Abstract
Summary: We present a unique finding of an elevated level of pyruvate at 2.37 ppm revealed by in vivo MR spectroscopy of a female neonate. Low fibroblast pyruvate dehydrogenase (PDH) complex activity subsequently confirmed a diagnosis of PDH deficiency. Abnormalities of brain development consistent with PDH deficiency were also evident on fetal and postnatal MR images. To our knowledge, this is the first report of pyruvate being shown in vivo in a child and the first report of MR spectroscopy aiding in the diagnosis of inborn error in pyruvate metabolism before confirmation by conventional enzymatic testing. This finding has potential implications for earlier diagnosis in patients with defects in mitochondrial metabolism.
Pyruvate dehydrogenase (PDH) deficiency is a defect of mitochondrial energy metabolism that results from mutations in one or more components of the PDH complex (1). Mutations of different components of the PDH complex have been reported and result in variable phenotypes. In >90% of patients, however, the E1∝ subunit is deficient or abnormal (1). Because the gene for this subunit is on the X chromosome, mutations in males tend to be lethal whereas affected females are common as a result of lyonization. Because variable clinical expression is present among those females who are affected, the diagnosis is often difficult to make.
Some children are born with brain and somatic malformations secondary to PDH deficiency (2). The brain malformations that are potentially identifiable by imaging include complete or partial agenesis of the corpus callosum, heterotopic gray matter, absence of the medullary pyramids and abnormal inferior olives, hydrocephalus, and cerebellar dysplasia (2–7). Clinical confirmation of the disorder requires highly specialized biochemical analyses of PDH complex activity in skin or muscle, which can take months to complete. We present the first report of a female infant with PDH deficiency in whom the CNS pyruvate was documented with MR spectroscopy well before biochemical confirmation was obtained.
Case Report
The patient was a 2.7-kg, 38-week gestation neonate born to a 33-year-old G3P1–2 Hispanic mother. Cerebral ventricular dilation was diagnosed by routine prenatal sonography at 18 weeks’ gestation. Serial sonography revealed progressive enlargement of the ventricular system (30-mm atrial diameter at 34 weeks’ gestation). Amniocentesis revealed a normal 46,XX karyotype and normal fluorescence in situ hybridization for chromosome 17p13.3 (LIS1, Miller-Dieker syndrome). Ultrafast fetal MR imaging performed at 34 weeks’ gestation on a 1.5-T clinical MR imaging system showed a markedly dysmorphic brain with ventriculomegaly, absence of the corpus callosum, cerebral parenchymal dysgenesis, brain stem hypoplasia, and cerebellar hypoplasia (Fig 1).
Ultrafast fetal MR images obtained at 34 weeks’ gestation.
A and B, Axial view half-Fourier single shot turbo spin-echo fetal MR images (12/95 [TR/effective TE]; number of acquisitions, one) show enlarged ventricular size and the diffusely abnormal gyral pattern.
C and D, Postnatal coronal view turbo spin-echo T2-weighted MR images (6000/99; number of acquisitions, two) highlight the findings along with the cerebellar subcortical cyst.
At birth, respiratory distress with significant lactic acidemia and hypoglycemia were present. The patient had mild dysmorphia, facial hypotonia, small, cupped ears, and pointed superior helices. Her head circumference plotted to the 75th percentile, with an enlarged anterior fontanelle (6 × 7 cm) and splayed sutures. Initial neurologic examination revealed an exaggerated Moro reflex, generalized hypotonia, and mild irritability.
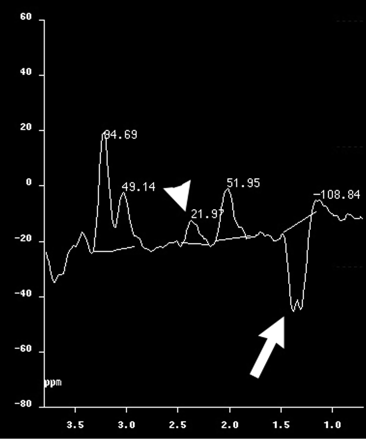
Postnatal MR imaging of the brain confirmed the malformations identified in utero and better delineated a subcortical cyst in the periphery of the hypoplastic right cerebellar hemisphere (Fig 1, Table 1). 2D chemical shift imaging MR spectroscopy (TE, 135 ms) showed a large lactate doublet at 1.33 ppm and a unique singlet at 2.37 ppm (Fig 2). Based on previous limited in vitro analysis, this was suspected to represent pyruvate (3). Phantom analysis of an aqueous solution containing 50 mmol/L sodium pyruvate and 1 mmol/L cupric sulfate reproduced the peak, supporting our conclusion that the clinically observed peak represented pyruvate.
2D chemical shift MR spectroscopy (TE, 135 ms). Arrow indicates lactate doublet at 1.33 ppm; arrowhead indicates pyruvate peak at 2.37 ppm.
Findings
Significantly elevated plasma lactate was present along with elevated CSF lactate and pyruvate (Table 2). Despite aggressive IV bicarbonate supplementation and antiepileptic therapy, the patient remained obtunded with minimal spontaneous activity, depressed gag reflex, and subclinical seizures.
Selected laboratory analyses
Because of the clinical course and probable diagnosis of PDH deficiency, the patient’s family withdrew mechanical support of the infant at 7 days. Postmortem examination, including pathologic examination of the brain, was performed (Fig 3, Table 1). Muscle and skin biopsies sent for biochemical analysis revealed significantly decreased PDH complex activity, confirming the diagnosis of PDH deficiency, but these results were not received until 8 months after the patient’s death (Table 3). Mutation analysis of the E1∝ subunit was not available.
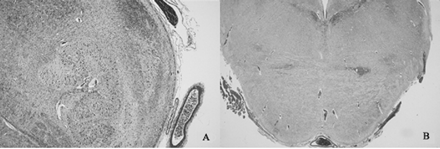
Photomicrographs from postmortem examination.
A, Transverse section of the medulla shows ectopic olivary nucleus in inferior cerebellar peduncle (Luxol fast blue cresyl violet; magnification, ×4).
B, Transverse section of the pons shows absence of descending cortical tracts in basis pontis (Luxol fast blue cresyl violet; magnification, ×1).
Fibroblast biochemical analyses
Discussion
Normal brain development requires temporal coordination of cellular migration, proliferation, and differentiation. It is becoming increasingly recognized that some children with inborn errors of metabolism may have characteristic developmental brain malformations (3). However, understanding of the mechanisms underlying these malformations and the reasons for the selective involvement of some children with biochemical disease is incomplete.
A broad array of differential diagnoses exists for an infant with abnormal brain development. Along with inborn errors of metabolism, specific gene mutations and environmental influences, such as viral infection, hypoxia, and maternal diabetes, may all result in malformations, such as pachygyria, polymicrogyria, or agenesis of the corpus callosum (3). Abnormal development of the corpus callosum, often in association with gyral malformations, has been observed in other inborn errors of metabolism, such as Zellweger syndrome, fumarase deficiency, and nonketotic hyperglycinemia (4–6). Genetic mapping of syndromes associated with specific patterns of brain dysgenesis, such as Miller-Dieker syndrome and MASA syndrome, suggest that nonmetabolic influences, such as cell adhesion and signal intensity transduction proteins, also are crucial for normal brain development (4, 8).
In this case, fetal MR imaging identified numerous brain malformations and dilated ventricles, resulting in a differential diagnosis that included nonmetabolic genetic syndromes and inborn errors of metabolism. Without a known family history or genetic testing, in utero diagnosis of PDH deficiency is difficult (9). Although in utero MR spectroscopy has been reported, it is technically challenging to perform and difficult to accurately interpret (10, 11). The range of normal lactate in the premature brain is not known, and the fetal CNS concentration of lactate and pyruvate may be falsely reduced by placental clearance (12).
Glycolytic metabolism is responsible for production of pyruvate from glucose. During anaerobic conditions, the oxidation and reducing equilibrium in mitochondria is reduced. Through end product inhibition, the increase in nicotinamide adenine dinucleotide decreases the PDH complex-mediated conversion of pyruvate to acetyl-coenzyme A, leading to an increase in pyruvate levels. Our patient had significant and persistent elevation of lactate, as appreciated by both blood and spinal fluid measurements. The MR spectroscopy observation of a large lactate doublet at 1.33 ppm and a narrow singlet at 2.37 ppm, corresponding to pyruvate, would be consistent with a defect in pyruvate metabolism. The long TE (135 ms) 2D chemical shift imaging technique makes it unlikely that the narrow peak seen is the result of glutamate or glutamine (2.0–2.45 ppm), which usually are broad, show coupling, and are better seen with shorter TE.
The only other report to suggest the observation of pyruvate with MR spectroscopy was in a series of adults with cerebral radiation injury that was progressive during an 18-month period. An unknown resonance in the 2.37- to 2.40-ppm region was detected in the temporal lobes of four of 10 patients. This finding was confined to spectra where lactate was present, which was in those patients with the highest severity of radiation injury. The authors surmised that the peak represented pyruvate, although their postulate was not confirmed by in vitro studies (13).
We reassessed MR spectroscopy studies of other patients with biochemically confirmed PDH deficiency for resonance in the 2.37 ppm region. No comparable peaks were seen. This is probably most reflective of the severity of the infant’s disease, as evidenced by plasma/CSF lactate values and minimal PDH activity (Tables 2 and 3).
Because MR spectroscopy is noninvasive and can be performed in conjunction with MR imaging, it is being used more frequently to assess children with known or clinically suspected metabolic disorders (14). MR spectroscopy has been used to aid evaluation in cases of galactosemia (15), nonketotic hyperglycinemia (16), and other inborn errors of energy metabolism (17, 18). Reduction in normal major metabolites (choline, creatine, and N-acetylaspartate) and elevation in lactate often are markers of CNS abnormality, although they are nonspecific (14). Previous MR spectroscopy studies of patients with PDH deficiency have shown the presence of lactate but not pyruvate (15, 16). One previous case report included abnormally elevated alanine (1.5 ppm), an alternate pathway product of pyruvate metabolism (14). This was not identified in our patient.
The levels of CSF and plasma lactate in this infant were suggestive but not diagnostic of PDH deficiency. In conjunction with prenatal and postnatal MR imaging showing brain malformations, including agenesis of the corpus callosum, consideration was heightened for inborn errors of metabolism involving energy metabolism. Pending biochemical confirmation, the MR spectroscopy added strong evidence toward the diagnosis. Considering that the specific finding of a pyruvate peak by proton MR spectroscopy has not been reported for respiratory chain defects or other defects in mitochondrial energy metabolism, our findings most likely reflected global reduction in PDH biochemical activity in this particular infant.
Conclusion
The use of MR spectroscopy of the brain should be an important adjunct in the evaluation of an infant with lactic acidosis and/or identified brain malformations suggestive of a metabolic disorder. In this patient, the elevated lactate and pyruvate revealed by MR spectroscopy, along with brain malformations and elevated serum and CSF lactate/pyruvate, had a direct impact on the clinical assessment, care, and decision making for this infant, months before the biochemical confirmation of PDH deficiency was received.
References
- Received November 26, 2002.
- Accepted after revision January 24, 2003.
- Copyright © American Society of Neuroradiology