Abstract
BACKGROUND AND PURPOSE: Third and fourth branchial apparatus anomalies are rare congenital anomalies. The purpose of this study was to investigate imaging features of these lesions on fetal MR imaging in comparison with lymphatic malformations, the major competing differential diagnosis in these cases.
MATERIALS AND METHODS: A retrospective review of our institutional fetal MR imaging database between 1997 and 2019 resulted in 4 patients with confirmed third and fourth branchial apparatus anomalies and 14 patients with confirmed lymphatic malformations. The imaging features were reviewed by consensus, and the Fisher exact test was used to evaluate statistically significant differences between these 2 populations.
RESULTS: Four cases of third and fourth branchial apparatus anomalies were imaged at 29 weeks 1 day (range, 23 weeks 1 day to 33 weeks 4 days). All 4 cases demonstrated unilateral, unilocular cysts without reduced diffusion or hemorrhage and a medially directed beaked contour that tapered between the spine and airway at the level of the piriform sinus. Compared with 14 cases of fetal lymphatic malformations imaged at 27 weeks 6 days (range, 21 weeks 3 days to 34 weeks 6 days), third and fourth branchial apparatus cysts were significantly more likely to be unilocular (P < .005) and to have a medially beaked contour (P < .005). The combination of features of unilateral, unilocular, and medially beaked contour was observed only in the fetuses with third and fourth branchial apparatus cysts (P < .001).
CONCLUSIONS: The presence of a left-sided unilocular cyst with a medially beaked contour tapering at the level of the piriform sinus suggests the diagnosis of third and fourth branchial apparatus anomaly. Accurate diagnosis in the prenatal period allows proper counseling, genetic work-up, and treatment, potentially sparing patients from recurrent infections and associated morbidity.
ABBREVIATION:
- SS
- single-shot
Anomalies of the branchial apparatus are among the most common congenital abnormalities of the head and neck, second only to thyroglossal duct cysts, and are thought to represent failure of obliteration of the branchial clefts or cervical sinus of His.1 Third and fourth branchial apparatus anomalies are rare, comprising only 1%–10% of all branchial apparatus anomalies.2⇓-4 These anomalies arise from the piriform sinus and course through the deep spaces of the neck, often descending into the mediastinum along the tracheoesophageal groove. Although third and fourth branchial apparatus anomalies are relatively less common, they are clinically important to recognize because they can present acutely with abscess, suppurative thyroiditis,5,6 and even life-threatening airway compromise and need to be appropriately treated.7,8 These anomalies typically manifest postnatally as fistulas or sinuses but can sometimes manifest in utero. There are few published reports on the prenatal imaging appearance of these lesions.
We present 4 cases of third or fourth branchial apparatus anomalies diagnosed prenatally with MR imaging and describe the prenatal MR imaging features suggestive of this diagnosis. We compare these imaging features with those seen in lymphatic malformations, the most common cystic lesion in the posterior cervical space, to investigate distinguishing features of these entities. Last, we correlate our prenatal findings with postnatal imaging, surgery, pathology, and clinical follow-up.
MATERIALS AND METHODS
In this institutional review board–approved, Health Insurance Portability and Accountability Act–compliant study, we searched the University of California San Francisco fetal MR imaging database for all reports of fetal neck MR imaging with cystic neck masses performed between 1997 and 2019. After correlation with available postnatal surgical reports and/or pathology, we identified 4 patients with surgically and/or pathologically confirmed third and fourth branchial apparatus anomalies and 14 patients with confirmed lymphatic malformations. Of the 4 patients with third and fourth branchial apparatus anomalies, 1 patient was scanned with 1.5T MR imaging (GE Healthcare Signa, Waukesha, WI) and the 3 others were scanned with 3T MR imaging (GE Healthcare Discovery 750, Waukesha, WI). One of the 4 patients underwent 2 fetal MRIs, both at 3T. Of the 14 cases of lymphatic malformation, 7/14 (50%) cases were imaged at 1.5T (GE Healthcare Signa, Waukesha, WI) and 7/14 (50%) were imaged at 3T (GE Healthcare Discovery 750, Waukesha, WI). The fetal neck MR imaging protocol at our institution includes a sagittal maternal large FOV single-shot (SS) FSE for localization, and then 2 or 3 acquisitions in each of the 3 planes of T2-weighted real-time SS FSE centered over the fetal neck (TR = 4000 ms, TE = 100 ms, 3-mm section thickness/0-mm section spacing, FOV = 24.0, matrix = 224 × 256). Additional sequences include axial and coronal diffusion-weighted imaging (b = 800, TR = 4000 ms, TE = minimum, 3-mm section thickness/0-mm section spacing, FOV = 32.0, matrix = 64 × 128) and axial echo-planar imaging (TR = 4000 ms, TE = 90 ms, 3-mm section thickness/0-mm section spacing, FOV = 25.0, matrix = 100 × 100).
Two radiologists with expertise in pediatric neuroradiology (Y.L., O.A.G.) and 1 radiologist with expertise in obstetric sonography (V.A.F.) reviewed the imaging by consensus and rated the location, size, locularity, laterality, vascular displacement, the presence of a medial beak, airway displacement, thyroid and mediastinal involvement, as well as T1, T2, DWI, and EPI characteristics of the masses. The Fisher exact test was used to evaluate statistically significant differences in these characteristics between third and fourth branchial apparatus anomalies and lymphatic malformations. Postnatal surgical and pathology reports were also reviewed, and clinical follow-up was obtained when available.
RESULTS
Fetal MR Imaging of Third and Fourth Branchial Apparatus Anomalies
We identified 4 cases of prenatally diagnosed third and fourth branchial apparatus anomalies. In all 4 cases, the patients were referred for fetal MR imaging for evaluation of a unilateral, unilocular, cystic neck mass detected on routine prenatal sonography (Figs 1–4) at a mean gestational age of 20 weeks 3 days (range, 18 weeks 6 days to 21 weeks 0 days). Fetal MRIs were performed following the ultrasounds at a mean gestational age of 29 weeks 1 day (range, 23 weeks 1 day to 33 weeks 4 days). In the case in which a second fetal MR imaging was performed to assist with delivery planning, the second MR imaging occurred 9 weeks after the first, at 37 weeks gestational age.
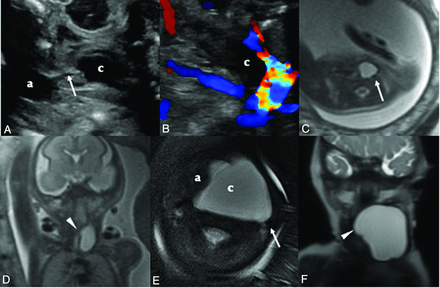
Patient 1 was initially identified as having a cystic neck mass on prenatal sonography. A, Sonography at 23 weeks demonstrates a left-sided cystic neck mass and communication (arrow) between the airway (a) and cyst (c). Doppler imaging (B) confirms the absence of intralesional vascular flow. Cine sonography imaging (Online Supplemental Video) shows fluctuation in the size of the cystic lesion with fetal breathing motion. Fetal MR imaging at 23 weeks’ gestation with axial T2 SS FSE demonstrates (C) a left-sided unilocular cystic lesion that approaches but does not displace the airway on the axial image. The carotid sheath (arrow) is posterior to the cyst (arrow). Coronal image (D) from the same scan highlights the tubular shape of the lesion with a medial beak (arrowhead). Postnatal axial T2 image (E) shows interval growth of the lesion, with better visualization of the posteriorly displaced carotid sheath (arrow) relative to the cyst (c). The airway is displaced to the right (a). Coronal T2 image (F) from the same postnatal scan demonstrates the medial beak (arrowhead) and tubular shape of the lesion.
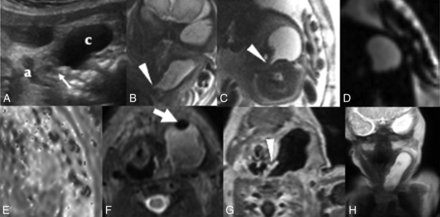
Prenatal sonography in patient 2 (A) demonstrates a left-sided cystic lesion in the neck. The lesion (c) lies lateral to the airway (a) and courses posterior to the airway as it extends medially (white arrow). Coronal (B) and axial (C) T2 SS FSE fetal MR images at 31 weeks’ gestation show a tubular cystic neck lesion with a beak (arrowhead) that extends medially and posterior to the airway. Axial EPI diffusion-weighted imaging (D) and an ADC map (E) demonstrate no abnormal susceptibility or reduced diffusion. Postnatal T2 imaging (F) in the same patient shows a nondependent hypointensity (white arrow) within the lesion after birth, compatible with air. Postcontrast T1 image (G) shows a communication with the airway (white arrowhead), confirmed intraoperatively following the postnatal scan. Coronal T2 image (H) demonstrates the tubular shape of the sinus.
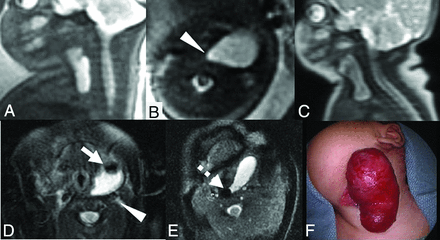
Fetal MR imaging for patient 3 at 20 weeks demonstrates a unilocular left-sided cystic neck lesion extending into the mediastinum in the sagittal (A) and axial (B) planes. The lesion tapers toward the midline with a medial beak (arrowhead) and extends into the mediastinum. Postnatal imaging demonstrates a similar craniocaudal extent of the cystic lesion (C) compared with the prenatal image (A). T2 fat-saturated postnatal imaging (D) demonstrates a focus of air (white arrow) within the lesion, with posterior displacement of the carotid artery (white arrowhead). Positioning the patient in the left lateral decubitus position (E) shows movement of the air bubble (dashed arrow) with repositioning. Postoperative photograph (F) of the cystic lesion in the neck before excision. Operative findings confirmed a sinus tract communicating with the piriform sinus.
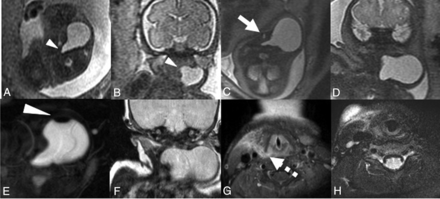
Fetal MR imaging of patient 4 in the axial (A) and coronal (B) planes at 28 weeks demonstrates a left-sided unilocular cystic lesion, with medial beaking (arrowhead), extending medially posterior to the airway, without mass effect on the airway. Axial (C) and coronal (D) T2 SS FSE fetal MR imaging at 37 weeks demonstrates interval growth of the cystic lesion, now displacing the airway (white arrow) to the right. Postnatal axial T2 fat-saturated image (E) demonstrates a nondependent hypointensity (white arrowhead), compatible with air within the cyst, which appears to have continued to enlarge on the coronal image (F) compared with the fetal MR imaging at 37 weeks’ gestation (D). A contralateral right-sided fistula in the same patient presented at 5 years of age, superiorly involving the piriform sinus (dashed arrow) on the axial T1 fat-saturated postcontrast image (G) and extending to the skin on the T2 fat-saturated image (H) more inferiorly. Bilateral third and fourth branchial anomalies are rare, but occur in 2%–3% of cases and are often familial.26 The right-sided fistula was also hypothesized to represent a pseudofistula acquired through multiple repeat infections of the branchial apparatus anomaly.
In all 4 cases, on sonography, the cysts were unilocular and anechoic without internal debris. Features of the cysts by fetal MR imaging are listed in the Table. On fetal MR imaging, the cysts were homogeneously T2-hyperintense and T1-hypointense. In the 2 cases in which DWI was performed, the lesions demonstrated increased diffusivity. No hemorrhage was seen on EPI in any of the 4 cases. All cysts were unilateral, left-sided, and involved the deep spaces of the neck and displaced the ipsilateral carotid space in a posterior-medial direction and the sternocleidomastoid muscle laterally. All 4 cases demonstrated a medially directed beaked contour that tapered between the spine and the airway at the level of the piriform sinus. There were varying degrees of mass effect on the airway in 3 cases, and the lesions crossed the midline posterior to the airway in 2 cases. In all 4 cases, the lesion extended inferiorly to contact the thyroid gland, and in 3 cases, the lesion extended into the mediastinum.
Imaging characteristics of postnatally confirmed fetal third or fourth branchial apparatus anomalies compared with consecutive cases of fetal lymphatic malformation
In the case with a follow-up fetal MR imaging 9 weeks later, the cyst demonstrated interval growth, but there was no change in its morphology or signal characteristics.
In one case on prenatal sonography, the lesion was noted to fluctuate in size with swallowing, suggesting communication with the airway (Online Supplemental Video).
Comparison with Fetal Lymphatic Malformation
Fourteen cases of fetal lymphatic malformations were imaged by fetal MR imaging at a mean gestational age of 27 weeks 6 days (range, 21 weeks 3 days to 34 weeks 6 days).
In comparison with the cases of fetal lymphatic malformations, the third and fourth branchial apparatus cysts demonstrated several distinguishing features (Table). Third/fourth branchial apparatus cysts were significantly more likely to be unilocular (P < .005) and to have a medially beaked contour (P < .005). Cystic lateral neck masses that demonstrated all 3 features in combination (unilateral, unilocular, with a medially beaked contour) were all subsequently confirmed to be third and fourth branchial apparatus cysts (P < .001). Two of the 14 lymphatic malformations were unilocular, but neither were in the expected location of the third and fourth branchial apparatus anomalies. One was in the left suboccipital region extending into the posterior neck, and the other involved the scalp near the vertex. Additionally, of the unilateral cystic lymphatic malformations that involved the deep spaces of the neck that were in reasonable locations for third or fourth branchial apparatus anomalies, none were unilocular or demonstrated the medially beaked appearance (Fig 5).
Axial (A) and coronal (B) T2 SS FSE fetal MR imaging from a representative patient with a postnatally confirmed lymphatic malformation, imaged at 34 weeks 6 days. Although the left-sided cystic neck lesion is in a location in which third and fourth branchial apparatus anomalies occur, the lesion is multiloculated with internal septations (arrowheads) and does not demonstrate a medially beaked contour, thus differentiating this case from the cases of third and fourth branchial apparatus anomalies.
Postnatal Follow-up
In all 4 cases of third and fourth branchial apparatus anomalies, the diagnosis was suggested prenatally on the basis of these imaging characteristics. All subjects underwent postnatal MR imaging after delivery to confirm the prenatally described findings and for surgical planning. In all subjects, the cyst had grown by postnatal MR imaging, and in 1 patient, the cyst demonstrated new extension across the midline. One of the 4 patients received contrast, which revealed thin peripheral enhancement typical of a cyst. Most interesting, postnatal imaging demonstrated air in the cyst in 3 of the 4 cases, indicating communication with the airway.
Operative reports, surgical pathology, and clinical follow-up were available for review in all patients. In patient 1 (Fig 1), the left neck cyst was noted to approximate the left piriform sinus, but no discrete tract was noted to ascend to the sinus, and no epithelialized tract was found by pathology. Pathology in this case initially diagnosed a thyroglossal duct cyst on the basis of the presence of thyroid tissue within the cyst. After we compared the pathologic findings with imaging findings, however, the cyst was thought more likely to represent a piriform sinus fistula. The thyroid tissue within the specimen was thought to be ectopic, likely due to a common embryologic origin. In patient 2 (Fig 2), microdirect laryngoscopy revealed a fistulous tract in the left piriform sinus that tracked directly to the cystic left neck mass. The mass was initially decompressed of air and fluid via the fistulous tract, and the tract was cauterized and oversewn. Several days later, the cyst recurred as an abscess, which was drained percutaneously and subsequently re-excised. In patient 3 (Fig 3), an operation confirmed the presence of a fistula to the piriform sinus in association with a cystic neck mass, establishing the diagnosis of third or fourth branchial apparatus anomaly. Patient 4 (Fig 4) underwent surgical repair of a left-sided third or fourth branchial cleft cyst in infancy, which was confirmed to communicate with the piriform sinus. This patient presented later in childhood with an abscess in the contralateral right side of the neck, and imaging demonstrated a fistulous tract extending from the surface of the skin to the airway. This fistula had not been detected prenatally or in infancy. On resection, this anomaly was also diagnosed as a branchial cleft remnant, and the fistula tract was thought most likely to represent a pseudofistula acquired through multiple infections.9
DISCUSSION
Third and fourth branchial apparatus anomalies are rare developmental anomalies that often present as cystic lateral neck masses, sinus tracts, or fistulas, most frequently on the left side.6 We present a series of 4 cases in which the diagnosis of third and fourth branchial apparatus anomaly was suggested prenatally on the basis of the appearance by fetal MR imaging. All lesions presented as homogeneously T2 hyperintense, unilateral, left-sided, unilocular cystic masses, extending posterior to the airway, with a medially beaked contour that tapered toward the piriform sinus. These cysts were typically located deep to the sternocleidomastoid muscle, anterior to the carotid sheath, and lateral to the visceral space of the neck. One cyst was noted to fluctuate in size on prenatal sonography, suggesting communication with the airway. In our cohort, the combination of a unilocular, unilateral cyst with a medial beak directed toward the region of the piriform sinus was seen only in patients with confirmed third or fourth branchial apparatus anomalies and not in any of the lymphatic malformations. This finding is consistent with previously published literature that stated that lymphatic malformations are only rarely unilocular.10
The differential diagnosis of congenital cystic lesions of the neck can be quite broad and includes thyroglossal duct cysts, branchial sinus anomalies, lymphatic malformations, dermoid/epidermoid cysts, ranulas, cervical thymic cysts, and cervical bronchogenic cysts.11 It is important to differentiate third and fourth branchial cleft cysts from these entities because treatment options may vary, and third and fourth branchial apparatus cysts are associated with specific genetic disorders.
Branchial apparatus cysts may become infected, leading to abscess formation and airway compromise; therefore, they are commonly surgically excised when they come to medical attention.11 Because branchial apparatus anomalies do not spontaneously regress with time, surgical excision is considered the definitive therapy.12 Although sclerosis has been shown to be safe13 and has been used in those with contraindications to an operation, sclerosis is not considered a definitive treatment because multiple sessions may be necessary and the fistulous connection to the piriform sinus must be definitively sclerosed to prevent recurrent infection.
Lymphatic malformations, though a more common cause of a lateral neck cyst,10,11 are not commonly superinfected, and at some institutions, they may be observed or sclerosed as opposed to resected, if they are otherwise not leading to immediate complications such as respiratory distress, because some rare cases may spontaneously regress in size with time.14
Furthermore, branchial apparatus anomalies can be associated with Treacher Collins syndrome, DiGeorge syndrome, Pierre Robin sequence, Goldenhar syndrome, and branchio-oto-renal syndrome.15 Lymphatic malformations involving the dorsal neck, commonly referred to as “cystic hygromas,” are often associated with underlying genetic abnormalities such as Turner syndrome, Noonan syndrome, or Trisomy 21. Macrocystic lymphatic malformations in the lateral neck, on the other hand, are not associated with these genetic abnormalities.14
Cervical thymic cysts, another entity on the differential diagnosis of cervical cystic lesions, account for 0.3% of all congenital neck masses16 and occur in the same location as third and fourth branchial apparatus anomalies because they arise from the thymopharyngeal duct but embryologically derive from the third branchial apparatus.17 Prior reports of thymic cysts have shown that they can be unilocular or multiloculated, are also more commonly left-sided, and can extend into the mediastinum and retropharyngeal space.18 Some authors have also described communication with the piriform sinus.17 Prenatally diagnosed cases have included prenatal sonography reports of mediastinal cysts,19 but to the best of our knowledge, there have been no published cases of prenatally diagnosed cervical thymic cysts or cases diagnosed with fetal MR imaging. Patients often present in childhood with swelling or mass effect on the airway.20 On the basis of imaging characteristics, cervical thymic cysts may be indistinguishable from third or fourth branchial apparatus anomalies but are exceedingly rare and are managed similarly with surgical excision. Thus, precise distinction of this entity from third and fourth branchial apparatus anomalies may not be possible or necessary.
Additionally, tracheoesophageal fistulas can present as a cystic midline neck mass anterior to the fetal spine. The cyst represents the dilated upper blind end of an atretic esophagus and is typically midline as opposed to the lateral neck and, thus, can be distinguished from third and fourth branchial apparatus anomalies on the basis of location.21 Tracheoesophageal fistulas are most commonly prenatally diagnosed on the basis of secondary findings of polyhydramnios and a small gastric bubble.22
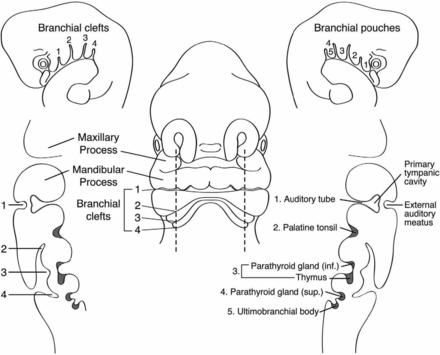
The branchial apparatus is an embryologic structure that appears during the fourth week of gestation in the walls of the embryologic pharyngeal digestive tract, forming 5 paired arches (Fig 6). The arches are separated by clefts and pouches, which reflect indentations of the ectoderm and endoderm, respectively. The arches are composed of embryonic mesoderm and neural crest cells and contain a neurovascular bundle. Thus, these structures serve as the embryonic origin of many different osseous, cartilaginous, muscular, vascular, neural, endocrine, mucosal, and cutaneous structures in the head and neck. By the seventh week of development, the second arch has overgrown the lower arches and, thus, creates the cervical sinus of His, an ectoderm-lined space that is obliterated during the course of normal development. The third branchial arch eventually gives rise to the stylopharyngeus muscle, internal and common carotid arteries, glossopharyngeal nerve, hyoid bone, inferior thyroid gland, and thymus.15 The fourth branchial cleft eventually forms the laryngeal cartilages, laryngeal and pharyngeal constrictor muscles, superior laryngeal nerve, calcitonin-secreting cells of the thyroid gland, superior parathyroid glands, left thoracic aorta, and the right proximal subclavian artery.23
Frontal and sagittal schematic representations of a 5-mm human embryo at the fifth week of gestation. The branchial apparatus with clefts and internal pouches is depicted, as well as the derivations of the major head and neck structures. Reprinted with permission from Waldhausen.12
Prior publications of third or fourth branchial cleft anomalies report they are approximately 80% left-sided, the reason being not fully understood but possibly due to asymmetric vascular development of the branchial arches.24 Bilateral anomalies, such as in one of our patients, are rare and occur in 2%–3% of cases, and, when present, are often familial.25,26 Imaging remains an important component of diagnosis of third and fourth branchial apparatus anomalies. As the case of the pathologically misdiagnosed thyroglossal duct cyst demonstrates, pathology can be misleading in the isolation of imaging findings. In one prior study, 88% of surgically excised third or fourth branchial anomalies contained ectopic thyroid tissue.9 In the absence of imaging, these cysts may be easily mistaken for thyroglossal duct cysts.
While both third and fourth branchial arch anomalies communicate with the piriform sinus, third branchial arch anomalies originate from the base of the piriform fossa and fourth branchial arch anomalies originate from the apex.27 Additionally, third branchial cleft anomalies originate cranial to the superior laryngeal nerve, whereas fourth branchial cleft anomalies originate caudal to the superior laryngeal nerve.11 While theoretically these features help to distinguish between third and fourth branchial apparatus anomalies, the size of the abnormality relative to the fetal or infant neck often results in difficulty resolving these anatomic differences.15 In practice, therefore, these entities are typically discussed together.
CONCLUSIONS
Our review of this series of surgically and pathologically proved cases of prenatal presentations of third and fourth branchial apparatus anomalies identified several features on fetal MR imaging that are highly suggestive of this entity. Namely, the presence of a medially beaked contour that tapers between the spine and airway at the level of the piriform sinus in a left-sided, unilocular cyst suggests the diagnosis of third and fourth branchial apparatus anomaly. Fetal MR imaging allows the prenatal diagnosis of these congenital anomalies, and accurate prenatal diagnosis allows proper counseling, genetic work-up, and earlier definitive management. Because these lesions do not spontaneously regress and carry the risk of superinfection, surgical excision is considered the definite therapy, and earlier treatment may potentially spare these patients from associated morbidity.
Footnotes
Paper previously presented at: Annual Meeting of the American Society of Pediatric Neuroradiology, January 10–12, 2020; Miami, Florida; it won a Cum Laude Award in Scientific Posters.
Disclosures: Yi Li—UNRELATED: Grants/Grants Pending: Radiological Society of North America, Comments: I am a recipient of the Radiological Society of North America Scholar Grant. Vicki A. Feldstein—UNRELATED: Employment: University of California, San Francisco, Department of Radiology and Biomedical Imaging, Payment for Lectures Including Service on Speakers Bureaus: University of California, San Francisco, Department of Radiology. Office of Postgraduate Education.
References
- Received April 13, 2021.
- Accepted after revision July 19, 2021.
- © 2021 by American Journal of Neuroradiology