Abstract
BACKGROUND AND PURPOSE: The brain distribution of 99mTc-hexamethylpropyleneamine oxime (HMPAO) correlates with regional brain perfusion, whereas 99mTc-ethyl cysteinate dimer (ECD) reflects not only perfusion but also the metabolic status of brain tissue. We compared 99mTc-ECD single-photon emission CT (SPECT) with 99mTc-HMPAO SPECT early after recanalization by local intraarterial thrombolysis (LIT) in patients with acute embolic middle cerebral artery occlusion. We also assessed the predictive value of 99mTc-HMPAO and 99mTc-ECD SPECT for the development of ischemic brain damage.
METHODS: 99mTc-HMPAO and consecutive 99mTc-ECD SPECT studies were performed in 15 patients within 3 hours of LIT. The two SPECT studies were obtained independently using a subtraction technique. SPECT evaluation was performed using semiquantitative region-of-interest analysis. Noninfarction, infarction, and hemorrhage were identified by follow-up CT or MR imaging.
RESULTS: Forty-five lesions were identified (21 noninfarctions, 19 infarctions, and five hemorrhages). Regardless of 99mTc-HMPAO SPECT findings, lesions showing isoactivity (count rate densities of 0.9 to 1.1 as compared with the contralateral side) on 99mTc-ECD SPECT were salvaged. Lesions with hypoactivity (values < 0.9) on 99mTc-ECD SPECT developed irreversible brain damage. Hemorrhage appeared in lesions with both hyperactivity (values > 1.1) on 99mTc-HMPAO SPECT and hypoactivity on 99mTc-ECD SPECT.
CONCLUSION: The brain distribution of 99mTc-ECD in a reperfused area identified by 99mTc-HMPAO SPECT early after recanalization of acute ischemic stroke is dependent on cerebral tissue viability. By combining 99mTc-ECD and 99mTc-HMPAO SPECT, performed within the first few hours of LIT, it is possible to identify patients at risk for hemorrhagic transformation reliably.
The past decade has seen the development of several radiotracers for imaging cerebral blood flow (CBF) with single-photon emission CT (SPECT), of which the most widely used is technetium-99m–labeled hexamethylpropyleneamine oxime (99mTc-HMPAO) (1). Technetium-99m–labeled ethyl cysteinate dimer (99mTc-ECD, or bicisate) is a new tracer that is thought to have properties similar to 99mTc-HMPAO (2, 3). Both radiotracers are stable, lipophilic, small, and circulate with the blood to the brain, at which point they readily pass through the blood-brain barrier. Intracellularly, the two tracers are metabolized and retained for a long time (4–6). ECD is transformed by intracellular esterases from a diester into a diacid complex. This polar metabolite does not cross the cell membrane and therefore is trapped in the neural cell (3, 7). In contrast to ECD, a steric transformation of the HMPAO chelate linked to intracellular glutathione activity has been proposed (4, 8), and the influence of the redox state on the interstitial space has recently been discussed (9). The distribution of both tracers in the brain correlates with regional brain perfusion in healthy volunteers and in patients with chronic stroke (10–14). However, early studies have reported that 99mTc-ECD SPECT does not show reflow hyperemia in patients with subacute stroke (14, 15), and that 99mTc-ECD reflects not only perfusion but also the metabolic status of brain tissue (16).
Thrombolysis using intravenous recombinant tissue plasminogen activator (rTPA) for acute ischemic stroke is a promising procedure in that it increased the number of patients who had a good functional outcome in the National Institute of Neurological Disorders and Stroke trial (17). Additionally, the recently published PROACT trial may renew interest in local intraarterial thrombolysis (LIT) in light of the fact that it was found to enhance the arterial recanalization rate (18). However, the greatest problem reportedly associated with thrombolytic therapy has been hemorrhagic transformation, which may occur as a result of early recanalization within a few hours after the insult in some patients and result in rapid deterioration of the patient's neurologic condition (19).
In the present study, we compared 99mTc-ECD SPECT with 99mTc-HMPAO SPECT early after recanalization by LIT in patients with acute embolic middle cerebral artery (MCA) occlusion. We also examined the predictive value of 99mTc-HMPAO and 99mTc-ECD SPECT for the development of ischemic brain damage.
Methods
Patients
Fifteen patients (six women and nine men, 50–72 years old; mean age ± SD, 63 ± 7 years) underwent recanalization by LIT for acute cerebral embolism. Informed consent was obtained from all participants or their next of kin, and the study was approved by the ethics committee of Kohnan Hospital. A diagnosis of embolic occlusion was made on the basis of clinical evidence of atrial fibrillation and/or other cardiovascular indications, as well as by the mode of onset of neurologic deficits. Angiographic criteria used to confirm embolic occlusion were 1) abrupt nontapering occlusion, 2) poor collateral circulation, 3) absence of atherosclerotic changes in remote vessels, and 4) absence of retrograde filling of bi- or trifurcations of the MCA trunk. LIT was indicated according to three criteria: 1) the patient had been hospitalized within 6 hours of onset; 2) no apparent hypodensity areas were observed on an admission CT scan; and 3) the site of occlusion was the MCA. Therapy was carried out using rTPA at a dosage of between 96 and 480 × 104 IU. The occluded vessel was recanalized and blood flow was successfully reestablished in all patients. Duration of angiographic recanalization after symptom onset ranged from 3.5 to 7.0 hours. CT was performed the next day and at 1 week, and MR imaging was performed 2 weeks and 1 month after treatment. The patient's neurologic status was evaluated upon admission, the following day, and 1 month after treatment according to the NIH Stroke Scale (20), which expresses the severity of neurologic status numerically.
Study Protocol
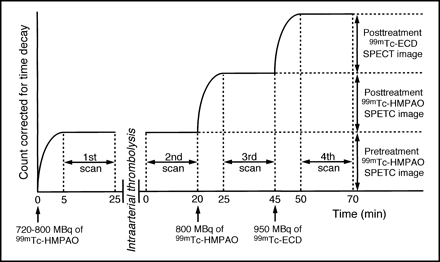
Upon admission, 99mTc-HMPAO (720 to 800 MBq) was injected intravenously and the first SPECT study was performed before initiating LIT. Posttreatment SPECT was performed within 3 hours after LIT. The patient's head was immobilized using a headholder so that bilateral external auditory meatuses were aligned with the machine-indwelling positioning crossed-light beam. The second SPECT study was begun without any additional injection of tracer. While the position of the subject's head was maintained, 800 MBq of 99mTc-HMPAO was injected and the third SPECT study was carried out. Immediately after the end of the third data acquisition, 950 MBq of 99mTc-ECD was injected and the fourth SPECT study was begun. After completing reconstruction, the second images were subtracted from the corresponding images obtained in the third study by using the pixel-by-pixel method after correcting for time differences in data acquisition. Posttreatment 99mTc-HMPAO SPECT scans were thus obtained. The tomographic images from the third study were then subtracted from the corresponding images obtained in the fourth part using the above method to obtain posttreatment 99mTc-ECD SPECT scans (Fig 1).
Schematic presentation of the protocol for SPECT; x axis is time (min) and y axis is decay-corrected regional counts
SPECT Studies
SPECT studies were performed using a multidetector ring-type scanner, which has a detector array consisting of 64 NaI crystals in a 38-cm-diameter circle. After tomographic reconstruction, the spatial resolution and slice thickness in the center of the plane were 9 mm and 16 mm, respectively, full width at half-maximum. The energy window used in this study was 140 KeV (15%). Projection data were processed using Ramachandran's filtered backprojection after introduction of a Butterworth prefilter. Data were processed using a 64 × 64 image matrix. The tracer was administered in a quiet, dimly lit room with patients lying down with their eyes open. SPECT was initiated 5 minutes after injection of tracer. Tomographic data acquisition was performed for 20 minutes (Fig 1).
Data Analysis
Posttreatment SPECT data were analyzed semiquantitatively using region-of-interest (ROI) analysis. Of six axial slices obtained, the slice showing the ischemic region most clearly on the pretreatment SPECT scans was selected for the posttreatment SPECT examination. The ROIs from the MCA territory on the affected (A) side were determined by using the posttreatment 99mTc-HMPAO SPECT scans. In addition, we placed mirror-image ROIs in the corresponding region on the contralateral (C) side. Identical sets of ROIs were transferred to the posttreatment 99mTc-ECD SPECT studies. To assess the regional distribution of both tracers objectively, the A/C ratio was calculated. The ROIs in the MCA territory on the affected side were determined by following two different methods: 1) one ROI outlining the entire MCA territory, and 2) three circular ROIs (8.5 cm2) covering those areas in which pretreatment SPECT scans revealed a perfusion deficit. On the basis of follow-up CT and MR findings, one observer blind to the SPECT findings categorized the size of the infarct in the entire MCA territory as noninfarct, small infarct (<33%), medium infarct (33–66%), or large infarct (>66%). In addition, each circular ROI on the affected side was analyzed by the same observer and then classified into three categories: noninfarction, infarction, or hemorrhage.
First, the relationship among the count densities of each tracer in the entire MCA territory on the affected side, size of infarct, and neurologic outcome was evaluated. Second, count densities in each circular ROI on the affected side were classified as hypoactivity, hyperactivity, or isoactivity if the A/C ratio was below 0.9, above 1.1, or 0.9 to 1.1, respectively. Finally, the findings obtained on follow-up CT and MR studies were compared with the regional distribution of tracers.
Results
The pretreatment SPECT studies revealed a perfusion defect in the unilateral MCA territory, as suggested by symptoms in all patients. Clinical characteristics, posttreatment SPECT findings, CT and MR findings, and neurologic outcomes are summarized in the Table. A/C ratios on 99mTc-HMPAO SPECT scans were more than 0.9 in all cases and did not differ among the various sizes of infarct. On the other hand, although A/C ratios on 99mTc-ECD SPECT scans were 0.9 or more in patients without infarction or in those with a small infarct, the values were less than 0.9 in patients with medium or large infarcts. Hemorrhagic transformation occurred in three patients and was detected on CT scans obtained the day after treatment. Although all hemorrhages were petechial, in two of these three patients (cases 3 and 8), the hemorrhages were accompanied by extensive ischemic brain edema, and neurologic deterioration was observed. The regions corresponding to petechial hemorrhage and surrounding brain edema showed high uptake of 99mTc-HMPAO and low uptake of 99mTc-ECD on posttreatment SPECT scans. One patient (case 8) died of transtentorial herniation 4 days after treatment.
Summary of patients with embolic middle cerebral artery occlusion treated by local intraarterial thrombolysis
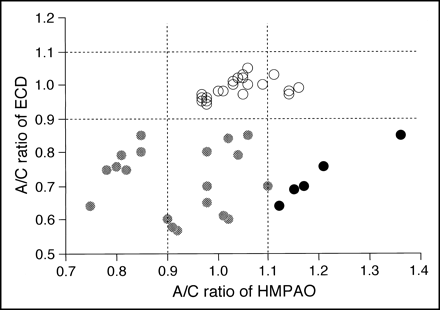
By comparing the circular ROIs distinguished on the SPECT scans with the noninfarcted, infarcted, and hemorrhagic regions determined on follow-up CT or MR studies, we identified 45 lesions (21 noninfarcted, 19 infarcted, and five hemorrhagic) in the 15 patients studied (Fig 2). The seven ROIs showing hypoactivity on posttreatment 99mTc-HMPAO SPECT scans also showed hypoactivity on 99mTc-ECD SPECT scans, with all the above areas resulting in infarction. Among the 29 ROIs showing isoactivity on posttreatment 99mTc-HMPAO SPECT scans, infarction developed in 12 ROIs showing hypoactivity on 99mTc-ECD SPECT scans but did not develop in 17 ROIs showing isoactivity. In addition, among the nine ROIs showing hyperactivity on posttreatment 99mTc-HMPAO SPECT scans, hemorrhagic transformation developed in five ROIs showing hypoactivity, whereas no abnormalities developed in four ROIs showing isoactivity on 99mTc-ECD SPECT scans. Representative studies appear in Figures 3 and 4.
Outcome versus posttreatment SPECT findings of both tracers. Count densities of the ROIs on the affected side were classified as hypoactivity, hyperactivity, or isoactivity if the A/C ratio was below 0.9, above 1.1, or 0.9 to 1.1, respectively. Open circles represent noninfarction; gray circles, infarction; black circles, hemorrhage
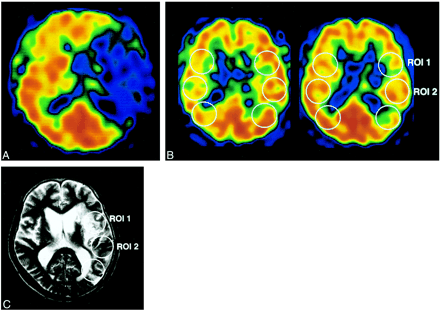
Patient 6: 58-year-old man with left-sided MCA occlusion.
A, Pretreatment 99mTc-HMPAO SPECT scan shows a perfusion defect in the left MCA territory.
B, Posttreatment 99mTc-HMPAO (left) and 99mTc-ECD (right) SPECT scans obtained 2.5 hours after recanalization by LIT.
C, MR image 2 weeks after the onset of stroke. Infarction did not develop in ROI 2, which showed hyperactivity (A/C ratio of 1.14) on 99mTc-HMPAO SPECT scan and isoactivity (A/C ratio of 0.97) on 99mTc-ECD SPECT scan. In ROI 1, however, which showed hypoactivity (A/C ratio of 0.85) on 99mTc-ECD SPECT scan, infarction did develop despite isoactivity (A/C ratio of 1.06) on 99mTc-HMPAO SPECT scan.
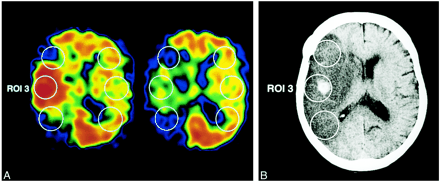
Patient 8: 61-year-old man with right-sided MCA occlusion.
A, Posttreatment 99mTc-HMPAO (left) and 99mTc-ECD (right) SPECT scans obtained 2 hours after recanalization by LIT.
B, CT scan 30 hours after the onset of stroke. Hemorrhagic transformation with ischemic brain edema developed in ROI 3, which showed hyperactivity (A/C ratio of 1.36) on 99mTc-HMPAO SPECT scan and hypoactivity (A/C ratio of 0.85) on 99mTc-ECD SPECT scan.
Discussion
We compared 99mTc-ECD SPECT with 99mTc-HMPAO SPECT early after recanalization by LIT in patients with acute embolic MCA occlusion. We also examined the predictive value of 99mTc-HMPAO and 99mTc-ECD SPECT for the development of ischemic brain damage.
Posttreatment 99mTc-HMPAO SPECT scans obtained within 3 hours of LIT showed focal iso- or hyperactivity in the region where the initial SPECT scans had revealed a perfusion deficit. These findings indicate that the ischemic regions were reperfused as a result of recanalization of occluded vessels by LIT, given that the brain distribution of 99mTc-HMPAO in patients with reflow of acute ischemic stroke could be correlated with regional brain perfusion by use of the reference method of CBF analysis (133Xe) (20). When a reperfused area showed isoactivity by 99mTc-ECD SPECT, no abnormalities developed. In contrast, in areas showing hypoactivity, both infarction and hemorrhage were observed. Therefore, our results demonstrate that the brain distribution of 99mTc-ECD in a reperfused area early after recanalization of acute ischemic stroke is dependent on cerebral tissue viability. The cerebral uptake of 99mTc-ECD reportedly reflects not only perfusion but also the metabolic status of brain tissue (16). Furthermore, the retention of 99mTc-ECD requires the presence of cytosolic esterase (16), which in turn depends on the viability of cells. Thus, the observed focal area of hypoactivity on 99mTc-ECD SPECT scans indicates a metabolic breakdown and ongoing irreversible tissue damage.
In studies attempting to determine the predictive value of early CBF imaging for tissue outcome in patients with cerebral ischemia (21–23), the physiological implications of focal hyperperfusion remain unclear. Some studies have reported improved outcome with hyperperfusion in the early stage of cerebral ischemia (24–26) while others have found no such beneficial effect (23). Different methodological and technical approaches most likely account for these discrepancies. In the present study, among the nine ROIs showing hyperactivity on posttreatment 99mTc-HMPAO SPECT scans, no abnormalities were observed in four ROIs, whereas hemorrhagic transformation developed in the remaining five ROIs. These findings suggest that early hyperperfusion in acute ischemic stroke may consist of both nutritional and nonnutritional components, as shown by Barber et al (28). The term nonnutritional hyperperfusion corresponds to luxury perfusion, because this reperfusion is into nonviable tissue. Nutritional hyperperfusion, in contrast, rescues potentially viable tissue from infarction. The limitations of 99mTc-HMPAO SPECT studies are that the proportion of nutritional and nonnutritional hyperperfusion cannot be determined on the basis of a single study early after reperfusion of acute ischemic stroke (27). In contrast, among the ROIs showing hyperactivity on posttreatment 99mTc-HMPAO SPECT scans, hemorrhagic transformation developed in ROIs showing hypoactivity, whereas no abnormalities were observed in the ROIs showing isoactivity on 99mTc-ECD SPECT scans. These findings suggest that by combining 99mTc-ECD and 99mTc-HMPAO SPECT, it is possible to distinguish nutritional hyperperfusion from nonnutritional hyperperfusion and to accurately predict the onset of hemorrhagic transformation.
Hemorrhagic transformation after thrombolytic therapy can be classified into petechial hemorrhage and parenchymal hematoma. Parenchymal hematomas often aggravate neurologic symptoms and adversely influence neurologic outcome, whereas petechial hemorrhage alone is usually asymptomatic (19, 29). In the present study, three patients developed hemorrhagic transformation, all of which were petechial hemorrhages. In two of these three patients, however, the hemorrhages were accompanied by extensive ischemic brain edema, and neurologic deterioration was observed. Posttreatment 99mTc-HMPAO SPECT scans showed that the regions corresponding not only to petechial hemorrhage but also to surrounding brain edema observed on the following day exhibited hyperperfusion early after thrombolysis. Therefore, we believe that a sudden rise in perfusion pressure in irreversibly damaged tissue caused extensive ischemic brain edema.
Reliable information after LIT as to the prognosis of an ischemic area is urgently required, because space-occupying infarctions, such as hemorrhagic transformation and extensive ischemic brain edema, may occur as a result of early recanalization within a few hours of the insult in certain patients, resulting in rapid deterioration in neurologic status (19). Specific but risky treatments, such as decompressive hemicraniectomy (30) or mild hypothermia (31), appear to be more appropriate for such patients. In cases in which a diagnosis of space-occupying infarction is confirmed early after LIT, the patient should be transferred immediately to an intensive care unit or to any specially equipped center where decompressive hemicraniotomy or mild hypothermia can be performed (32). A combination of 99mTc-HMPAO and 99mTc-ECD SPECT performed within the first few hours of LIT enables accurate identification of patients at risk for space-occupying infarction, which in turn allows clinicians to initiate treatment immediately that may be specific for increasing intracranial pressure. In the present study, parenchymal hematoma, a condition that often aggravates neurologic symptoms, unexpectedly did not develop. However, since hyperperfusion leading to an increase in perfusion pressure in irreversibly damaged tissue appears to cause parenchymal hematoma after recanalization (33), a combination of 99mTc-HMPAO and 99mTc-ECD SPECT may also identify patients at risk for parenchymal hematoma.
The posttreatment SPECT procedure used in the present study is time-consuming and uncomfortable for critically ill patients. Therefore, practical difficulties limit its routine use. Improved methods of data acquisition should be investigated to reduce the total examination time.
Conclusion
The brain distribution of 99mTc-ECD in a reperfused area identified by 99mTc-HMPAO SPECT obtained early after recanalization of acute ischemic stroke was found to be dependent on cerebral tissue viability. Moreover, combining 99mTc-ECD and 99mTc-HMPAO SPECT performed within the first few hours of LIT allowed us to distinguish nutritional from nonnutritional hyperperfusion and to identify patients at risk for hemorrhagic transformation reliably.
Footnotes
↵1 Address reprint requests to Kuniaki Ogasawara, MD, Department of Neurosurgery, Iwate Medical University, Uchimaru 19-1, Morioka 020-8505, Japan.
References
- Received January 31, 2000.
- Copyright © American Society of Neuroradiology