by water shifting from extra- to intracellular compartments (18); however, this fluid shift must require finite time to occur and little is known about the behavior of the diffusion tensor while the shift is in progress. It is conceivable that the transmembrane flux of water might be anisotropic even in gray matter and that there might be a transient increase in apparent directional diffusivity associated with this flux, up to the point where cellular swelling becomes the predominant effect, mean diffusivity falls, and anisotropy normalizes. Both the follow-up CT and MR imaging indicated the subsequent development of cortical swelling and normalization of anisotropy.
Complicating the interpretation further is the potential contribution of vasogenic edema. In contrast to cytotoxic edema, vasogenic edema has been shown to increase mean diffusivity but decrease anisotropy (19). Vasogenic edema favors the white matter because the microstructural environment there is more susceptible to the accumulation of extracellular fluid; cerebral cortex is not immune to vasogenic edema, but it is seldom observed on MR images, and the diffusion tensor appearance of vasogenic edema in gray matter has not been well studied. Furthermore, vasogenic and cytotoxic edema have been shown to coexist and develop along different time scales after traumatic brain injury, with a transient increase in vasogenic edema being followed by a more slowly developing cellular edema (20). Therefore, the relative contributions of cytotoxic and vasogenic edema to the diffusion tensor in the present case are uncertain, and our diffusion tensor findings could reflect a complex, dynamic interplay between the two.
A vascular explanation for the findings is worth considering, because perfusion at the capillary level is known to be a potential source of pseudodiffusion effects on diffusion-weighted images (21). This, however, is an unlikely explanation, because diffusion-weighted imaging is relatively insensitive to perfusion effects at b values as high as 1000 s/mm2 (21) and the relative volume fraction of the intravascular compartment is likely too small to have an effect as large as we observed.
Conclusion
We speculate that the increased diffusion anisotropy and mean diffusivity preceding the development of overt brain swelling reflect one moment in a complex and dynamic response to traumatic injury, characterized by cellular and vasogenic edema coexisting in unknown proportions within tissue of ordered microstructure. Shear forces sustained at the time of blunt trauma may have ruptured cellular membranes in a directionally ordered manner. Systematic study of the diffusion tensor in traumatic brain swelling will likely elucidate the nature of this complex phenomenon and allow firmer conclusions than what can possibly be drawn from this isolated case.
Acknowledgments
We wish to thank Drs. Victor Haughton and Howard Rowley for their comments on the manuscript.
References
- Received August 12, 2002.
- Accepted after revision December 16, 2002.
- Copyright © American Society of Neuroradiology