Abstract
BACKGROUND AND PURPOSE: Presence of bone marrow edema (BME) in osteoporotic vertebral compression fractures (VCF) detected by MR imaging as selection criterion for percutaneous vertebroplasty (PV) is speculative. To clarify significance of BME in VCF, we assessed pain response after PV in patients with VCF with full BME versus patients with VCF with absent BME.
METHODS: From a cohort of patients with painful VCF selected for PV, pain response in 14 patients with absent BME in VCF was prospectively compared with pain response in 31 patients with full BME in VCF. Pain was evaluated before PV and at 1 and 3 months after PV with visual analog scores and analgesics used. Back pain in general and at treated vertebral levels was assessed.
RESULTS: Pain decrease after PV at treated levels was observed in 10 (71%) patients with absent BME in VCF at both follow-up periods and in 29 (94%) patients with full BME 1 month after PV and 30 (97%) at 3 months after PV. Differences between the groups were significant (P = .04 at 1 month; P = .01 at 3 months). Pain response was not affected by other patient or imaging characteristics. General back pain after PV was comparable in both groups after PV (P = .08 at 1 month; P = .4 at 3 months).
CONCLUSION: Pain decrease after PV in patients with VCF is more frequently observed when full BME is present. Because 71% of patients with VCF with absent BME responded favorably on pain, PV should not be withheld based on absence of BME alone.
The role of imaging procedures before percutaneous vertebroplasty (PV) in patients with symptomatic osteoporotic vertebral compression fractures (VCF) remains largely speculative. In patients with multiple VCF or in patients with discrepancy in location of pain and level of VCF, MR imaging can be helpful in selecting the symptomatic VCF. MR imaging is sensitive for the detection of bone marrow edema (BME) in VCF.1-4 Presence of BME in VCF is associated with (sub)acute fractures.1-4 Some investigators have suggested that presence of BME in VCF is predictive of a favorable response to PV.1,5,6 Most VCF demonstrate no BME on MR imaging and are not considered for PV, unless back pain in patients is immobilizing and can be related to collapsed vertebral bodies on fluoroscopically guided examination. The relation between preprocedural BME in painful VCF with pain response after PV has not been evaluated prospectively.
In the present study, we investigated the relation between BME and pain response after PV. Because we expected maximal differences to be found between patients with absent BME and those with full BME, patients with partial BME were excluded. From a cohort of 127 patients with painful VCF selected for PV, we prospectively included 31 patients with full BME in VCF on preprocedural MR imaging and 14 patients with absent BME in VCF. Relief of pain in the first trimester after PV was compared across groups.
Patients and Methods
Patients
Between March 2002 and March 2005, 127 consecutive patients underwent PV of painful osteoporotic VCF in our hospital. All patients were discussed by a multidisciplinary team consisting of endocrinologists, orthopedic surgeons, and radiologists. The inclusion criteria for VCF to be treated by PV were: (1) VCF with height loss of the vertebral body, (2) immobilizing back pain refractive to medical therapy for at least 6 weeks and related to the VCF, (3) focal tenderness on fluoroscopy-guided physical examination related to the level of the VCF, and (4) bone attenuation T scores less than −2.0 with no indication of underlying disease.
Preprocedural Imaging Protocol
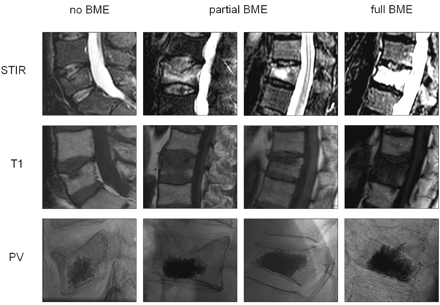
Preprocedural imaging consisted of anteroposterior and lateral radiographic examinations of the spine. When PV was considered, total spine MR imaging was performed on a 1T or 1.5T MR imaging scanner. MR imaging sequences consisted of sagittal T1-weighted (repetition time [TR], 400 ms; echo time [TE], 13 ms), T2 turbo spin echo (TSE)-weighted (TR, 3500 ms; TE, 120 ms) and short τ inversion recovery (STIR) images (TR, 2500 ms; TE, 70 ms), and transverse T2 TSE weighted images (TR, 2500 ms; TE, 120 ms) at the level of VCF with BME. All MR images had 512 matrices. Section thickness was 4 mm. Field of view in sagittal plane images was 350 mm and 225 mm in transverse images. BME in the collapsed vertebral body was defined as decreased signal intensity on T1-weighted images and increased signal intensity on STIR images.1 BME extent was assessed as a percentage of volume of the collapsed vertebral body and classified into absent (0%), partial (5%–95%), and full (100%) (Figs 1–4). Shape and grade of every treated VCF was scored by 2 radiologists in consensus using the semiquantitative visual grading scale of vertebral deformities according to Genant et al.7 Shape of VCF was classified on the basis of reduction in anterior height (wedged), middle height (biconcave), and posterior height (crush). Grade of VCF was classified as a percentage of height reduction in mild (15%–25%), moderate (26%–40%), and severe (>40%).
Lateral STIR and T1-weighted MR images of various osteoporotic vertebral compression fractures with absent, partial, and full bone marrow edema (BME) before percutaneous vertebroplasty (PV) and lateral radiographic images after PV.
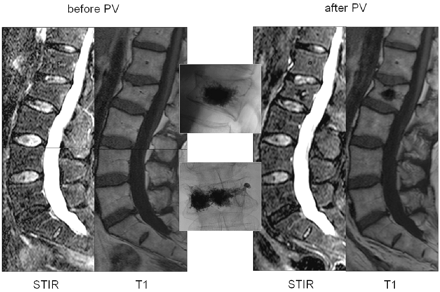
Osteoporotic vertebral compression fracture of L2 with absent bone marrow edema (BME) treated by percutaneous vertebroplasty (PV). Lateral STIR and T1-weighted MR images of the lumbar spine before and after PV. In the middle, lateral and anteroposterior radiographic images after PV.
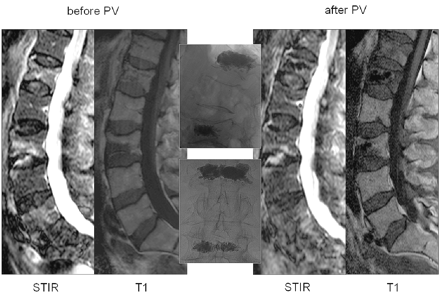
Osteoporotic vertebral compression fractures of L1 and L3 with partial bone marrow edema (BME) treated by percutaneous vertebroplasty (PV). Lateral STIR and T1-weighted MR images of the lumbar spine before and after PV. In the middle are lateral and anteroposterior radiographic images after PV.
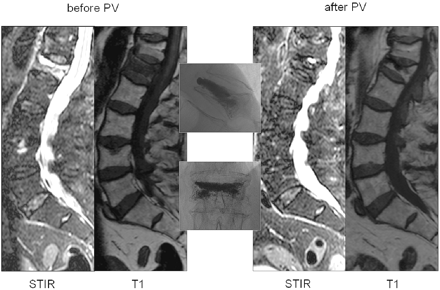
Osteoporotic vertebral compression fracture of L1 with full bone marrow edema (BME) treated by percutaneous vertebroplasty (PV). Lateral STIR and T1-weighted MR images of the lumbar spine before and after PV. In the middle, lateral and anteroposterior radiographic images after PV.
Study Population
Of 127 consecutive patients selected for PV, 45 were prospectively included in this study: 14 patients had absent BME in treated VCF (group I) and 31 patients had full BME in treated VCF (group II). The 14 patients with absent BME were treated with PV because focal tenderness related to the level of the VCF was present on physical examination and no other underlying spinal cause for their pain was evident. Before the procedure, Institutional Review Board approval and patient informed consent were obtained.
Procedure
PV was performed under local anesthesia on a biplane angiographic unit (Integris BN 3000 Neuro; Philips Medical Systems, Best, The Netherlands). In most cases, a bilateral transpedicular approach was used; in 1 case, a unilateral transpedicular approach was used. Under continuous fluoroscopic guidance, polymethylmethacrylate (PMMA) bone cement was injected manually using 1.0-mL syringes and 11 or 13 gauge bone biopsy needles (Cook Europe, Bjaeverskov, Denmark). Until June 2003, Simplex-P (Howmedica, Limmerick, Ireland; n = 3) and Palacos LV-40 (Schering-Plough Europe, Brussels, Belgium; n = 11) PMMA bone cements were used, to which 6.0 g of sterile barium sulfate was added for radio-opacity. Later, custom-made Osteopal V (Biomet Merck, Ried b. Kerzers, Switzerland; n = 16) and Osteo-Firm (Cook Europe; n = 30) were used. The amount of cement injected in each vertebral body was noted. Immediately after PV, a CT scan with multiplanar reconstructions of treated levels was made to assess cement deposition and to identify possible cement leakage or other local complications that might not have been noted under fluoroscopy.
Clinical Follow-Up
All 45 patients filled out pain questionnaires before PV and at 1 and 3 months after PV. Pain questionnaires consisted of a Visual Analog Score (VAS) for back pain and recording of analgesic use. VAS was indicated on a 10-point scale ranging from 0 (no pain) to 10 (worst pain in the patient’s life).8 Prescribed analgesic use was classified into (0) no medication, (1) use of paracetamol (acetaminophen), (2) use of nonsteroidal anti-inflammatory drugs, and (3) use of opiate derivatives. In addition, at follow-up, after PV patients were asked whether pain was located at the treated level or at another level. Three months after PV, MR imaging was performed to evaluate presence of new VCF at other levels or other spinal pathology at another level. Follow-up MR imaging consisted of sagittal T1-weighted and STIR images and transverse T2 TSE-weighted images at the level of new VCF or other spinal pathology if present.
Statistical Analysis
Characteristics of both groups were compared to evaluate possible other differences besides BME extent in treated VCF that could have influenced pain evolution after PV with χ2 (categorical variables) or unpaired t test (continuous variables). Changes in VAS at different points in time were assessed by subtraction of scores at both follow-up intervals. Differences in type of analgesic use at every follow-up period were quantified by defining analgesic use as an ordinal variable from 0 (no analgesic use) to 3 (use of opiate derivatives). Paired t test was used to compare changes in VAS and Wilcoxon paired sample test to compare type of analgesics used before and after PV. Differences in VAS score and type of analgesics used at follow-up intervals between patients from both groups were evaluated by linear regression analysis. Regression coefficients for patients with VCF with absent BME versus full BME were calculated with corresponding 95% confidence limits.9 Differences in characteristics (see Table 1) in patients with unchanged pain and in patients with relief of pain at treated levels after PV were tested with χ2 (categorical variables) or unpaired t test (continuous variables).
Characteristics of patients with absent bone marrow edema (no BME) in treated osteoporotic vertebral compression fractures (VCF) (group I) and patients with full BME (full BME) in treated VCF (group II) prior to percutaneous vertebroplasty (PV)
Results
Preprocedural Patient and Imaging Characteristics
Characteristics in patients with absent BME (group I) and with full BME (group II) in VCF are summarized in Table 1. Most patients were elderly women with wedged osteoporotic VCF of 6 months’ duration. Most patients indicated high VAS pain scores and used strong analgesics. Besides the extent of BME in treated VCF, patients in group II had on average more severe graded VCF treated by PV and had nearly 1 cc less cement injected per VCF. All other characteristics were comparable in the 2 groups. Vertebral levels treated by PV ranged from T7 through L5.
Postprocedural Clinical Outcome
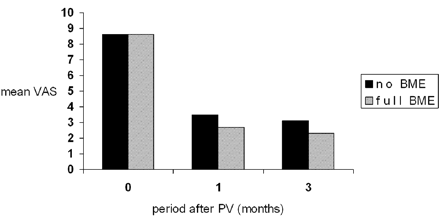
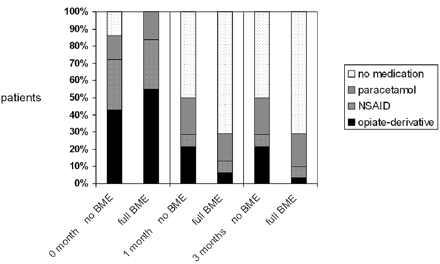
No technical failures or procedural morbidity occurred. During follow-up, pain and use of analgesics was significantly less compared with scores before treatment in both groups (P < .001) (Figs 5 and 6). Mean change in VAS for pain after PV was not different in both groups at both follow-up periods. Patients with full BME in treated VCF used fewer analgesics 1 month after PV. At 3 months after PV, analgesic use was comparable in both groups. Table 2 shows decrease in mean VAS and analgesics used during follow-up in both groups.
Visual analog score (VAS) for pain both preprocedure (0 months) and at follow-up periods after percutaneous vertebroplasty (PV) in patients with absent bone marrow edema (no BME) in treated vertebral compression fractures and patients with full BME in treated VCF.
Analgesic use both before percutaneous vertebroplasty (PV) (0 months) and at follow-up periods after PV in patients with absent bone marrow edema (no BME) in treated vertebral compression fractures (VCF) and patients with full BME in treated VCF. NSAID indicates nonsteroidal anti-inflammatory drug.
Decrease in mean visual analogue score (VAS) for pain and analgesics used at 1 and 3 months after percutaneous vertebroplasty (PV) in patients with vertebral compression fractures (VCF) with absent bone marrow edema (no BME) prior to PV and patients with full BME (full BME) in treated VCF
At both follow-up intervals, 10 patients (71%) with absent BME in treated VCF indicated no pain at treated levels or considerably less pain. Twenty-nine (94%) and 30 patients (97%) with full BME in treated VCF had good clinical response of treated levels at 1- and 3-month follow-up, respectively. The rest of the patients experienced unchanged pain and had no response to PV treatment. Some patients from both groups indicated back pain at other untreated levels. Five patients (11%), all with full BME in treated VCF, developed new VCF in untreated vertebral bodies. Of these 5 patients, 1 (2%) had immobilizing pain; the others indicated no new pain symptoms from their new VCF. One patient with absent BME and 6 patients with full BME in treated VCF had pain at untreated levels with a normal MR imaging at painful levels. Table 3 demonstrates pain outcome scores at follow-up after PV in both groups. Patients with full BME in treated VCF had significantly better relief of pain on the treated level than patients with absent BME. No difference in back pain in general after PV was found in either group.
Pain response at both follow-up intervals after percutaneous vertebroplasty (PV) in patients without bone marrow edema (BME) in treated vertebral compression fractures (VCF) prior to PV and patients with severe BME in treated VCF prior to PV
Apart from BME in treated VCF before PV, basic characteristics in patients with unchanged pain after PV (n = 6 at 1-month and n = 5 at 3-month follow-up) did not differ from patients with relief of pain. In the group of patients with absent BME in treated VCF before PV, characteristics between patients with unchanged pain after PV (n = 4) and patients with relief of pain (n = 10) were comparable.
Discussion
We found a favorable response on pain in VCF treated with PV in 94%–97% of patients with full BME and in 71% of patients with absent BME. These results indicate that presence of BME in VCF is a strong predictor of favorable response on pain relief after PV and that PV in patients with absent BME should not be withheld based on absence of BME alone. Pain response after PV was independent of other patient or imaging characteristics. Relief of back pain in general after PV treatment was comparable in patients with full and absent BME.
In the preprocedural work-up of symptomatic VCF considered for PV, MR imaging has additional diagnostic value in evaluation of soft tissue, musculoskeletal and myelum, or nerve root compromise. Moreover, the most significant aspect of MR imaging is its sensitivity to show presence, location, and extent of BME in collapsed osteoporotic vertebral bodies. Characteristic changes in marrow signal intensity in VCF supposedly indicate age and healing stage of VCF.1-4 In general, fractures less than 30 days old are hypointense on T1-weighted images and hyperintense on T2-weighted and STIR sequences. Approximately 1 month after vertebral collapse, most osteoporotic VCF become isointense to normal bone marrow on T1- and T2-weighted sequences. Fully healed VCF demonstrate either return to normal marrow signal intensity or, in the presence of sclerosis, may appear hypointense on T1- and T2-weighted sequences. Based on these observations, VCF with BME are considered (sub)acute and candidates for PV with favorable response to pain.1,5,6 On the other hand, VCF without BME chronic or healed and therefore not good candidates for PV.
Early PV of subacute VCF with presence of BME on MR imaging has given excellent clinical results regarding pain.10-12 In earlier years of PV treatment, cases of chronic symptomatic osteoporotic VCF were not treated because of poorer clinical outcomes in individual cases. However, recent studies have demonstrated good clinical outcomes in patients with chronic VCF, even though not as excellent as in patients with acute VCF.13,14 A recent study in chronic VCF of more than 1 year’s duration without BME on MR imaging demonstrated clinical benefit from PV in 80% of patients.5 Patients with absent BME in treated VCF in our prospective study also showed substantial relief of pain in 71%. Equally important was that in all studies, clinical symptoms of patients did not worsen after PV. Therefore, absence of BME in symptomatic VCF with pain related to the collapsed level on fluoroscopically guided imaging, is no absolute contraindication for successful clinical outcome after PV, but the chance of a good outcome after PV is less likely. In our small patient group with absent BME in VCF, we found no difference in characteristics in patients with pain relief and unchanged pain after PV. Characteristics of patients with symptomatic VCF with absent BME need to be evaluated in large series to select more properly patients with VCF with absent BME that would benefit from PV.
The aim of our study was to compare differences in pain response after treatment by PV of patients with absent or full BME in treated VCF, not to assess trends of pain response after PV depending on percentage of BME in treated VCF. To evaluate maximum differences in patient groups with painful VCF selected for PV on preprocedural MR imaging, we have purposely selected patients with 0% BME and 100% BME and excluded patients with partial BME (5%–95%). Because significant difference in pain response is found in patients with full BME compared with absent BME, it would be of further interest to investigate trends in pain response of patients with painful VCF with various percentages of BME ranging from absent to full BME.
In our study, patients with either full or absent BME in treated VCF had fractures of less than 6 months’ duration (median), and no significant difference between both groups existed. In all patients treated, VCF still caused severe pain, although the period of 1 to 3 months after fracture was already passed. In contradiction to previous studies on marrow changes from the acute to the chronic phase of osteoporotic VCF, we observed full BME in VCF up to 2 years after initial fracture. In addition, absence of BME in VCF was not always associated with absence of pain symptoms at the fractured level. Observations from previous studies need to be re-examined in larger populations with lasting incapacitating pain from osteoporotic VCF to determine the moment of bone marrow signal intensity change to normal. Besides, it would be interesting to examine the relation between change of BME to normal and relief of pain symptoms in the natural history of osteoporotic VCF.
Effect of pain response after PV is within the first day after treatment and remains constant thereafter. Follow-up period after PV chosen in our study was accordingly 3 months. Measurement of pain relief after PV was not evaluated with standardized back pain questionnaires. However, we believe differences in preprocedural and postprocedural VAS for pain and use of analgesics is indicative of pain outcomes after PV. In particular, changes in VAS for pain at painful treated vertebral levels gives accurate pain assessment compared with more general back pain questionnaires, in our opinion. This is illustrated by comparable outcomes in general back pain after PV in patients from both groups in our study. Commonly, elderly patients with osteoporotic VCF not only have pain symptoms caused by the VCF but also have musculoskeletal back pain caused by degenerative disease and stature changes.
As a guideline for clinical practice, we propose that patients with absent BME in painful VCF on MR imaging but with local back pain upon fluoroscopy-guided manual palpation at the level of the vertebral compression fracture should be informed about the reasonable chance of unaffected pain symptoms after PV treatment. Because a substantial number of patients with absence of BME in VCF do have benefit from the procedure, PV can be performed in selected cases with positively motivated and well informed patients.
Our study is in concordance with the observation that PV in VCF with presence of (severe) BME gives pain relief, irrespective of duration of VCF. Except for extent of preprocedural BME in VCF on MR imaging, no other characteristic was associated with pain outcome. Physical examination, particularly fluoroscopy-guided manual palpation, may help to select the symptomatic vertebral level but is not always conclusive. The role of MR imaging required before PV is important and no longer empiric in patient selection.
Conclusion
Decrease of pain after PV in patients with VCF is more frequently observed when BME is fully present. Because 71% of patients with VCF with absent BME responded favorably in regards to pain relief, PV should not be withheld based on absence of BME alone.
References
- Received September 25, 2005.
- Accepted after revision November 22, 2005.
- Copyright © American Society of Neuroradiology