Abstract
SUMMARY: The PulseRider is a novel endovascular device specifically designed to treat bifurcation intracranial aneurysms with wide necks. In an international series, we report the results of PulseRider stent-assisted coiling of 15 patients (9 women and 6 men; mean age, 62.6 years) with 15 unruptured wide-neck (median dome size, 8 mm; median neck size, 5 mm) bifurcation aneurysms. Failure of PulseRider treatment occurred in 1 case, and 1 intraprocedural thromboembolic complication was observed. There was no mortality or neurologic permanent morbidity at discharge and at 1 month. Immediate angiographic outcome showed 12 complete occlusions and 2 neck remnants. Follow-up at 6 months was available for 3 aneurysms and demonstrated 2 complete aneurysm occlusions and 1 growing neck remnant. In this small series of selected patients, PulseRider stent-assisted coiling of wide-neck bifurcation aneurysms was feasible with low procedural complication rates. Angiographic follow-up will be required to evaluate the efficacy of the PulseRider device.
ABBREVIATION:
- IA
- intracranial aneurysm
Endovascular treatment with coils is the reference therapy for ruptured intracranial aneurysms (IAs).1⇓–3 Although no randomized study demonstrated the superiority of endovascular treatment compared with clipping for unruptured IAs, endovascular treatment is also often the preferred therapeutic option.3 However, endovascular treatment with coils of IAs with wide necks is difficult or simply not feasible. In this specific situation, balloon-assisted and stent-assisted techniques have widened the indications for endovascular treatment.4⇓⇓–7 Endovascular treatment of bifurcation IAs often requires stent placement with double stents in “Y” or “X” configurations, which could increase the risk of clinical complications,7,8 whereas some authors reported low rates of complications compared with the balloon-remodeling technique.9,10
Recently, 3 devices have been specifically developed for the endovascular treatment of such aneurysms arising at bifurcations: the WEB (Sequent Medical, Aliso Viejo, California), the pCONus (phenox, Bochum, Germany), and the PulseRider device (Pulsar Vascular, San Jose, California). The WEB is an intrasaccular braided-wire flow disruptor,11,12 and the pCONus is a new stent-like self-expanding nitinol implant with 4 distal petals allowing coiling of the aneurysmal sac.13 The PulseRider has a unique frame configuration that opens to conform to the vessel walls. It is specifically designed to preserve luminal patency and hemodynamic flow through the parent vessel bifurcation, while minimizing exposed metal to encourage early endothelialization while securely retaining coils within the aneurysm sac. It received a CE mark for intracranial aneurysms but has not been approved by the FDA. To date, a single published article on aneurysms treated with the PulseRider reported a series including 3 IAs.14 The aim of this study was to evaluate the results of the treatment of wide-neck bifurcation IAs with the PulseRider in an international series.
Case Series
The PulseRider is a self-expanding nitinol implant (Fig 1) that is delivered via a standard microcatheter with an inner diameter of 0.021 inches. The device is retrievable and may be repositioned by retracting it into the microcatheter at any time during or after deployment. It is deployed at the parent vessel bifurcation and across the aneurysm neck to provide a supporting framework, bridging the aneurysm neck while retaining coils within the aneurysm. The PulseRider is electrolytically detached from the delivery wire. The T or Y configurations are available according to the geometry of the daughter vessels arising at the bifurcation with 8- or 10-mm diameters.
The PulseRider device is intended for use with embolic coils for the treatment of unruptured wide-neck intracranial aneurysms originating on or near a vessel bifurcation. The PulseRider has a unique arch design with concentrated coverage at the neck allowing attenuated coil packing, and open architecture in the branch vessels eliminates struts crossing through the lumen of the branch vessels. Reproduced with permission from PulsarVascular.
From June 2014 to February 2015, 15 consecutive patients (9 women and 6 men; mean age, 62.6 years) with 15 unruptured bifurcation IAs (median dome size, 8 mm; median neck size, 5 mm) at 1 US center (Charleston, South Carolina) and 5 European institutions (Lyon and Besançon, France; Florence, Italy; Recklinghausen, Germany; Salzburg, Austria) who were treated with the PulseRider device for IAs were retrospectively analyzed (under institutional review board approved protocol in United States and without approved ethics committee protocols in the European Union). The decision to assist coiling by a PulseRider device was made at the discretion of the senior author. All patients were treated under general anesthesia and full anticoagulation. In addition, double antiplatelet therapy was administered preoperatively according to the operator's protocol.
Endovascular Procedure
A Prowler Select Plus 0.021-inch microcatheter (Codman & Shurtleff, Raynham, Massachusetts) was navigated over a 0.014-inch microwire and positioned at the neck of the aneurysm. In a suitable working projection, the appropriately sized PulseRider was then deployed across the neck of the aneurysm with limbs in the daughter vessels arising at the bifurcation or in the aneurysm, or in a hybrid fashion with one limb in the branch vessel and the other limb in the aneurysm. Thereafter, a second microcatheter was inserted through the shaft into the aneurysm fundus, and coiling was performed. The PulseRider was detached either at or near the final coiling.
Treatment Failure
Treatment failure with the PulseRider occurred in 1 patient (patient 11) with a wide-neck carotid terminus aneurysm and a 4.5-mm dome. The deployment of the PulseRider was achieved, but on control angiograms, suboptimal positioning of the device was seen with incomplete protection of the neck. The PulseRider was replaced several times without success, and the aneurysm was then treated with stent-assisted coiling (Y-stent placement).
Procedural Complications
One thromboembolic event after detachment of the device occurred. After successful PulseRider-assisted coiling of a right MCA aneurysm, a thrombus formation occurred at the limbs of the device and caused a stenosis of the distal M1 segment. After immediate administration of glycoprotein IIb/IIIa inhibitors, the stenosis remained but the patient woke up without neurologic deficits and remained in this status at 1 month follow-up. No intraoperative rupture was observed.
Outcome
Among the 14 patients treated with the PulseRider, no neurologic impairment was observed at discharge and at 1-month follow-up (11 patients). There were no delayed neurologic deficits or deaths at follow-up.
One reader (B.G.) independently evaluated all the angiograms by using a simplified 3-point scale (total occlusion, neck remnant, aneurysm remnant).15 Immediate angiograms showed complete occlusion in 12 patients and neck remnant in 2 patients. Angiographic follow-up at 6 months was available for 3 patients and demonstrated 2 complete aneurysm occlusions and 1 growing neck remnant. During follow-up, retreatment was performed in 1 case due to a significant increase in the size of the neck remnant, with complete occlusion at the end of procedure. No in-stent stenosis and 1 jailed branch occlusion were observed.
Illustrative Cases
Case 1.
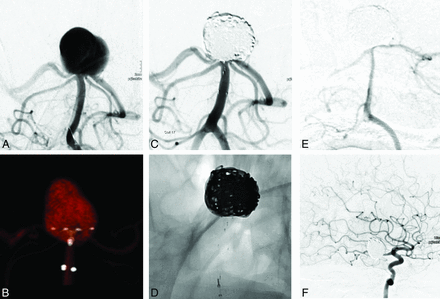
A 60-year-old woman (Fig 2, patient 9) presented with an incidental large basilar artery aneurysm. Angiography revealed a 17-mm aneurysm with an 8-mm neck. Endovascular treatment was performed by using a PulseRider device with final complete aneurysm occlusion. Angiographic follow-up at 6 months revealed complete aneurysm occlusion with an occlusion of the P1 segment of the posterior cerebral artery, which was supplied by the internal carotid artery via the posterior communicating artery. The patient was asymptomatic.
Incidental large basilar artery aneurysm in a 60-year-old woman (case 1). A, The angiogram shows a 17-mm aneurysm with an 8-mm neck. B, 3D reconstruction after rotational angiography shows the PulseRider device placement before coiling. C and D, An angiogram at the end of the procedure shows final complete aneurysm occlusion. E, Angiographic follow-up at 6 months reveals complete aneurysm occlusion with an occlusion of the P1 segment of the posterior cerebral artery, which was supplied by the internal carotid artery via the posterior communicating artery (F). The patient was asymptomatic.
Case 2.
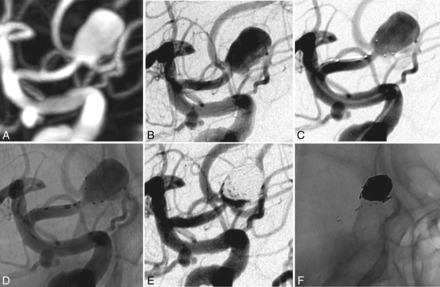
A 50-year-old woman (Fig 3, patient 6) presented with an incidental finding of a 4.2-mm wide-neck anterior communicating artery aneurysm. Endovascular PulseRider deployment and then coiling was performed. The final angiogram showed a neck remnant.
A wide-neck anterior communicating artery aneurysm in a 50-year-old woman (case 2). A, 3D reconstruction after rotational angiography shows a large anterior communicating artery with a 4.2-mm neck. B, Subtracted angiography shows a large anterior communicating artery. C and D, PulseRider stent-assisted coiling was performed. E and F, Subtracted angiographies at the end of the procedure show a neck remnant. The PulseRider device retained coils within the aneurysm sac.
Case 3.
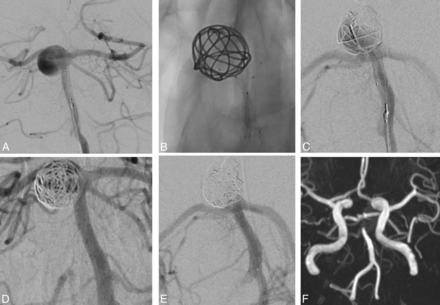
A 60-year-old woman (Fig 4, patient 14) presented with an unruptured basilar tip aneurysm. Angiography showed a large basilar tip aneurysm with a 4-mm-wide neck. Endovascular treatment was performed by using a PulseRider. The final angiogram showed complete aneurysm occlusion, and MRA at 1 day showed complete aneurysm occlusion.
An unruptured basilar tip aneurysm in a 60-year-old man (case 3). A, Subtracted angiography shows a large basilar tip aneurysm with a 4-mm-wide neck. B–D, Endovascular treatment was performed by using a PulseRider device. E, Subtracted angiography at the end of the procedure shows complete aneurysm occlusion. F, MRA at 1 day shows complete occlusion.
Discussion
This initial study reports a series of patients with unruptured bifurcation IAs with wide necks treated by PulseRider stent-assisted coiling.
Feasibility and Patient Selection
This report suggests that PulseRider-assisted coiling of IAs with wide necks is feasible; however, the selection of patients should be well-considered before deciding on a PulseRider treatment. In fact, in this small series of 15 patients, the rate of failure was 6.7% (patient 11). This patient presented with a wide-neck terminus carotid aneurysm with a small-size dome. The PulseRider deployment was feasible, but it did not provide protection of the aneurysmal neck even after several attempted placements. The pCONus device was placed and not detached due to the same problem occurring repeatedly; the aneurysm was then successfully treated with Y-stent-assisted coiling without complications.
Clinical and angiographic outcomes
Selection of patients is also important when using a WEB device. In the recent series of Gherasim et al11 dealing with 10 patients with unruptured anterior communicating artery aneurysms (mean dome size, 5.8 mm; range, 3.8–8.2 mm; mean neck size, 5.4 mm; range, 3.6–8 mm), WEB deployment failed in 3 of 10 patients because of unfavorable anatomy and the use of a much larger and stiffer microcatheter for the WEB device than usually used for coiling.11
Periprocedural Complications
Our results show that endovascular treatment of IAs with the PulseRider is safe despite the very specific population with a median neck size of 5 mm. There was neither device-related mortality nor permanent morbidity. Similar results have also been reported in the first published series.14 The safety of the PulseRider was also highlighted because no clinically evident complications were associated with its use in 3 wide-neck aneurysms.14 Contrary to sidewall IAs, bifurcation IAs with wide necks are difficult or impossible to treat with simple coiling and often need double stent placement in “Y” and “X” configurations. However, the risk of procedure-related morbidity and mortality is not negligible. The rate of procedure-related permanent neurologic deficits was 10% in 97 patients with complex and wide-neck bifurcation aneurysms.8 Compared with regular intracranial stent placement, PulseRider treatment also needed dual antiplatelet therapy during the perioperative period despite a very low amount of metal. The safety of this device seems very good in our small series. However, the safety of PulseRider stent-assisted coiling remains to be assessed in larger series.
Anatomic Results
As previously reported in the small series of Spiotta et al,14 complete initial aneurysm occlusion was achieved in most cases (84.6%). These immediate anatomic results are encouraging, given the unfavorable angiographic aspects of IAs included in our series. In this series of Spiotta et al with 3 wide-neck bifurcations aneurysms, complete aneurysm occlusion was observed in all cases.14 In fact, large or giant IAs treated with coils presented low initial angiographic occlusion rates and high rates of recanalization.3,15,16 However, in our series, angiographic control at follow-up was obtained in only 3/13 patients (23%). A follow-up is mandatory to evaluate the efficacy of this treatment. Although angiography remains the criterion standard, there is a role for MRA in following up these patients because small artifacts were introduced by metal as illustrated in Fig 4 (patient 14).17 Much more data with the PulseRider device are clearly required to evaluate the mid- and long-term results of this new endovascular approach.
The limitations of our study were a small number of patients with a relatively short follow-up period to evaluate the efficacy of the PulseRider device. In addition, a small percentage of patients were followed in our series. However, we believe it is important to have a preliminary evaluation for this new endovascular treatment device dedicated to challenging IAs with wide necks and/or complex anatomy. So far, the preliminary results are encouraging.
Footnotes
Disclosures: Alejandro M. Spiotta—UNRELATED: Consultancy: Penumbra, Pulsar, MicroVention, Stryker; Grants/Grants Pending: MicroVention (research grant)*; Travel/Accommodations/Meeting Expenses Unrelated to Activities Listed: Penumbra, Pulsar, MicroVention, Stryker. *Money paid to the institution.
References
- Received April 10, 2015.
- Accepted after revision June 8, 2015.
- © 2016 by American Journal of Neuroradiology