Abstract
BACKGROUND AND PURPOSE: Sex differences in white matter structure are controversial. In this MR imaging study, we aimed to investigate possible sex differences in language and motor-related tracts in healthy preterm neonates by using DTI and probabilistic tractography.
MATERIALS AND METHODS: Thirty-eight preterm neonates (19 boys and 19 girls, age-matched), healthy at term-equivalent age and at 12 months were included. TBV was measured individually. Probabilistic tractography provided tract volumes, relative tract volumes (volume normalized to TBV), FA, MD, and λ⊥ in the SLF, in the TRs, and in the CSTs. Data were compared by using independent t tests, and Bonferroni corrections were performed to adjust for multiple comparisons.
RESULTS: We showed that healthy preterm boys had larger TBV than girls. However, girls had statistically significantly larger relative tract volumes than boys bilaterally in the parieto-temporal SLF, and in the left CST. Moreover, in the left parieto-temporal SLF, a trend toward lower MD and λ⊥ was observed in females.
CONCLUSIONS: Structural sex differences were found in preterm neonates at term-equivalent age in both sides of the parieto-temporal SLF and in the left CST. Further studies are necessary to investigate whether these structural differences are related to later sex differences in language skills and handedness or to the effect of prematurity.
ABBREVIATIONS
- ATR
- anterior thalamic radiation
- CST
- corticospinal tract
- DTT
- diffusion tensor tractography
- FA
- fractional anisotropy
- GA
- gestational age
- MD
- mean diffusivity
- PTR
- posterior thalamic radiation
- SENSE
- sensitivity encoding
- SLF
- superior longitudinal fasciculus
- STR
- superior thalamic radiation
- TBV
- total brain volume
- TR
- thalamic radiation
- λ//
- longitudinal diffusivity
- λ⊥
- transverse diffusivity
Substantial interest in sex differences in neural structures has been generated in recent years by observations of sex differences in cognitive functions.1⇓–3 A male advantage for spatial abilities has been widely observed in humans and other animals,4 whereas a female advantage has been seen for verbal abilities such as verbal fluency and verbal memory in adult life.5⇓–7 This difference also has been found in children, with girls having better language development at an early age7⇓⇓–10 and boys experiencing more frequent language impairments.11,12
Therefore, postmortem pathologic and in vivo quantitative brain imaging studies have been looking for differences between males and females. In adults, several studies have shown that men have larger (by ∼10%) brains than women.13⇓–15 Interestingly, these differences are already present in children16⇓–18 and neonates.19 In adults, the regional volumetric gray matter distribution patterns tend to show an enlargement in females when adjusting for brain size.14,20⇓⇓⇓–24 In children, findings of sex differences in relative gray matter volume have shown enlargement in females, most prominently in the temporal and parietal cortices.25,26
Studies on sex effects on global and regional WM are controversial; both significant22,27,28 and nonsignificant interactions29,30 have been reported. It is possible that the measured WM volumes, as determined from conventional MR imaging, reflect changes in macrostructure only, and may not be sensitive to WM microstructure.31,32 Such microstructural changes are within the reach of DTI, an MR imaging technique that allows studying the in vivo microstructure and the volume of the major WM tracts. DTI assesses and quantifies water diffusion at a microstructural level, given that water diffuses more easily in the direction of the fibers than orthogonally.33⇓–35 Diffusion indices, such as FA, MD, and λ// and λ⊥, allow us to indirectly quantify brain microstructure.36,37 Results for sex differences in diffusion indices in adults, either global or regional, have been inconsistent. One study showed no sex difference,38 whereas others showed significant sex differences, but only when focusing on predefined brain regions, such as the frontal lobe or the corpus callosum.39⇓⇓–42 Nevertheless, it should be noted that all these studies by using either ROI analysis or voxel-based morphometric techniques have an error related to anatomic ambiguity in the ROI definition, WM segmentation, and other postprocessing steps such as spatial normalization and smoothing.22,43 These methods focus on predefined brain regions but not on specific WM tracts. DTT provides a 3D reconstruction of specific WM tracts and is able to overcome these confounding effects. Moreover, to our knowledge, no diffusion imaging studies have yet investigated whether sex differences are present in neonates.
In this study, we investigated, by using DTI and DTT, whether sex-related differences were present in the language and motor related fibers in healthy preterm neonates at term-equivalent time.
Materials and Methods
Subjects
Among preterm neonates born between June 2005 and June 2009 who underwent brain MR imaging to detect lesions related to premature birth,44 78 preterm neonates with acceptable (see below) DTI were studied. The inclusion criteria for normality were as follows: 1): normal head circumference at birth (>5th and <95th percentiles), 2) 5-minute Apgar score >6, 3) lack of evidence for congenital infection or multiple congenital anomaly syndrome, 4) normal structural brain MR imaging as assessed by 2 board-certified neuroradiologists (D.B., P.D.), and 5) normal physical and neurologic examination at term-equivalent age and at 12 months corrected for GA as assessed by a board-certificated neuropediatrician (A.A.). On the basis of these criteria, 28 neonates were excluded. Furthermore, 12 normal neonates were excluded to obtain sex groups of equal sample size, that were matched for GA at birth and corrected GA at the time of MR imaging. Thirty-eight healthy preterm neonates (19 boys and 19 girls) were finally included in this study (Table). The study was approved by the ethics committee of our institution (reference P2004/207 and P2009/234), and informed written parental consent was obtained for each participant.
Gestational age at birth and at the time of MRI scan
MR Imaging Data Acquisition
MR imaging data were acquired by using a 1.5T magnet (Achieva; Philips, Best, the Netherlands) equipped with an 8-channel SENSE head coil. The following sequences were acquired for all subjects: 1) sagittal 3D T1-weighted gradient-echo images, 2) coronal T2-weighted turbo-spin-echo images, 3) spin-echo echo-planar images (DTI): TR/TE = 5888/92 ms, FOV = 220 × 220 mm2, 32 noncollinear diffusion-sensitizing gradient directions with diffusion sensitivity of b = 600 s/mm2 and a 2 × 2 mm2 in-plane resolution, acceleration factor (SENSE) of 2.2, section thickness = 2.3 mm, and the scanning time for DTI acquisition of 3 minutes 40 seconds.
No sedation was used, and the neonates were spontaneously asleep, positioned in a vacuum immobilization pillow to minimize body and head movements. Ear-muffs were placed to minimize noise exposure. Oxygen saturation and electrocardiography were monitored throughout the acquisition.
Data Postprocessing
Data analysis was performed by using FSL software.45
Image Preparation
Image artifacts due to eddy current distortions and head movements were minimized by registering the DTI from 32 directions to the B0 images.46 DTI images corresponding to directions with motion artifacts was excluded from further data processing. DTI was considered as acceptable when <5 directions had to be excluded. Extraction of the brain parenchyma from scalp and skull was performed with the FSL Brain Extraction Tool; any small errors identified in the masks were manually corrected.14 Maps of the diffusion indices were obtained by using FSL Diffusion Toolbox.47
Probabilistic Tractography
The bundles were reconstructed in each subject by a single investigator (Y.L.) by using multitensor probabilistic tractography.31 Seed masks and waypoint masks were generated on color-coded FA maps, placed carefully by one radiologist (Y.L.) and checked by a second radiologist (D.B.).48 The SLF was separately tracked into 2 parts48,49: the frontoparietal SLF and the parieto-temporal SLF. For the frontoparietal SLF, a seed mask covered the frontal WM and the waypoint mask covered the frontoparietal WM; for the parieto-temporal SLF, the seed mask was the same as the waypoint mask of the frontoparietal SLF, and the waypoint mask covered the temporal lobe.48 The TRs were studied separately in 4 subradiations48: ATR, the motor and sensory STR, and the PTR. A seed mask was positioned in the bottom of the thalamus and a waypoint mask was positioned in the anterior limb of the internal capsule for the ATR; in the precentral gyrus for the motor STR; in the postcentral gyrus for the sensory STR, and in the occipital lobe for the PTRs. The CST was isolated as a whole, by using a seed mask positioned in the cerebral peduncle and a waypoint mask in the precentral gyrus.50
The original tracts were normalized by the total number of samples going from the seed mask to the target mask.51 Finally, the obtained connectivity distributions were thresholded with a probability of 2%.48,52,53
We calculated the TBV by measuring the volume of the voxels located in the brain mask.14,54 To assess tract macrostructure, tract volumes and relative tract volumes (defined as the ratio between individual tract volume and TBV) were computed. The microstructure of the tracts was evaluated with diffusion indices (FA, MD, λ//, and λ⊥) by using FSL maths.48
Statistical Analyses
All variables were analyzed with the SPSS software (SPSS, Chicago, Illinois). A 1-sample Kolmogorov-Smirnov test was performed to detect a possible departure from normality of our variables. Sex-related differences in the TBV, the volumes, the relative volumes, and the diffusion indices (FA, MD, λ//, and λ⊥) of each tract were analyzed by using a t test for independent samples. Adjustment for multiple comparisons was performed by using the Bonferroni correction,55 statistical significance was reached when P < .004. A trend toward significance was reported when P < .05.
Results
Brain Volume
The TBV of the 38 participants ranged from 367 to 614 cm3 (mean ± SD, 438 ± 52 cm3). TBV values in males (461 ± 59 cm3) were 10.7% larger than those in females (414 ± 30 cm3; P = .004).
Sex Differences in Principal WM Tracts
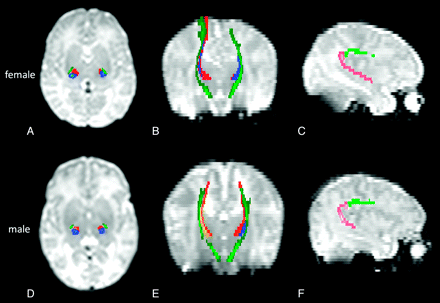
WM tracts related to sensorimotor and language functions are shown in Fig 1.
Sensorimotor and language-related tracts are shown on B0 images in a healthy preterm girl born at 31 weeks GA and imaged at 37 weeks GA (A–C) and in a boy born at 30 weeks GA and imaged at 37 weeks GA (D–F). Axial (A, D) and coronal (B, E) images show the CST (dark green) and the STR (motor STR in yellow-red and sensory STR in blue). Sagittal images (C, F) show the frontoparietal SLF (light green) and parieto-temporal SLF (pink).
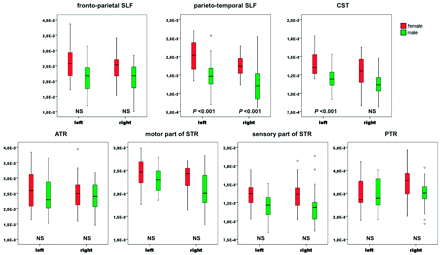
Relative tract volumes were statistically significantly larger in females than in males (Fig 2) bilaterally in the parieto-temporal SLF (left, P < .001; right, P < .001) and in the left CST (P < .001). Moreover, trend toward larger tract volumes (On-line Table 1) was found bilaterally in the parieto-temporal SLF (left, P = .034; right, P = .011).
Sex effects on relative tract volumes of the SLF, the CST, and the TR in both hemispheres are shown by boxplots, with median (line), first and third quartiles (extremities of the box), sample minimum and maximum (vertical bars) and outside of the box (outliers and extreme cases) values. Differences between sex are significant after Bonferroni correction when P < .004.
A trend toward lower MD (P = .041) and λ⊥ (P = .033) in females was observed in the left parieto-temporal SLF (On-line Table).
Discussion
In this in vivo brain MR imaging study, we investigated sex differences in the TBV and WM tracts with DTI probabilistic tractography in the language and motor networks in a population of healthy preterm neonates scanned at term-equivalent age. We found, like other studies in neonates19 and in adults,14,23 that at term-equivalent age healthy preterm male neonates had larger TBV than females. The original findings of our study were that female neonates had larger relative tract volumes bilaterally in the parieto-temporal SLF and in the left CST, with a trend toward lower MD and λ⊥ in the left parieto-temporal SLF after Bonferroni correction.
Previous studies have shown that the temporal cortex is larger in females than in males. This has been demonstrated in children by using structural imaging,25,26 and also in adults through pathologic studies showing larger planum temporale56 and Heschl gyrus,57 and a greater attenuation of neurons58 in females. Given that the parieto-temporal SLF is supposed to transmit auditory information from the superior temporal gyrus to the inferior parietal lobe, we suggest that our results may reflect an early established difference in favor of female neonates in the number or size of axons in these language-related regions.
The SLF is one of the slowest maturing WM tracts, being not yet myelinated at birth.59,60 Lower MD and λ⊥ are probably caused by a decrease in brain water content and an increase in membrane attenuation, and they suggest an advanced premyelination stage characterized by proliferation and maturation of oligodendrocytes.61,62 Therefore, we propose that this microstructural sex difference might be caused by an advanced maturation in the left parieto-temporal SLF in female neonates.
The finding of different tract relative volumes with no significant difference in diffusion indices is a feature with no straightforward interpretation. In the right parieto-temporal SLF, a larger relative tract volume associated with a trend toward larger tract volume in females was found in the absence of difference in diffusion indices: this might possibly reflect macrostructural changes (more axons at the same myelination stage). In the CST, a larger relative tract volume in females was observed together with no significant difference in either tract volume or diffusion indices, suggesting a similar maturation and number of axons in a smaller female brain. In other published series, differences in tract volumes were not always associated with differences in diffusion indices.33,48,63,64 Moreover, we used probabilistic tractography, which does not directly rely on diffusion index values, but on the uncertainty orientation of the distribution function, enabling it to progress across regions with principal direction uncertainty and through regions with crossing fibers. Therefore, in probabilistic tractography, volume measurement is not directly linked to diffusion indices.51
Language acquisition and processing have shown sex-related differences in infants as young as 2 years old.8,10,65 Maccoby and Jacklin66 reported that girls outperformed boys during preschool and early years in articulation, length of sentences, verbal fluency, grammar, and spelling. In giving the California Verbal Learning Test to children between 5 and 16 years old, girls were found to use more semantic clustering, to recall and recognize more items, and to relate words together more as a recall aid than did boys.67 In addition, language impairments have been found to occur more frequently in boys than in girls.11,12 Because of its implication in language function, we suggest that the sex effect on parieto-temporal SLF relative tract volume and microstructure might explain the more rapid development of language skills hitherto reported in females.
Although we could not evidence a significant sex effect on the volume of the CST, we showed that the relative volume of the left CST is larger in females. Interestingly, studies in adults have already shown, after adjusting for the TBV, an increased volume21 and gray matter concentration14 in the precentral gyri in females. A relatively larger left CST in females might explain why meta-analyses showed more right-handed females than males in the general population.68 However, the relationship between handedness and asymmetry in the adult brain is not established, because some studies have found such relationship69,70 but others have not.71
Sex differences in the volume and microstructure of certain WM tracts, as observed in this study, might result from genetic factors as well as from effects of sex steroids on brain development, both factors being known to affect regional tissue composition.72⇓–74
Another hypothesis is that our results may have been influenced by the effect of prematurity. Indeed, even if the normality of our preterm population was based on robust structural and clinical criteria, as in previous studies,48,52 we cannot exclude the possibility that the sex related differences observed in the language and motor networks may have been caused by subtle cerebral lesions, because certain studies seem to suggest that preterm males may be more sensitive to brain injuries than females.75,76 Therefore, it would be of interest to investigate whether these sex differences are also present in healthy term neonates.
Because the first years of life are perhaps the most dynamic phase of postnatal brain development, with rapid development of a wide range of cognitive and motor functions,77 the link between structural sex differences at term-equivalent age with later functional differences should be interpreted with great caution. Longitudinal studies combining cognitive evaluation with structural and functional imaging may provide insights into the structure-function relationship in sex differences.
Another limitation of our study is that the reproducibility of mask placement was not assessed. Nevertheless, mask placements were checked by 2 radiologists and in probabilistic tractography, by using the approach of normalization, the size of seed and target masks can be ignored.51
Conclusions
In this DTI and probabilistic tractography study on healthy preterm neonates, we demonstrated that sex differences are present in language and motor-related tracts at term-equivalent age. Further studies are needed to investigate whether these structural differences are related to later sex differences in language skills and handedness or to the effect of prematurity.
Acknowledgments
We are grateful to Doni Tamblyn for assistance in language editing.
Footnotes
-
Disclosures: Viviane De Maertelaer. Research Support (including provision of equipment or materials): University of Brussels; Vincent Denolin. Consultant: Advice on sequence parameters and data analysis.
-
This work was supported by grants from the Fonds Xénophilia (ULB) and the Fond de la Recherche Scientifique of Belgium (grant 1.5.149.10).
Indicates open access to non-subscribers at www.ajnr.org
References
- 1.↵
- 2.↵
- 3.↵
- 4.↵
- 5.↵
- 6.↵
- 7.↵
- 8.↵
- 9.↵
- 10.↵
- 11.↵
- 12.↵
- 13.↵
- 14.↵
- 15.↵
- 16.↵
- 17.↵
- 18.↵
- 19.↵
- 20.↵
- 21.↵
- 22.↵
- 23.↵
- 24.↵
- 25.↵
- 26.↵
- 27.↵
- 28.↵
- 29.↵
- 30.↵
- 31.↵
- 32.↵
- 33.↵
- 34.↵
- 35.↵
- 36.↵
- 37.↵
- 38.↵
- 39.↵
- 40.↵
- 41.↵
- 42.↵
- 43.↵
- 44.↵
- 45.↵
- 46.↵
- 47.↵
- 48.↵
- 49.↵
- 50.↵
- 51.↵
- 52.↵
- 53.↵
- 54.↵
- 55.↵
- 56.↵
- 57.↵
- 58.↵
- 59.↵
- 60.↵
- 61.↵
- 62.↵
- 63.↵
- 64.↵
- 65.↵
- 66.↵
- 67.↵
- 68.↵
- 69.↵
- 70.↵
- 71.↵
- 72.↵
- 73.↵
- 74.↵
- 75.↵
- 76.↵
- 77.↵
- Received February 9, 2011.
- Accepted after revision March 28, 2011.
- © 2011 by American Journal of Neuroradiology