Abstract
BACKGROUND AND PURPOSE: The presence of edema will result in increased brain volume, which may obscure progressing brain atrophy. Similarly, treatment-induced edema reduction may appear as accelerated brain tissue loss (pseudoatrophy). The purpose of this study was to correlate brain tissue properties to brain volume, to investigate the possibilities for edema correction and the resulting improvement of the precision of automated brain volume measurements.
MATERIALS AND METHODS: A group of 38 patients with clinically isolated syndrome or newly diagnosed MS were imaged at inclusion and after 1, 2, and 4 years using an MR quantification sequence. Brain volume, relaxation rates (R1 and R2), and proton density were measured by automated software.
RESULTS: The reduction of normalized brain volume with time after inclusion was 0.273%/year. The mean SDs were 0.508%, 0.526%, 0.454%, and 0.687% at baseline and 1, 2, and 4 years. Linear regression of the relative change of normalized brain volume and the relative change of R1, R2, and proton density showed slopes of −0.198 (P < .001), 0.156 (P = .04), and 0.488 (P < .001), respectively. After we applied the measured proton density as a correction factor, the mean SDs decreased to 24.2%, 4.8%, 33.3%, and 17.4%, respectively. The observed atrophy rate reduced from 0.273%/year to 0.238%/year.
CONCLUSIONS: Correlations between volume and R1, R2, and proton density were observed in the brain, suggesting that a change of brain tissue properties can affect brain volume. Correction using these parameters decreased the variation of brain volume measurements and may have reduced the effect of pseudoatrophy.
ABBREVIATIONS:
- BPF
- brain parenchymal fraction
- BPV
- brain parenchymal volume
- ICV
- intracranial volume
- NEDA
- no evidence of disease activity
- PD
- proton density
- R1 and R2
- relaxation rates
- SR1, SR2, and SPD
- correlation slopes
Brain atrophy and accelerated brain volume loss are present early in the disease course of MS and correlate with the presence and development of physical disability and cognitive impairment.1 When assessing disease activity in MS, the term “no evidence of disease activity” (NEDA) is increasingly used, where NEDA-3 is defined by the combination of the following: 1) no relapses, 2) no brain MR imaging activity (no new or enlarging T2 lesions or gadolinium-enhancing lesions), and 3) no sustained disability worsening (Expanded Disability Status Scale progression). However, given the clinical importance of brain atrophy, it is desirable to expand the NEDA concept to NEDA-4, in which “no increased brain volume loss/brain atrophy rate” is added.2 Disease-modifying immunomodulatory treatment options in MS vary in efficacy and adverse effect profile. Personalized treatment for a patient with MS requires clinically feasible and reliable assessment of disease activity status at treatment initiation and treatment follow-up. Although there is not yet enough evidence to support the use of brain volume measures to monitor treatment response or for making treatment decisions in individual patients, brain volume loss is now often incorporated in large clinical trials of potential new MS drugs.3 Monitoring brain volume and brain volume changes with time in clinical practice, however, is often hampered by the additional time and effort burden of brain segmentation methods and their inherent precision. Moreover, disease activity induces changes in the intra- or extracellular water content, which, in turn, can lead to changes in total brain volume. Therefore, resolution of inflammatory edema after a successful treatment may appear as atrophy, known as “pseudoatrophy,” as previously reported in patients with early-onset MS.4,5 Likewise, an increase in disease activity may increase the total edema, visible only as diffuse hyperenhancement on T2-weighted or FLAIR images. This may lead to swelling of the brain, obscuring the progressing atrophy.
Recent progress in quantitative MR imaging permits measurement of physical properties of the brain, such as relaxation rates (R1 and R2) and proton density (PD) in a reasonable scan time.6 Axonal damage, gliosis, inflammation, and edema are related to changes in these values.7⇓⇓⇓⇓–12 Therefore, it can be expected that the mean values of R1, R2, and PD have a relationship with processes that may lead to global volume changes in the brain. SyntheticMR (SyMRI; SyntheticMR, Linköping, Sweden) is a brain segmentation tool with high precision,13⇓–15 which is based on quantitative MR imaging. It therefore combines the ability to fully automatically measure brain volumes and simultaneously obtain values for mean R1, R2, and PD of brain tissue.
The aim of this study was to investigate the relation between brain volumes of patients with clinically isolated syndrome and those with MS using a fully automatic brain volume segmentation method and the mean R1, R2, and PD values in these brains. Provided that these 2 measures correlated, the second aim was to use R1, R2, and PD as a correction factor for brain volume and investigate whether the precision of the brain segmentation method would improve.
Materials and Methods
Subject Group
The study included 44 patients (29 women) who were consecutively enrolled in a prospective longitudinal cohort study of patients with clinically isolated syndrome and those with newly diagnosed MS. All patients fulfilled the revised McDonald criteria for clinically isolated syndrome or MS at inclusion.16 Brain MR imaging was performed at baseline and at 1, 2, and 4 years. Retrospectively, 1 subject was excluded due to severe hydrocephalus; for 5 subjects, the study was prematurely terminated due to moving to another place or pregnancy. The remaining 38 all had 4 time points (baseline, 1, 2, and 4 years), a total of 152 examinations. Examinations were performed during the daytime, between 8 am and 4 pm. Of these, 104 (68%) were performed in the morning, and 48 (32%), in the afternoon. The included subjects had a mean age of 35 ± 11 years (range, 21–67 years) and a median Expanded Disability Status Scale of 2.0 (interquartile range, 1.0–2.0) at baseline. Further patient characteristics are presented in Table 1.
Patient diagnoses, relapse status, and treatment status at baseline and at follow-up MRI
Ethics Statement
The study was approved by the regional ethics committee, and written informed consent was obtained from all participants.
Acquisition
The MR imaging quantification method was a multiecho, multidelay saturation recovery spin-echo sequence, which has been described previously.6 The multisection sequence consisted of section-selective saturation pulses, interleaved with a Carr-Purcell-Meiboom-Gill acquisition of 5 echoes at multiples of 14-ms TE. The saturation pulse acts on a section n, while the subsequent acquisition acts on a section m. By a variable shift between sections n and m, multiple effective delay times were created between the saturation and acquisition of each section. The sequence was repeated 4 times, resulting in a matrix of 20 images per section, at 5 different TEs and at 4 different saturation delay times. All these images had a different effect of R1 and R2 relaxation. The TR was 4280 ms with 43 sections of 3-mm thickness. The FOV was 230 mm with an in-plane resolution of 1.4 mm. The scan time was 6 minutes and 20 seconds on an Achieva 1.5Tscanner (Philips Healthcare, Best, the Netherlands).
Postprocessing
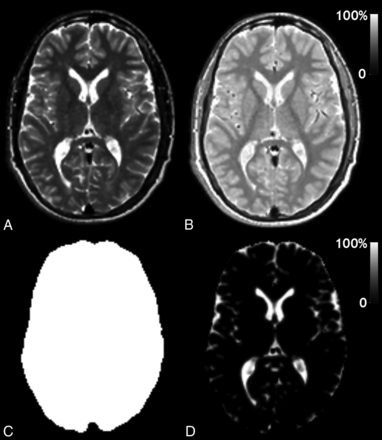
The calculation of the intracranial volume (ICV), the brain parenchymal volume (BPV), and brain parenchymal fraction (BPF = BPV / ICV) is an automatic function of SyMRI 9.0. The same software also shows the R1, R2, and PD maps of the entire acquisition volume. Typical images and maps from this software in our patients are shown in Fig 1. An adjustment was made in the software to extract the average R1, R2, and PD values for the entire brain and gray and white matter. This was done by taking the ICV and removing all CSF (clipped at 50% partial volume) and subsequently eroding 1 pixel on all sides of the volume to remove the partial volume effects of CSF. The available GM and WM segmentation maps were used to retrieve the average R1, R2, and PD of GM and WM inside the eroded volume.
Typical output of the SyMRI software: A, Synthetic T2-weighted image of a section of the brain of one of the patients. This synthetic image was generated from the R1, R2, and PD maps in combination with a TE/TR = 100/4500 ms. B, PD map on a scale of 0%–100%, in which 100% corresponds to pure water at 37°C. C, Automatically generated intracranial mask (ICV). D, Automatically generated CSF partial volume map on a scale of 0%–100%. The brain parenchymal volume is calculated as the sum of all ICV minus the sum of all CSF. The brain parenchymal fraction is calculated as BPV/ICV.
Finding the Correlation of BPF with R1, R2, and PD
The average value and SD of ICV, BPV, BPF, R1, R2, and PD were calculated for each time point. In the relative change of BPFi for all measurements, i was found by dividing each of these measurements by the average <BPF>s of each subject s. This removed individual differences in BPF size. Subtraction by 1 centered the relative change of BPF around zero. The resulting data points were assumed to change linearly as a function of time after inclusion of t, expressed as an intercept and slope. A noise term ε accounts for the observed variation of the data.
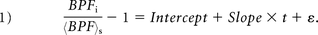
We believe that the noise term ε does not only contain random noise but may have a correlation with the observed relative change of R1, R2, and/or PD, independent of the time after inclusion. The relative change of R1, R2, and PD can be calculated in the same way as the relative change of BPF. Then, the correlation slopes SR1, SR2, and SPD were found according to
 and equivalently for R2, and PD.
and equivalently for R2, and PD.
Correction of BPF
Once the slopes SR1, SR2, and SPD are found, they can be used as correction terms for BPF to reduce ε. In other studies, it is unlikely that there is an average R1, R2, or PD per subject available; hence, the terms <R1>s, <R2>s, and <PD>s must be replaced by an expected R1exp, R2exp, and PDexp value, which can be taken from the average values of all our subjects. The corrected BPF is then calculated as the observed BPF multiplied by a factor (1–ε).
Results
Finding the Correlation of BPF with R1, R2, and PD
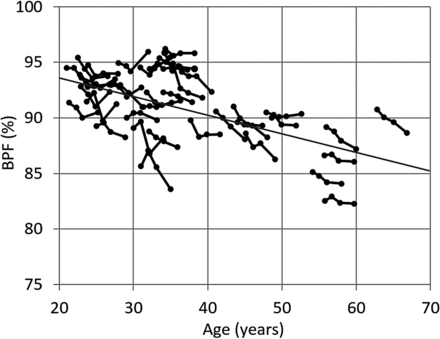
The average ICV of all subjects at all times was 1407 ± 151 mL; the average BPV was 1278 ± 129 mL, resulting in an average BPF of 90.9% ± 3.2%. The BPF of all subjects at all times is plotted in Fig 2. Linear regression on all data as a function of age showed a slope of BPF of −0.167%/year (95% confidence interval, −0.131 to −0.204; P < .001).
Brain parenchymal fraction of all subjects at all times. The 4 time points of each individual are connected with a line. The gray line indicates the linear regression slope.
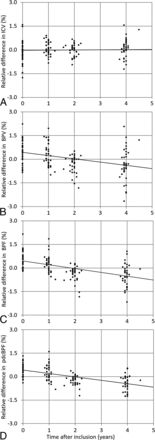
In Fig 3, the normalized ICV, BPV, and BPF are shown as a function of time after inclusion. For the separate time points, at 1, 2, and 4 years after the time of inclusion, the decrease of BPV and BPF was 2.2 mL (P = .4) and 0.37% (P < .001) after 1 year, 12.2 mL (P < .001) and 0.87% (P < .001) after 2 years, and 12.3 mL (P = .007) and 0.97% (P < .001) after 4 years. Linear regression as a function of all 4 years showed that the ICV had a nonsignificant slope of 0.02%/year (95% confidence interval, −0.03–0.08; P = .6). The BPV showed a significant decrease with time after inclusion, with a slope of −0.236%/year (95% CI, −0.372 to −0.173; P < .001). Also, the BPF showed a significant decrease with time after inclusion, with a slope of −0.273%/year (95% CI, −0.211 to −0.335; P < .001). The intercept was 0.472%.
Relative difference of intracranial volume (A), brain parenchymal volume (B), and brain parenchymal fraction (C) of all subjects as a function of time after inclusion of the study. D, The relative difference in BPF of all subjects is shown again as a function of time after inclusion is shown, but corrected for PD. This figure is comparable with C, albeit with less variation in the data. The gray lines indicate the linear regression slopes.
The relative changes in BPF were corrected for time using the observed slope of −0.273%/year and an intercept of 0.472% (Equation 2). Then, the resulting values for BPF were compared with the relative changes in the R1, R2, and PD to obtain the slopes SR1, SR2, and SPD. For SR1, a significant slope of −0.198 was observed (P < .001). For SR2, this was 0.156 (P = .04); and for SPD, a significant slope of 0.488 was observed (P < .001). Similar-but-weaker correlations were found when only GM or WM were taken into account. All data are summarized in Table 2. No correlation was found between relative BPF and time of day of the examination (P = .7).
Observed correlation slopes SR1, SR2, and SPD of the relative change in BPF as a function of relative change in R1, R2, and PD of the entire brain and gray and white matter
Correction of BPF
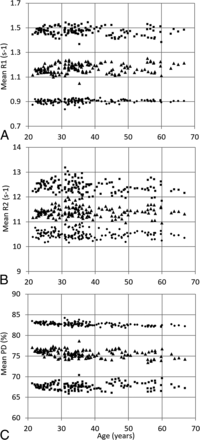
The observed mean R1, R2, and PD of the entire brain as a function of subject age are shown in Fig 4 (triangles). Only minor changes as a function of subject age were observed; linear regression showed a slope in R1 of 0.00054 seconds−1/year. For R2, this was −0.0001 seconds−1/year; and for PD, it was −0.026%/year. The R1 intercept for all subjects was 1.151± seconds−1, the R2 intercept was 11.44 seconds−1, and the PD intercept was 75.65%. All data, including for whole brain and GM and WM only, are summarized in Table 3. Using these values, one can establish the expected R1 value for the whole brain of each subject, R1exp = 1.151 + 0.00054 × age; the expected R2 value for the brain of for each subject, R2exp = 11.44 − 0.0001 × age; and the expected PD value for the brain of each subject, PDexp = 75.65 − 0.026 × age.
Observed mean R1 (A), mean R2 (B), and mean proton density (C) of the entire brain (triangles), gray matter (dots), and white matter (squares) as a function of age for all subjects.
Observed mean values and slopes of R1, R2, and PD of the entire brain and gray and white matter as a function of subject age
On the basis of these expected values for R1, R2, and PD, a correction of the BPF values can be made using Equation 2. The PD-corrected BPF values, using SPD, are shown in Fig 3D. In comparison with the uncorrected BPF values, as shown in Fig 3C, the variation of the data points was reduced. The SDs at baseline and at 1, 2, and 4 years after inclusion, when corrected for R1, R2, or PD, are given in Table 4. The highest reductions were achieved with R1 and PD, for which SDs decreased, on average, more than 20%. The baseline values especially changed substantially. If one used the correction, the observed atrophy rate decreases slightly—for example, PD-corrected BPF shows a slope of −0.238%/year, and R1-corrected BPF shows a slope of −0.256%/year, decreases of 12.8% and 6.2%, respectively, in comparison with the original slope of −0.273/year.
Observed SDs of the relative changes in BPF at baseline and at 1, 2, and 4 years after inclusiona
Discussion
In this work, brain volume measurements were combined with measures reflecting the physical properties of the brain. Generally, the variation in brain volume measurements is high because it includes the normal variation in human head size. Thus, BPF is used for clinical follow-up because it normalizes brain volume with the intracranial volume, hence removing head size differences.17 An additional factor for brain volume variations is the placement of the MR imaging acquisition volume. The acquisition coverage in our case included 43 sections, with a total of 129 mm, which was not sufficient for all subjects to cover the entire cranium. Differences of the placement of the acquisition volume may therefore have resulted in differences of the included ICV and BPV. This is a second reason that BPF is more reliable to use, because it is a ratio of the included volumes and hence less sensitive to the exact positioning of the acquisition volume. The stability of BPF over BPV is confirmed by the higher significance of the observed atrophy rate in our data.
Yet an additional factor, which is generally ignored, is that brain tissue composition may be different at each examination. Brain volume may not be as invariant as one may expect and may change with, for example, hydration, dehydration, time of day, blood pressure, physical activity, eating habits, or drug and alcohol consumption. Previous studies have shown that brain volume can exhibit a diurnal variation on the order of 0.4%–0.7%,18,19 though this could not be verified in all studies.20 In our data, no evidence of a relation with the time of day was found either (P = .7). Besides the general factors that can influence brain volume, patients with MS may exhibit additional effects. Starting treatment to suppress the inflammatory processes in the CNS is expected to reduce brain tissue edema, which hence may lead to an apparently accelerated brain atrophy directly after onset. The existence of the so-called pseudoatrophy has been described previously4,5 and is confirmed in, for example, natalizumab treatment, in which effective inflammation reduction leads to a higher initial brain volume loss, compared with the subsequent brain volume loss.21,22
For our study, we hypothesized that the average R1 and R2 and proton density of the brain were associated with water content of brain tissue, including the component due to edema. Our MR quantification approach has the advantage of simultaneously measuring R1, R2, and PD and automatically calculating brain volume and BPF using the same single sequence. We found that the average R1, R2, and PD values are relatively stable over the investigated age range. The total change of the observed values was <2 SDs during a complete lifetime (Fig 4). The mean values of WM and GM and the observed slow decrease of PD were very similar to previously reported values.23
A significant correlation was found between the relative change in observed BPF and the relative change in average R1 and PD of the brain. No correlation was found for R2, in contrast to a previous study in which only R2 (T2) was used in a study with substantial dehydration and rehydration.24 The presence of these relations provides evidence that BPF is indeed affected by changes in brain tissue composition. The correlation is stronger in WM than in GM; this finding suggests that the average value for brain tissue is mainly affected by changes in WM for patients with MS. It means that when a subject exhibits a lower BPF, it is at least partly related to a higher R1 and a lower PD. In our study, no relation with the time of day was found, suggesting that the effect of disease activity is a stronger factor than diurnal fluctuations.
The observation that the variation in measured BPF can be reduced by correction of the average PD of the entire brain or the average PD of WM suggests that the level of edema in patients with MS varies, leading both to changes in total water content of brain parenchyma and changes in brain volume. Especially, the reduction of variation in BPF at baseline indicates that correction for edema could play an important role in the interpretation of the atrophy rate after the time of inclusion. If the effect of pseudoatrophy can be reduced, there would be no need to exclude an initial period of time to avoid the observation of an exaggerated atrophy rate, as was proposed as an alternative strategy.25 In our study, 13 patients had a clinical relapse within 2 months before MR imaging at baseline, compared with only 3, 1, and 2 at follow-up MR imaging. The higher proportion of patients in clinical relapse at baseline MR imaging was, in part, because the relapse was often the cause of neurologic evaluation and inclusion in the cohort study, in part because of a considerable number of patients receiving disease-modifying MS drugs during follow-up.
Limitations in our study were the low number of included patients, the specific selection of patients, and the inevitable differences in individual patient management, leading to additional factors that were treated as random variables in the analysis. The choice of a group of early-onset patients with MS precludes any general conclusions on patients with MS or other neurodegenerative diseases. No distinction was made using the Expanded Disability Status Scale because all patients had relatively low scores (interquartile Expanded Disability Status Scale range, 1.0–2.0). Also, no control group was included to assess the normal decrease of BPF or any correlation with R1, R2, or PD with the healthy brain, precluding a reference value for normal variation. The time between measurements was at least a year, which is too long to monitor the dynamic behavior of the disease activity. Because brain volume loss is a critical parameter in MS research and clinical management, however, this work must be seen as an indicator that brain volume measurements may be confounded by other factors and that the precision of such measurements may benefit from monitoring the brain tissue characteristics as well. Our study indicates that edema correction may improve the precision of monitoring brain volume loss for patients with MS. Possibly, other neurologic, psychiatric, and geriatric conditions could also benefit from such a correction, and we intend to include larger groups to investigate further possibilities. A faster rate of probing would potentially show disease dynamics, which would be interesting to investigate, especially in the first 12 months after onset, when the effect of pseudoatrophy is expected to predominate.
Conclusions
Edema correction for brain volume using the mean R1 and R2 or proton density of the brain reduced the variation in brain volume measurement values in our cohort with up to 33% and may have reduced the effect of pseudoatrophy.
Footnotes
Disclosures: J.B. Marcel Warntjes—UNRELATED: Employment: SyntheticMR AB, Comments: part-time employee; Stock/Stock Options: SyntheticMR AB. Peter Lundberg—UNRELATED: Employment: University Hospital; Grants/Grants Pending: Conventional National Science Council funding*. *Money paid to the institution.
References
- Received July 5, 2017.
- Accepted after revision October 8, 2017.
- © 2018 by American Journal of Neuroradiology