Abstract
BACKGROUND AND PURPOSE: Nonimaging transcranial Doppler sonography (TCD) and imaging TCD (TCDI) are used for determination of the risk of stroke in children with sickle cell disease (SCD). The purpose was to compare angle-corrected, uncorrected TCDI, and TCD blood flow velocities in children with SCD.
MATERIALS AND METHODS: A total of 37 children (mean age, 7.8 ± 3.0 years) without intracranial arterial narrowing determined with MR angiography, were studied with use of TCD and TCDI at the same session. Depth of insonation and TCDI mean velocities with and without correction for the angle of insonation in the terminal internal carotid artery (ICA) and middle (MCA), anterior (ACA), and posterior (PCA) cerebral arteries were compared with TCD velocities with use of a paired t test.
RESULTS: Two arteries were not found on TCDI compared with 15 not found on TCD. Average angle of insonation in the MCA, ACA, ICA, and PCA was 31°, 44°, 25°, and 29°, respectively. TCDI and TCD mean depth of insonation for all arteries did not differ significantly; however, individual differences varied substantially. TCDI velocities were significantly lower than TCD velocities, respectively, for the right and left sides (mean ± SD): MCA, 106 ± 22 cm/s and 111 ± 33 cm/s versus 130 ± 19 cm/s and 134 ± 26 cm/s; ICA, 90 ± 14 cm/s and 98 ± 27 cm/s versus 117 ± 18 cm/s and 119 ± 23 cm/s; ACA, 74 ± 24 cm/s and 88 ± 25 cm/s versus 105 ± 23 cm/s and 105 ± 31 cm/s; and PCA, 84 ± 27 cm/s and 82 ± 21 cm/s versus 95 ± 23 cm/s and 94 ± 20 cm/s. TCD and angle-corrected TCDI velocities were not statistically different except for higher angle-corrected TCDI values in the left ACA and right PCA.
CONCLUSION: TCD velocities are significantly higher than TCDI velocities but are not different from the angle-corrected TCDI velocities. TCDI identifies the major intracranial arteries more effectively than TCD.
Vasculopathy and narrowing of the major cerebral arteries in children with sickle cell disease (SCD) evolve over time and lead to cognitive decline and cerebral infarction if preventive treatment is not introduced in a timely fashion.1,2 The Stroke Prevention Trial in Sickle Cell Anemia (STOP) established that chronic transfusion therapy can reduce the risk of a first stroke by 92% in high-risk children, who were selected by screening with conventional nonimaging transcranial Doppler sonography (TCD).1,3 Although the incidence of stroke has been dramatically impacted by the implementation of STOP study guidelines for TCD testing, there is still a need to find a better tool to estimate the risk of stroke because TCD is limited by poor reliability of flow velocity measurements.4
A newer method, imaging transcranial Doppler sonography (TCDI), has the potential to be more accurate in the estimation of risk of stroke because, in contrast to TCD, it allows outlining of parenchymal structures and visualization of the examined vessels.5 These features can improve the reliability of identification of vessels and accuracy in placing the sample volume in a particular vascular segment. Consequently, many centers use TCDI because they assume that it provides comparable information on velocity.6–8 TCDI also enables an operator to determine an angle between the course of an artery and the ultrasound beam and correct measurements for cosine of the angle. However, angle-corrected TCDI velocities are not widely used because it has been suggested that they may overestimate the risk of stroke.9,10 Although the quality of information provided by TCDI measurements is probably better than that provided by TCD, the suggestion by Nichols et al10 is likely because angle-corrected TCDI velocities are not compatible with commonly used TCD diagnostic thresholds. The purpose of this study was to compare angle-corrected and uncorrected TCDI blood flow velocities with TCD velocities in children with SCD who had no neurologic deficits and no intracranial arterial narrowing.
Materials and Methods
Patient Population
The study population has been drawn from the Comprehensive Sickle Cell Center (CSCC) at our institution on the basis of the following criteria: 1) SCD genotype: homozygous SS (SCD-SS, confirmed by isoelectric focusing with DNA-based confirmatory testing or parental studies), 2) age: minimum 2 years, maximum 12 years, 3) no deficits on neurologic examination, 4) no history of stroke, and 5) approval and informed consent of subject's caretaker and subject's assent. The following exclusion criteria were also used: 1) history of major head injury requiring visit to an emergency department, 2) history of seizure disorder requiring anticonvulsant therapy, 3) chronic transfusion therapy, 4) history of prenatal and perinatal hypoxic-ischemic brain injury, and 5) evidence of HIV infection.
Thirty-seven children with SCD (mean age, 7.8 ± 3.0 years; 20 male, 17 female), recruited during a 2-year period, underwent TCD, TCDI, and MR angiography (MRA) studies. Three children, who underwent all the studies twice in a 1-year interval, were also included in the analysis.
Protocol Procedures
We obtained approval from our institutional ethics committee for this prospective study. After obtaining informed consent, we collected demographic information including age, sex, race, developmental history, information on the health status of the child from the last clinic visit, and last sickle cell crisis. We also collected data on hemoglobin concentrations and hematocrit levels. Children with no abnormalities on neurologic examination were scheduled for brain assessment with TCD, TCDI, and MRA. MRA in our institution is routinely scheduled once a year for children with SCD.
The consecutively recruited children underwent 2 complete transcranial Doppler examinations during the same visit and 10 minutes apart from each other: one performed with TCD and one performed with TCDI. One of 3 well-trained sonographers performed both examinations. An expert (J.K.) with more than 10 years of experience in TCD and TCDI supervised the imaging examinations. In each case, the same sonographer performed the TCD and TCDI examinations. The children were not permitted to sleep during the examinations and were not sedated.
Nonimaging Transcranial Doppler Sonography
TCD studies were performed according to the STOP protocol, described in detail below, by a STOP II-certified sonographer,10 who was supervised by a senior pediatric neuroradiologist. The TCD defaulted to a 6-mm sample volume, while gain settings were optimized to measure the peak-systolic velocity. The angle of insonation was assumed to be 0° degree. Blood flow velocities from the major cerebral arteries were measured through transtemporal and occipital windows with the use of a 2-MHz probe (Pioneer TC 8080; Nicolet Vascular, Madison, Wis). The mean time-averaged maximum velocity (Vmean) of the terminal portion of the internal carotid artery (ICA), M1 segment of the middle cerebral artery (MCA), A1 of the anterior cerebral artery (ACA), P1 or P2 of the posterior cerebral artery (PCA) and basilar artery (BA), and V4 segments of the vertebral arteries were measured over at least 3 complete cardiac cycles. Insonation of the arteries was performed in 2-mm increments, and at each depth, a sweep of at least 3 spectral waveforms were recorded. The mean blood-flow velocity was calculated manually as the midpoint between the peak systolic and end-diastolic velocity. The sonographer selected the most representative waveform with the highest velocity from each arterial segment to determine manually the mean velocity by moving a horizontal cursor to visually equalize the area of the waveform above and below the cursor. Electronic readings were available, but we selected the manual readings for the study to be consistent with the STOP protocol.
Imaging Transcranial Doppler Sonography
The TCDI study was performed immediately after TCD examination by the same sonographer supervised by the expert. The role of the expert was to assure proper placement of the sample volume in a specific arterial segment and accurate measurement of the angle of insonation. We identified the terminal portion of the ICA, M1 segment of the MCA, A1 segment of the ACA, and P1 segment of the PCA via the temporal acoustic windows in an oblique axial plane using standards previously described in detail.5 The BA and V4 arteries were visualized via the occipital window. We used an HDI 5000 sonographic scanner (Philips Medical Systems, Bothell, Wash) equipped with a 1.8–3.6 MHz 90° phased-array probe for both B-mode and Doppler imaging. To determine the angle of insonation, we placed a sample volume adjusted to the size of the insonated artery and a linear marker provided by the scanner software under visual guidance on the color image of the artery being insonated and fitted its direction to be oriented along the long axis of the vessel. This allowed the angle-corrected blood flow velocities to be measured.
At each arterial segment, angle-corrected velocities were measured once. The angle of insonation was recorded in each arterial segment. The 3-mm sample volume was placed on the point of the highest velocity acceleration of a particular artery segment, as determined by the color-aliasing artifact. The depth of the sample volume placement was recorded. The Vmeans were calculated by automatic or, in cases of weak Doppler signal intensity, manual tracing of the maximum frequency envelope of the Doppler waveform. The investigators performed the tracing during the time of examination. The number of manual tracings was recorded in each case. Uncorrected velocities were calculated for each artery segment as a product of angle-corrected velocities and cosine of the recorder angle of insonation.
Magnetic Resonance Angiography
To exclude patients with intracranial arterial narrowing, we performed a time-of-flight (TOF) 3D gradient-echo sequence (provide TR/TE/flip angle and spatial resolution) covering the intracranial ICAs, vertebrobasilar arteries, and the circle of Willis in the axial plane. We transferred the raw data from 3D TOF MRA to an on-line workstation for the generation of segmented 2D arterial reprojections using a commercially available maximum intensity projection (MIP) ray-trace and multiplanar reconstruction algorithms. The segmented 2D reprojections and raw data of the ICAs and branches of the circle of Willis were displayed on a 1024 × 1024 pixel workstation and evaluated for potential segmental narrowing and flow restriction by the 2 trained readers.
Statistical Analysis
We entered all data into an Excel data base (Microsoft, Redmond, Wash) and then imported the data into a SYSTAT 10 (Systat Software, San Jose, Calif) for all statistical analyses. To compare proportions of successful identification of arteries for both sonographic methods, we used the exact sign test by Liddell. We used the repeated-measures ANOVA to test the null hypothesis that the TCD and TCDI velocities (corrected and uncorrected) did not differ between the 2 methods. However, in the case of significant differences, we used a paired t test with the optional P value adjustment (Dunn-Sidak) for multiple comparison tests. A linear regression analysis was used to quantify relationships between the TCD and TCDI parameters. Levels of probability <.05 were considered statistically significant.
Results
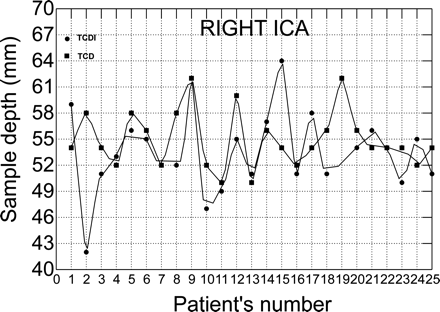
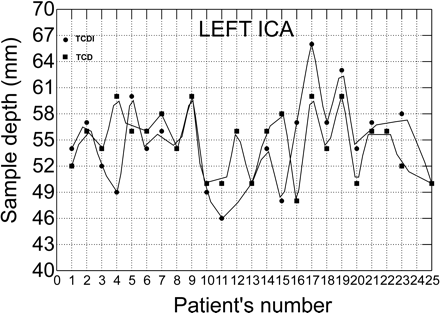
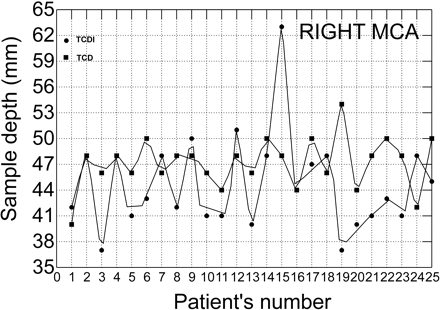
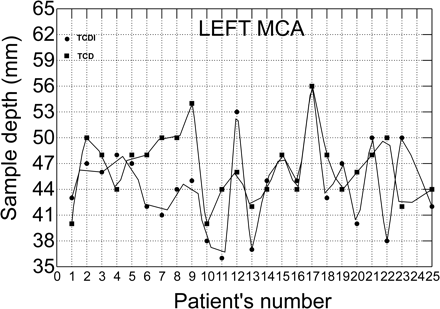
On MRA, no narrowing of the intracranial arteries was found in the children. The proportion of successfully insonated arteries was higher for the TCDI than for TCD. All major cerebral arteries in our patients were identified with the TCDI except for 2 ACAs, whereas 15 vessels (2 MCAs,1 ICA, 3 ACAs, and 9 PCAs) were not found with the TCD (P<.05). TCDI data from 1 MCA are not shown because they were inadvertently deleted in the TCDI machine. The average angle of insonation varied from 23° to 49° depending on the artery (Table 1). On the basis of paired data from 25 patients, the mean depth of insonation of the ICA and MCA on nonimaging TCD was 54.8 ± 3.4 mm and 47.1 ± 2.9 mm, respectively, on the right side and 54.6 ± 3.7 mm and 46.3 ± 4.2 mm, respectively, on the left side. On TCDI, the mean depth of insonation of the ICA and MCA was 54.3 ± 7.0 mm and 44.7 ± 5.5 mm, respectively, on the right side and 53.9 ± 5.9 mm and 44.6 ± 4.9 mm, respectively, on the left side. Although the differences were not significant in mean depth of insonation of these arteries, the differences in the individual arteries were as high as 15 mm (Figs 1–4).
Scatterplot with data points connected by lines shows differences in sample volume depths between TCDI and TCD in the right internal carotid arteries.
Scatterplot with data points connected by lines shows differences in sample volume depths between TCDI and TCD in the left internal carotid arteries.
Scatterplot with data points connected by lines shows differences in sample volume depths between TCDI and TCD in the right middle cerebral arteries.
Scatterplot with data points connected by lines shows differences in sample volume depths between TCDI and TCD in the left middle cerebral arteries.
Mean blood flow velocities in major cerebral arteries obtained with imaging transcranial Doppler sonography (TCDI) versus nonimaging transcranial Doppler sonographic (TCD) velocities in children with sickle cell disease
The angle-corrected TCDI velocity values did not differ significantly from the respective TCD velocity values (Table 1). The biggest discrepancy was observed in both ACAs. However, the TCDI velocity values were substantially lower than the respective TCD velocities (average 20%; range, 12%–32%), as shown in Table 1. In Table 2, the results of linear regression analysis to convert angle-corrected and uncorrected velocity values obtained with TCDI to TCD values are presented.
Values of coefficients and constants obtained by univariate linear regression analysis (y = ax + b) to predict nonimaging transcranial Doppler sonographic (TCD) velocities based on angle-corrected imaging transcranial Doppler sonographic (TCDI) velocities and TCDI velocities without angle correction in major cerebral arteries of children with sickle cell disease
The mean TCD flow velocity values in all studied arteries were below 170 cm/s, except in 3 patients in whom the velocities in the ICA, ACA, and MCA were 180, 181, and 229 cm/s, respectively (Table 3). In 16 arteries, the angle-corrected TCDI flow velocities ranged between 170 and 200 cm/s, and in an additional 5 arteries the angle-corrected TCDI velocities were above 200 cm/s (Table 3). In 2 patients, the TCDI velocities were above 170 cm/s, whereas in 1 patient the flow velocity in the MCA was higher than 200 cm/s.
Discrepancies in mean blood flow velocities in cm/s in major cerebral arteries obtained with angle corrected imaging transcranial Doppler sonography (TCDI) and nonimaging conventional transcranial sonography (TCD) in individual children with sickle cell disease*
In most of the patients, the TCDI velocity waveforms were manually traced (Table 4). There were no significant differences between the mean velocity values obtained by manual and automatic tracing except for the left ACA (unpaired t test, (Tables 3 and 4).
Numbers of automatic and manual measurements and average imaging transcranial Doppler sonography mean blood flow velocities obtained with automatic and manual tracings in major cerebral arteries of children with sickle cell disease*
Discussion
As has been shown in previous studies, our study confirmed that in children with SCD, the TCDI velocities were lower by approximately 20% compared with TCD velocities. Angle-corrected TCDI velocities and TCD velocities were not statistically different, though in many patients the discrepancies were high.
The STOP study showed that the risk of stroke can be decreased by more than 90% if transfusions are instituted on the basis of TCD criteria.1,3 Some centers have started using TCDI for screening SCD patients because machines are readily available in many radiology departments and the technique is easier to learn. These centers adopted the STOP criteria for TCDI velocities mostly because several recent publications showed agreement between TCD and TCDI measurements.5,7,8,11–13 However, our data showed not only discrepancies between the TCDI and TCD measurements but also overlapping of the TCDI “reference” values (eg, MCA 82–186 cm/s) with the lowest STOP criterion (170 cm/s). Thus, the STOP criteria may not work accurately with the use of TCDI velocities.
Our results were consistent with those of previous studies, which showed that TCDI velocities were lower than TCD velocities. The discrepancy ranged from 10% to 15%.6,14–18 The most discrepant readings were in the ACA, PCA, and ICA, which, compared with the MCA, had an unfavorable course in relationship to the ultrasound beam. The variable vessel trajectory can explain the variability in velocity measurements between the arteries because velocity measurements depend on the site of sampling of a particular vascular segment.5,9,19 Furthermore, in curved and tortuous arteries the distribution of blood flow across the artery is not uniform.20 Therefore, it is important to sample the entire lumen of the artery to obtain the most reliable results. In TCD, the sample volume placement is poorly controlled, which can lead to errors in identification and proper sampling of the arteries, and subsequently to inaccuracy in velocity measurements. Such situations obviously may lead to erroneous estimation of the risk of stroke.
Our data clearly showed that arteries could be identified and sampled more precisely with TCDI than with TCD. The rate of vessel identification with TCD in our study was similar to those reported in other studies.3,7 It is likely that extremely variable vessel course or size and location of the temporal acoustic window, or both, accounted for failures in identification. In our patients, differences in depth of sampling between TCDI and TCD varied from zero to as high as 15 mm. Such magnitude of discrepancy indicates that not only the sites of sampling of a particular vascular segment were different but also that the 2 readings could have come from completely different arteries. Although TCDI is not the standard criterion for imaging of the major arteries of the brain, the identification of arteries and placement of sample volume in a particular vascular segment leaves little room for errors.5
Also, other potential sources of discrepancy in velocity measurements between the TCDI and TCD have been suggested.6,7,15 For instance, a bigger TCDI transducer may result in less optimal positioning in the temporal window.7,14 However, we did not encounter any problem with positioning of the transducer. A way of outlining the Doppler waveform to obtain velocity data may contribute to discrepancy in velocity measurements between the 2 methods.21 If the signal-to-noise ratio is poor, automatic outlining of the waveforms is not precise, and an operator has to manually outline the waveform. Signal-to-noise ratio, and subsequently the number of manual tracings, can vary between the TCDI and TCD, depending on the machine. On the basis of TCD data, it has been demonstrated that automatic tracing provides 6% to 8% higher values compared with manual tracing.21 We do not know whether the difference pertains only to TCD measurements. On the basis of the TCDI data, we did not find any difference between the manual and electronic tracing except for the ACAs, in which we found that manual tracing provides higher velocity values. Nevertheless, variation in the method of waveform tracing may contribute to the discrepancy between TCD and TCDI.
Angle-corrected TCDI and TCD velocities were not statistically different, though in many patients we noticed substantial discrepancies. It is assumed in the TCD method that the angle between the course of a vessel and the ultrasound beam is 0°. However, in our patients, the angle varied on average from 30° for the MCA to 40° for the ACA. Our findings confirmed previous studies, which showed that almost half of MCAs require angles greater than 35° for angle-corrected measurements.17,18 Angle correction itself increases the variability of flow velocity measurements because determination of the angle is operator dependent, and variability in the course of a vessel is taken into account in the Doppler equation.5 Although we did not estimate an interobserver and intraobserver variability of measurement of the angle itself on the basis of the same arteries in the same children, our opinion is that the variability is small compared with the variability related to various trajectory of vessels. This opinion is supported by higher variability of angle-corrected velocities in the ACA and PCA than in the MCA. Although measurements of the angle can introduce some variability in velocity measurements, the angle-corrected TCDI velocities can be more accurate than the TCDI and TCD velocities. Angle correction can have a substantial impact on therapeutic decisions, because a child classified as having a low risk of stroke on the basis of TCDI measurements can be at high risk on the basis of angle-corrected TCDI measurements. For instance, if a measured velocity is 170 cm/s and the angle is 40°, the true velocity is 222 cm/s. Thus, it is clear that errors introduced by lack of angle correction would result in an underestimation of velocity readings. In our opinion, however, this and other studies published so far do not provide data to support a recommendation of using angle-corrected velocities. A trial is needed to demonstrate that a protocol on the basis of an angle-corrected TCDI is not worse in estimation of risk of stroke than the actual STOP protocol. Actually, design of such a trial, on the basis of TCDI or any other method, is a major problem in SCD research because, after implementations of STOP guidelines, the number of children with SCD who experience a stroke is relatively small.
One can question whether the “true velocity” is pertinent in the context of a large body of reported TCD and clinical outcome data (Medical College of Georgia and STOP), which was prospective and validated by long-term observation of children for outcome of stroke.6 The main drawback of such an approach is that by essentially relying on the STOP protocol to “define” the risk of stroke, it is difficult for the new TCDI protocol to seem more effective, even if it allows one to predict stroke better than the STOP protocol. Indeed, in many other applications such as diagnosis of intracranial vasospasm, stenosis, and occlusion, angle-corrected TCDI replaces the TCD.11,22 To be consistent with the STOP protocol, we offered regression equations in Table 2 for those who want to convert TCDI velocity values to values usable in the STOP criteria. However, it should be recognized that the regression equations can be specific for a TCDI machine because there may be a difference in velocity measurements between different machines and vendors.
Contrary to previous studies, our comparison of both methods was based on a homogeneous population of patients with no narrowing of the intracranial arteries, as determined with MR angiography.6,7,15 In cases of substantial narrowing, both TCDI and TCD can provide very similar measurements, whereas the major discrepancy may exist in borderline narrowing and in “normal” arteries. It can be argued that a TCDI study is more time consuming than a TCD study because an operator has to devote some time to determine the angle of insonation. However, this extra time is counterbalanced by time savings on easier and faster identification of arteries and selection of a proper sampling site. It should also be stressed that inappropriate angle correction, in particular when the angle is more than 60°, may provide erroneously high velocity values. To avoid such situations, an operator has to place a sample volume in a site such as an arterial segment, which allows not only keeping the angle as low as possible but also allows reliable determination of the angle of insonation.
The relatively small patient population in our study was mainly related to the well-known problem with recruitment of children with SCD. However, we believe that our sample size was sufficient to provide valid comparison of both methods. Also, the fact that both TCD and TCDI examinations were performed by the same investigator at the same time can be perceived as yet another limitation of our study. However, the identification and sampling of an artery with TCDI under supervision of the expert left very limited room for subjective measurements, whereas the comparison of both examinations performed at the same time reduced the risk of paradoxical discrepancy between TCD and TCDI related to hemodynamic and hematologic changes.
Conclusions
Similar to previous studies in the literature, our study confirmed that in children with SCD, the TCDI velocities were lower by about 20% compared with the TCD velocities. The angle-corrected TCDI velocities do not differ significantly from the respective TCD velocities, but those who want to use the angle-corrected velocity in clinical practice should be warned that in individual patients, the differences between angle-corrected TCDI and TCD velocities can be very high.
Our study also showed that TCDI could identify the major cerebral arteries more effectively than TCD because TCD could not depict the 15 major vessels, whereas on TCDI, all but 2 vessels could be identified. Furthermore, a risk of inaccurate velocity sampling with TCD is higher than with TCDI.
Acknowledgments
We thank Tom Flynn, Jerilyn Radcliffe, Jiongjiong Wang, Kwaku Ohene-Frempong, Abbas Jawad, Daniel Licht, and Robert Zimmerman for their valuable contribution to the study.
Footnotes
This work was supported in part by a grant from the National Institutes of Health, 5-R01-NS-046717, and a fellowship grant from the American Society of Pediatric Neuroradiology (Devang Gor).
References
- Received October 29, 2006.
- Accepted after revision February 6, 2007.
- Copyright © American Society of Neuroradiology