Abstract
BACKGROUND AND PURPOSE: Large endolymphatic duct and sac (LEDS) is one of the most common anomalies seen in patients with congenital sensorineural hearing loss (SNHL), and is known to occur with other inner ear findings. Our purpose was to use high-resolution T2-weighted fast spin-echo (FSE) MR imaging to describe the features and prevalence of specific anomalies that occur in association with LEDS.
METHODS: We retrospectively reviewed MR images of the inner ear obtained in 63 patients with LEDS and in 60 control subjects. We evaluated each image for features of cochlear and vestibular dysplasia, including deficiency of the cochlear modiolus, gross cochlear dysmorphism, asymmetry of the cochlear scalar chambers, enlargement of the membranous vestibule, gross vestibular dysmorphism, and abnormality of the semicircular canals (SCC).
RESULTS: Cochlear anomalies were present in 76% of ears with LEDS. Modiolar deficiency, gross dysmorphism, and scalar asymmetry were seen in 94%, 71%, and 65% of abnormal cochleas, respectively. Vestibular abnormalities were present in 40% of ears with LEDS. Simple enlargement, gross dysmorphism, and distortion of the lateral SCC were seen in 84%, 16%, and 32% of abnormal vestibules, respectively.
CONCLUSION: Coexistent cochlear anomalies, vestibular anomalies, or both are present in most ears with LEDS, and appear as a spectrum of lesions, ranging from subtle dymorphism to overt dysplasia. The presence of coexistent anomalies in LEDS affects treatment decisions and prognosis. Newer techniques of high-resolution FSE MR imaging provide a means of exquisite characterization of LEDS, as well as more sensitive detection of associated vestibulocochlear anomalies.
Large vestibular aqueduct with large endolymphatic duct and sac (LEDS) is an anomaly that was first described as a distinct clinical entity more than 20 years ago by Valvassori and Clemis (1). Since that time, LEDS has come to be recognized as one of the most common morphologic findings associated with congenital sensorineural hearing loss (SNHL) (2) as well as the most commonly identified radiologic anomaly on cross-sectional imaging studies of the inner ear (3).
The defining bony morphologic feature of this condition is enlargement of the vestibular aqueduct, which was initially described on polytomographic scans (1, 4, 5) and subsequently on high-resolution temporal bone CT scans (2, 6, 7) (Fig 1A). The underlying process that causes this bony change of the vestibular aqueduct is enlargement of the endolymphatic sac and duct. The classical term for this entity is large vestibular aqueduct (1), which reflects the key bony feature of early tomographic descriptions. The alternative term, LEDS, however, is a broader descriptive name that reflects the underlying changes of the endolymphatic structures as seen on MR images, and is used preferentially here.
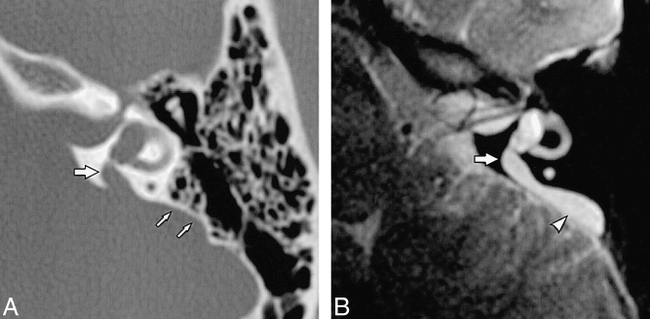
Large endolymphatic duct and sac.
A, High-resolution axial temporal bone CT scan of the left ear in a patient with LEDS shows enlargement of the bony vestibular aqueduct (large arrow). The scalloped contour of the posterior temporal bone (small arrows) is created by the enlarged endolymphatic sac, although the sac itself is not discernible on the CT scan.
B, Corresponding high-resolution axial temporal bone T2-weighted FSE MR image shows enlargement of the endolymphatic duct (arrow) and sac (arrowhead).
(Reprinted from [22] with permission.)
A specialized high-resolution MR technique using thin-section T2-weighted fast spin-echo (FSE) MR imaging has been developed to evaluate the acoustic structures in patients with SNHL (8). This technique takes advantage of the intrinsic fluid contrast of the membranous labyrinth to yield exquisite inner ear characterization (Fig 2), and has been used to assess the membranous labyrinth in patients with LEDS (9, 10) (Fig 1B).
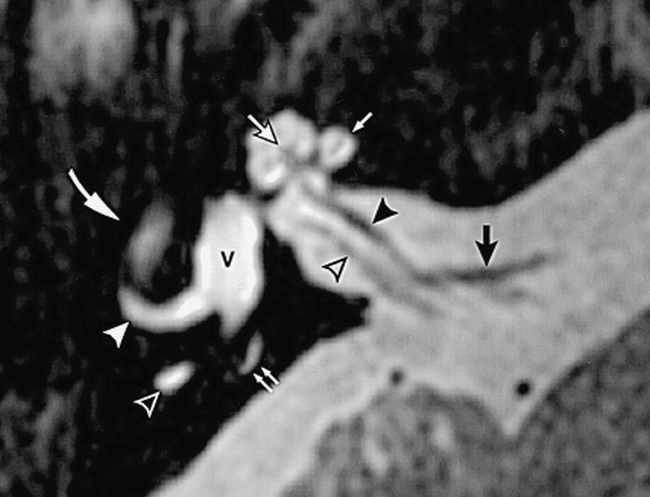
Axial high-resolution T2-weighted FSE MR image of a normal right inner ear at the midmodiolar level shows the normal cochlea and vestibule. The fluid-filled chambers of the cochlea are hyperintense and display the characteristic cochlear outline. The modiolus (white arrow with black outline) appears as a hypointense soft-tissue plug at the base of the cochlear spiral. Single straight white arrow indicates interscalar septum. The membranous vestibule (V) is ovoid in shape. The lateral (solid white arrowhead) and posterior (open white arrowhead) semicircular canals are visible. Double arrows indicate small segment of the normal endolymphatic duct; cochlear nerve (solid black arrowhead) and inferior vestibular nerve (open black arrowhead) are branches of the eighth cranial nerve; the tympanic segment of the seventh cranial nerve (curved arrow) is faintly seen; and the anterior inferior cerebellar artery (black arrow) is seen in its typical location near the porus acusticus.(Reprinted from [22] with permission.)
LEDS is commonly associated with other cochlear and vestibular anomalies, although the frequency with which these anomalies is reported to occur has been inconsistent among authors and has changed over time (1, 3–6, 10–15). The recognition of associated anomalies in LEDS is critical to the imaging assessment of these patients, since the presence of other abnormalities, especially of the cochlea, will affect prognosis and therapeutic planning.
The goal of this study was to use high-resolution T2-weighted FSE MR imaging to describe the features and prevalence of specific anomalies of the cochlea and vestibule that occur in association with LEDS. We compared these results with reports in the literature and assessed how the perspective of LEDS has changed over time with the evolution of newer imaging techniques.
Methods
We retrospectively reviewed MR images of 63 inner ears from 33 patients with a diagnosis of LEDS (30 patients with bilateral and three patients with unilateral LEDS). Imaging studies were included if the diameter of the endolymphatic duct at its midpoint exceeded 1.5 mm (1, 9) and if gross enlargement of the endolymphatic sac was present (10). We also reviewed MR images of 60 inner ears from 30 control subjects. Control images were selected from age-matched patients who were referred for evaluation of hearing loss but whose MR examinations appeared normal. All images were performed on a 1.5-T superconducting MR unit between July 1994 and March 1997. Axial FSE sequences of each inner ear were obtained using a unique protocol developed at our institution, with the following parameters: 4000/95/6 (TR/TEeff/excitations), echo train length of 32, 2-mm section thickness with 1-mm overlap, triple interleaved acquisition, 20 × 10-cm field of view, 512 × 512 image matrix, right-to-left frequency encoding, and anterior and posterior saturation bands to eliminate phase-wrap artifacts. The images were obtained using a 3-inch dual phased-array coil centered over the external auditory canals. Acquisition time for this sequence was approximately 8 minutes.
The presence of cochlear anomalies was assessed on each image in terms of three criteria. The cochlear modiolus was determined to be normal, deficient, or absent (Fig 3). The cochlea was examined for gross dysmorphism, defined as distortion of the normal cochlear contours (Fig 4). The individual scalar chambers and interscalar septa were assessed, noting the relative sizes of the anterior and posterior scalar chambers and whether the bony septa were deficient. Scalar symmetry was expressed as a ratio of the diameter of the anterior to the diameter of the posterior chambers on axial cross sections, and was considered abnormal when the ratio exceeded 1.1 (Fig 5).
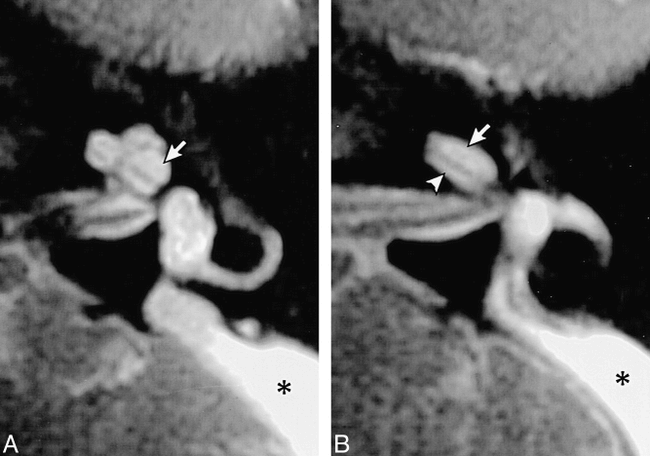
Modiolar deficiency with LEDS.
A, Axial T2-weighted FSE MR image of the left inner ear in a patient with LEDS shows mild modiolar deficiency. The modiolus (arrow) appears attenuated and flattened, unlike the normal modiolus seen in figure 2. The endolymphatic duct (section mark) and sac (asterisk) are markedly enlarged.
B, Axial T2-weighted FSE MR image in another patient shows moderate modiolar deficiency with near absence of the normal soft-tissue core at the central cochlear axis (arrow). Asterisk indicates endolymphatic sac.
(Reprinted from [22] with permission.)
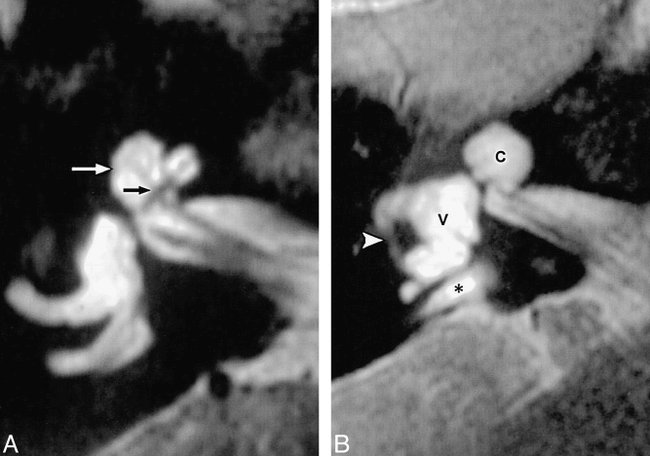
Cochlear dysplasia with LEDS.
A, Axial T2-weighted FSE MR image of the right inner ear in a patient with LEDS shows mild dysplasia. The cochlear outline is distorted, and the normal notch between the middle and apical turns laterally (white arrow) is blunted. Note that the modiolus is deficient (black arrow).
B, Axial T2-weighted FSE MR image in another patient shows severe dysplasia. The cochlea (C) appears as a common cavity, the internal architecture is lost, and the modiolus is absent. The vestibule also shows severe dysplastic changes, including gross vestibular enlargement (V) and hypoplasia of the lateral semicircular canal (arrowhead). A portion of the enlarged endolymphatic duct is also apparent (asterisk).
(Reprinted from [22] with permission.)
Scalar asymmetry with LEDS.
A, Axial T2-weighted FSE MR image of the left inner ear in a patient with LEDS shows enlargement of the anterior scalar chamber (arrow). The anterior chamber is composed of the scala vestibuli and scala media. The endolymphatic sac (asterisk) is markedly enlarged.
B, Axial image 2 mm below A also shows asymmetric enlargement of the anterior chamber (arrow). The posterior chamber (arrowhead) comprises the scala tympani, which is normal in size. Asterisk indicates endolymphatic sac.
(Reprinted from [22] with permission.)
The presence of vestibular anomalies was assessed by several criteria. To evaluate the size of the membranous vestibule, we measured the transverse diameter of the vestibule in the axial plane and expressed that measurement as a ratio with the inner diameter of the lateral semicircular canal (SCC). The vestibule was considered abnormally large when the ratio exceeded 1.2 (Fig 6A). The vestibule was evaluated for gross dysplasia, defined as distortion of the normal vestibular contour apart from simple enlargement, with or without abnormality of the lateral SCC (Fig 6B). The size and shape of the endolymphatic sac and duct were assessed, which were by definition enlarged in patients with LEDS.
Vestibular dysplasia with LEDS.
A, Axial T2-weighted FSE MR image of the right inner ear in a patient with LEDS shows enlargement of the membranous vestibule. The ratio of the transverse dimension of the membranous vestibule (black arrow) to the inner diameter of the lateral semicircular canal (white arrow) is increased. Dysplasia of the cochlea is also apparent, manifest as a scala communis, or unpartitioned scalar chamber at the apex (arrowhead).
B, Axial T2-weighted FSE MR image in another patient shows severe vestibular dysplasia. The membranous vestibule (V) is enlarged and distorted, and the lateral semicircular canal (arrowhead) is hypoplastic. Marked enlargement of the endolymphatic duct and sac is apparent (asterisk).
(Reprinted from [22] with permission.)
The thresholds for determining abnormality of ratios for both scalar asymmetry and vestibular enlargement were chosen empirically; namely, the threshold values were defined as the upper limit of measurements obtained from control images.
The 123 images of the inner ear were reviewed independently by two neuroradiologists. Agreement between the two reviewers was measured using the correlation coefficient r. In cases in which the radiologists' interpretations differed, consensus was reached during a joint review session. Statistical significance of the differences in findings between normal and LEDS images was determined using a two-tailed Student's t-test.
Results
Among all ears with LEDS, 53 (84%) had either cochlear or vestibular anomalies or both. Cochlear anomalies were present in 48 (76%) of the images from patients with LEDS. Most abnormal cochleas featured a combination of modiolar deficiency, dysmorphism, and scalar asymmetry (Table 1). Of these findings, deficiency or absence of the modiolus was the most common, present in 45 cases (71% of all cases, 94% of abnormal cochleas) (Fig 3). Modiolar deficiency was the sole finding in 13 cases (21% of all cases, 27% of abnormal cochleas). Gross cochlear dysmorphism was present in 34 cases (54% of all cases, 71% of abnormal cochleas), all but one of which also showed modiolar deficiency (Fig 4). Scalar asymmetry was present in 31 cases (49% of all cases, 65% of abnormal cochleas), but occurred as an isolated anomaly in only one case (Fig 5). The average value of scalar ratios for images with positive findings was 1.45, compared with 0.97 for images with negative findings. Scalar asymmetry was accompanied by gross cochlear dysmorphism in all but four cases.
Prevalence of anomalies of the cochlea in patients with LEDS
Anomalies of the vestibule were present in 25 (40%) of the images from patients with LEDS (Table 2). Simple enlargement of the membranous vestibule was present in 21 cases (33% of all cases, 84% of abnormal vestibules) (Fig 6A). The average value of vestibule-to-semicircular canal (SCC) ratios for positive images was 1.52, compared with 1.04 for negative images. Gross dysplasia in addition to enlargement was present in four cases (7% of all cases, 16% of abnormal vestibules). All four cases with gross vestibular dysplasia also showed abnormal configuration of the lateral SCC, manifest as flattening of the normal arc and irregular caliber of the canal (Fig 6B). Four cases with simple vestibular enlargement also showed similar but less striking abnormalities of the lateral SCC. Anomalies of the vestibule were present in 20 (42%) of the LEDS cases that had cochlear anomalies and in five (33%) of the LEDS cases that had a normal cochlea.
Prevalence of anomalies of the vestibule in patients with LEDS
The frequency of occurrence of each of these findings showed a statistically significant difference between LEDS and control images (P < .0001). The correlation coefficients of these findings between the two observers ranged from 0.85 to 0.94, indicating a high degree of interobserver agreement.
Discussion
LEDS has come to be recognized as the most common radiologic finding in patients with early-onset SNHL (1–3, 9, 10). The distinctive imaging features of this syndrome are enlargement of the bony vestibular aqueduct on CT scans and enlargement of the endolymphatic sac and duct on MR images (Fig 1). Because many of these children have useful hearing initially that gradually diminishes over the first several years of life, the question of therapeutic intervention to interrupt the progression of hearing loss has been raised, although the results of such intervention have been disappointing (2). Associated anomalies of the membranous labyrinth, especially of the cochlea, significantly affect the nature and outcome of any therapeutic plan; for example, successful cochlear implantation depends on an intact underlying cochlea. In this study, we reviewed the high-resolution MR images of the inner ear in a cohort of patients with LEDS in an attempt to refine the types and prevalences of inner ear anomalies associated with this condition further.
The coexistence of other inner ear anomalies with LEDS has been observed since this condition was first described, although the relative frequency of specific anomalies reported in the literature has been inconsistent. Early studies based on polytomographic scans of patients with LEDS showed a 15% to 25% prevalence of cochlear anomalies associated with bony vestibular aqueduct enlargement (1, 11). Several recent studies based on CT and MR imaging have reported cochlear anomalies in 25% to 30% of patients with LEDS (10, 12–14). A recent report of a study using high-resolution CT has suggested that the cochlea may be abnormal in all ears with LEDS (15). Coexistent anomalies of the vestibule in LEDS have been reported to occur more often than cochlear anomalies, although the cited frequencies vary widely. Polytomographic, CT, and MR imaging studies have shown anomalies of the vestibule ranging from 5% to 60% of patients with LEDS, with no evidence of a greater sensitivity of detection for any particular imaging technique (1, 3–5, 10–14).
The association between LEDS and other inner ear anomalies has also been suggested by histopathologic studies of congenital cochlear lesions. Enlargement of the endolymphatic sac and duct is seen as part of the constellation of histologic findings in cochlear dysplasia (16, 17).
To appreciate fully the subtle imaging findings of the cochlear and vestibular anomalies associated with LEDS, a clear understanding of the normal anatomy is necessary (Fig 2). The normal cochlea on axial FSE MR images has a characteristic contour that follows each of the cochlear turns; on midmodiolar section, the apical and middle turns are clearly distinguishable. The normal modiolus is composed of spongy bone and cochlear nerve ganglia at the central axis of the cochlear spiral. On FSE MR images, the modiolus appears as a bowtie- or crown-shaped low-signal core at the central base of the cochlea, with thin fingerlike projections extending into the interscalar septa and between the individual turns of the cochlea (Fig 2). The scalar chambers on either side of the interscalar septum are similar in size. The normal membranous vestibule is ovoid and flared anteriorly; the width of the vestibule in the coronal dimension is roughly the same as the inner bony diameter of the adjacent lateral SCC. In this study, the ratio of the vestibular width to the SCC diameter on control images ranged from 0.70 to 1.1, with a mean of 0.92.
The normal control images in this study were selected from a pool of patients with hearing loss who were referred for imaging evaluation, inasmuch as the retrospective method of this study did not allow the opportunity to evaluate asymptomatic volunteers. Selecting the control group in this way introduces a source of potential bias. Future investigations should include prospective evaluation of asymptomatic control subjects, although the benefit of including such subjects must be weighed against the potential risk of sedation in very young volunteers.
Cochlear Anomalies
In discussing the specific features of cochlear dysplasia, it is useful to refine the nature of cochlear changes associated with LEDS by looking beyond the general description of dysplasia to the specific features of modiolar deficiency, gross dysmorphism, and scalar chamber asymmetry.
Modiolar Deficiency
The modiolar abnormalities in this study appeared as a spectrum, ranging from mild flattening and attenuation to complete absence (Fig 3). Modiolar deficiency was present in three fourths of the cochleas in our cohort group. Modiolar deficiency was a feature of nearly all cochleas that had multiple abnormal findings, although it occasionally occurred by itself. Modiolar deficiency probably represents the mildest form of cochlear dysplasia. An understanding of normal modiolar appearance on high-resolution MR images enhances the sensitivity in detecting and recognizing previously underdiagnosed subtle cochlear dysplasias.
Gross Dysmorphism
Gross cochlear dysmorphism was present in just over half the cases in this study. Dysmorphism occurred as a spectrum, although the changes of most affected cochleas could be classified as mild or severe. The mildly dysmorphic cochleas typically showed subtle distortion of the normal cochlear outline, often with a broad apical contour and loss of the notch between the apical and lateral middle turns (Fig 4A). The severely dysmorphic cochleas showed obvious dysplastic changes, with loss of internal architecture, common cavity, and severe modiolar deficiency (Fig 4B). Mild cochlear dysmorphism, which was present in roughly one third of the cases in our series, was more common than severe dysmorphism, which was present in roughly one fifth of the cases. The prevalence of severe dysmorphism in our series is comparable to the overall prevalence of cochlear anomalies reported in other early studies of LEDS, which suggests that only the more severe cochlear anomalies may have been recognized previously. The notable exception in the recent literature is the study by Lemmerling et al (15), which showed cochlear anomalies on CT scans in all patients with LEDS. That study emphasized evaluation of the modiolus in the detection of an abnormal cochlea.
Scalar Asymmetry
Scalar chamber asymmetry was also a frequent finding in dysplastic cochleas in this study. When present, asymmetry was manifest as relative enlargement of the anterior chamber (Fig 5). The anterior scalar chamber comprises primarily the perilymphatic scala vestibuli, but also contains the smaller endolymphatic scala media (cochlear duct). These two scalar structures are separated by Reissner's membrane, which is not visible on MR images. The posterior chamber comprises the perilymphatic scala tympani.
The scalar asymmetry we observed in LEDS mimics that seen in dysplastic cochleas, and this finding in LEDS is most likely a manifestation of coexistent cochlear dysplasia rather than of diffuse endolymphatic enlargement. When we initially noted the asymmetry of the scalar chambers on MR images of LEDS, we postulated that the anterior chamber enlargement might be due to enlargement of the scala media as an expected manifestation of generalized endolymphatic enlargement (the scala media comprises the endolymphatic cochlear duct, which is in contiguity with the endolymphatic sac and duct). Histologic sections from patients with cochlear dysplasia, however, show that distortion of the scalar structures involves enlargement of the scala vestibuli, not the scala media (16–18). One report presented histologic findings of endolymphatic sac enlargement in a patient with Mondini disorder but with no evidence of cochlear endolymphatic hydrops (19).
Dysplastic cochleas frequently show incomplete formation of the interscalar septum, known as incomplete partition, which typically involves the apical turns and results in a common apical scalar chamber, or scala communis. Toward the base, the partially partitioned scalar structures are distorted, with relative enlargement of the anterior chamber (Fig 5).
Vestibular Anomalies
Anomalies of the vestibule were relatively common in this series, present in approximately 40% of cases, which is comparable to results of the majority of other studies of LEDS. Simple enlargement of the membranous vestibule is the most common type of vestibular anomaly associated with LEDS, and has been observed since the earliest studies on the subject (1, 6, 11). Our criterion for anomaly used the ratio of the transverse dimension of the vestibule to the inner bony diameter of the lateral SCC (Fig 6). The use of this ratio was based on the assumption that the SCC provides an internal control for judging vestibular size. This assumption may not be valid in all cases, as distortion of the SCC has been described as a feature of complex inner ear anomalies associated with LEDS (1, 12, 13). Indeed, eight of our patients with vestibular dysplasia showed abnormally formed lateral SCCs. Nonetheless, there was a statistically significant difference between the LEDS and control groups in our series, indicating that enlargement based on this definition represents either coexistence of an associated anomaly or a secondary manifestation of the condition itself. In either case, this criterion provides a reasonable objective measure of a finding that otherwise would be left to subjective interpretation alone.
Isolated LEDS
Isolated LEDS is an uncommon lesion, seen in only 14% of affected ears. The coexistence of other anomalies is a distinguishing feature that plays an important role in the clinical evaluation of and therapeutic planning for LEDS. The presence of cochlear anomalies complicates the surgical evaluation of LEDS and is a relative contraindication to endolymphatic sac surgery. In fact, poor surgical response may in part be due to underrecognition of coexisting anomalies that compromise therapeutic outcome. The concept of a “fragile cochlea” has been used to explain sudden incremental hearing loss due to minor trauma in patients with LEDS (2, 7), which also may be attributable to undetected coexistent cochlear anomalies. Newer imaging techniques with higher sensitivities for detecting coexistent anomalies may allow better refinement of the classification of isolated and complex LEDS. Such refinements will yield more selective patient populations for consideration of therapeutic intervention. Coexisting anomalies may also impact the response to related surgical therapies, such as cochlear implantation.
Surgical occlusion and obliteration of the endolymphatic sac have been tried as therapeutic options in these patients, but even for isolated LEDS, endolymphatic sac surgery has met with limited success in most cases (2). Recently, there have been promising reports of stabilization of the progressive hearing loss that affects patients with LEDS (20). Other authors, however, have not been able to show a beneficial effect of long-term follow-up of these patients (21).
Conclusion
Coexistent inner ear anomalies are common in LEDS, seen in 84% of cases. Cochlear anomalies are present in most ears with LEDS and appear as a spectrum, ranging from subtle modiolar deficiency to gross dysplasia. Modiolar deficiency is the most frequent cochlear finding, and its prevalence has been previously underreported; it represents a mild form of dysplasia. Scalar asymmetry is commonly seen in conjunction with gross cochlear dysmorphism, both of which are also manifestations of cochlear dysplasia. Anomalies of the vestibule, particularly enlargement of the membranous vestibule, are quite common, and have been recognized since LEDS was first described. Gross vestibular dysplasia and SCC abnormality are also part of the spectrum of associated vestibular anomalies. Isolated LEDS is an uncommon lesion, present in only one of six affected ears. The presence of coexistent anomalies in LEDS affects treatment decisions and prognosis. Newer techniques of high-resolution FSE MR imaging provide a means of exquisite characterization of LEDS and more sensitive detection of associated vestibulocochlear anomalies.
The mechanism by which LEDS causes progressive SNHL is still unknown. Several possible mechanisms of cochlear damage have been proposed, including injury caused by pressure effects, physiologic endolymph dysfunction, and susceptibility to minor trauma (2). The recognition of associated cochlear anomalies may lead to a better understanding of the clinical course of hearing loss in these patients as well as provide guidance in selecting clinical intervention. It is likely that the high prevalence of coexistent cochlear anomalies represents a manifestation of a common underlying embryologic insult, and that LEDS patients with and without other anomalies simply have different manifestations of the broad spectrum of inner ear dysplasia.
Footnotes
↵1 Address reprint requests to H. Christian Davidson, MD.
References
- Received January 26, 1998.
- Accepted after revision April 19, 1999.
- Copyright © American Society of Neuroradiology