Abstract
SUMMARY: We report percutaneous cryoablation of spine tumors in 7 consecutive patients (5 men, 2 women [mean age, 47 years; range, 17–68 years]) by using intraprocedural image monitoring of ice ball margins to protect adjacent neural elements. Complete tumor ablation was achieved in all patients without neurologic complication. Pain relief was achieved in 4 of 5 (80%) patients; the patient with persistent pain was later found to have enlarging metastases at other sites.
Risk of sensory and motor damage is a concern when thermally ablating tumors adjacent to major neural elements.1 Cryoablation is a promising technique for the treatment of such tumors given that ice ball margins can be visualized intraprocedurally with CT or MR imaging, allowing for better prevention of injury to adjacent structures compared with other forms of thermal ablation.2 A recent case series has demonstrated safety and efficacy of CT-guided cryoablation in spine tumors with the use of nerve monitoring and thermal protection techniques,3 and another successful case has been reported using a similar technique followed by cement augmentation and radiation therapy.4 We expand upon that literature by describing a feasible and potentially safe cryoablation method using near–real-time intraprocedural ice ball visualization, preferably with MR imaging, allowing for dynamic adjustment of cryogen gas flow to shape the ice ball to the spine tumor while preserving immediately adjacent neural structures.
Materials and Methods
We retrospectively reviewed the medical records and diagnostic imaging of 7 patients (5 men, 2 women) with a mean age of 47 years (range, 17–68 years) who underwent 9 consecutive percutaneous image-guided cryoablation procedures on spine tumors adjacent to major neural elements at our institution between January 2014 and September 2015. Patient demographics and tumor information are presented in Table 1. Because of concern for surgical morbidity, surgery was either determined to be not indicated or was refused by the patient. The study was approved by our institutional review board and performed in compliance with the Health Insurance Portability and Accountability Act.
Demographics of spine ablations in chronological order
All procedures were performed in a multimodality image-guided procedure suite, the capabilities of which have been previously described.5 Intraprocedural CT fluoroscopy (Biograph mCT 64 or Somatom Sensation 64; Siemens, Erlangen, Germany) or DynaCT (Artis zee; Siemens) was performed in the axial plane. Sagittal, coronal, and oblique reconstructions were generated from the axial dataset at the scanner whenever necessary. Intraprocedural MR imaging (3T Magnetom Verio; Siemens) monitoring was typically performed with intermittent axial T2 TSE (TR, 3000; TE, 87; section thickness, 3 mm) and sagittal T2 TSE (TR, 3000; TE, 106; section thickness, 3 mm) or axial and sagittal T2 BLADE (Siemens) (TR, 2000; TE, 113; section thickness, 4 mm) sequences during active cryoablation.
When necessary, bone access was obtained with an Arrow OnControl Power Driver drill (Teleflex, Limerick, Pennsylvnania). Cryoablation was performed using IceSphere, IceSeed, or IceRod cryoprobes with the SeedNet cryoablation system (Galil Medical, Arden Hills, Minnesota). An anesthesiologist performed general endotracheal anesthesia for all cases. Antibiotics were not administered in any case.
In general, CT or PET/CT was used to guide drilling and cryoprobe placement because they offer superior visualization of bone architecture, whereas MR imaging was favored for monitoring because it offers superior visualization of ice ball formation (Fig 1). MR imaging guidance and monitoring in patient 1 was previously reported in a review paper.6 The procedure on patient 4 was performed entirely with CT because of a large paraspinal component simultaneously ablated by our abdominal interventionalists. The entire procedure on patient 5 was performed with PET/CT (Fig 2) because of simultaneous ablation of a head tumor. Image-guidance and monitoring modalities and technical goals, either pain relief or complete tumor ablation for local disease control, are presented in Table 2.
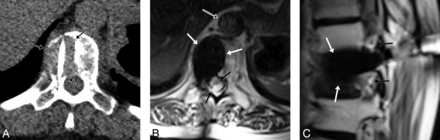
51-year-old woman with lytic T10 lung adenocarcinoma metastasis. A, Intraprocedural axial prone CT image rotated for visual convenience demonstrates destructive lesion (black arrows) extending to the thecal sac with a cryoprobe within the mass. B, Intraprocedural TSE T2-weighted image in the axial plane demonstrates ice ball (white arrows) covering the lesion with preservation of the thecal sac (black arrows) and aorta (black arrow with white border), and C, sagittal plane demonstrates the ice ball (white arrows) with preservation of the neural foramina (black arrows).
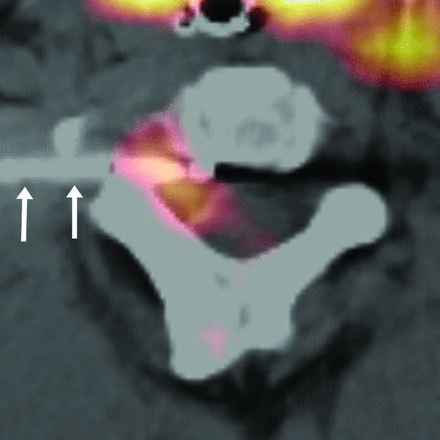
50-year-old man with PET-avid C2–C3 neural foramen adenoid cystic carcinoma metastasis largely occult on CT. Intraprocedural axial fused PET/CT image demonstrates cryoprobe (white arrows) coursing immediately posterior to the styloid process within FDG-avid lesion at the end of the second freeze; the ice ball was not well visualized. The carotid artery was immediately anterolateral to the styloid process.
Methods and results of spine ablations in chronological order
Results
A total of 9 procedures were performed in 7 patients. Outcomes are presented in Table 2. There were no temporary or permanent neurologic or functional deficits.
Two patients required 2 procedures to achieve complete ablation and pain relief. Conservative approaches were initially taken with a 17-year-old man with cervical osteoid osteoma and a 22-year-old man with lumbar osteoblastoma. Both patients had slight residual pain after ablation of the tumor nidus, and both patients opted to pursue ablation of small residual components immediately adjacent to the thecal sac and nerve root. To minimize the risk of complication when ablating the deep regions of residual tumor, preprocedure planning with advanced visualization techniques and simulation with a 3D model were performed.7 Complete cryoablation resulted in complete pain relief in both patients. Complete ablation was achieved in the other 5 patients after a single cryoablation procedure.
Four of 5 patients with preablation pain experienced complete and sustained pain relief after ablation. Patient 6, who experienced persistent pain after cryoablation, was found on 1-month postprocedure MR imaging to have complete ablation of the targeted T10 tumor, but also new tumor in T9 and T12.
Patient 5 had a left neck lymph node metastasis ablated simultaneously to the right C2–C3 neural foramen metastasis.
Follow-up imaging ranging from 1 month to 22 months postprocedure was available for 5 of the 7 patients and demonstrated no new or enlarging residual tumor at the ablation site in any patient.
Discussion
This report describes the methods and early outcomes of percutaneous image-guided cryoablation of spine tumors in 7 consecutive patients by using intraprocedural MR imaging to visualize and protect adjacent neural elements. Near–real-time monitoring of the cryogenic ice ball, particularly with MR imaging, allows for adjustment of cryogen gas flow to shape the ice ball to the tumor while preserving immediately adjacent neural structures. Such monitoring should reduce the risk of the neural damage that has been described after CT-guided radiofrequency ablation1 because the ice ball margin is not as distinct on CT. The major drawback to our approach is that an MR imaging–safe power drill bit for percutaneous procedures is not currently commercially available, and, therefore, CT guidance is necessary for cryoprobe placement. When MR imaging is not available as a monitoring technique, use of thermoprotective techniques as previously described3,8⇓⇓–11 should be considered if the ice ball is not easily visualized with CT, particularly with ice ball formation in bone.
Complete tumor ablation was achieved in all 7 of our patients, and complete pain relief was achieved in 4 of 5 (80%) patients with preprocedure pain. There were no temporary or permanent neurologic complications or functional deficits. First ablation of the central nidus of an osteoid osteoma and central nidus of an osteoblastoma resulted in partial pain relief, and complete pain relief was achieved in both patients after second ablation of small residual tumor components, consistent with prior evidence that ablation of the tumor–bone interface correlates with successful pain relief.12
The primary limitations of this study are the small number of treated patients and short-term follow-up. Larger studies and increased radiologist experience, hopefully achievable through 2 active prospective trials,13,14 will help establish the safety profile and long-term outcomes of this technique.
In summary, percutaneous cryoblation of spine tumors by using intraprocedural MR imaging monitoring is a feasible treatment option, even when tumors are adjacent to major neural elements and surgery is not a good option. Continued research is encouraged to establish this treatment as an effective and safe option for patients with nonsurgical or surgically morbid disease.
Footnotes
Disclosures: Kemal Tuncali—UNRELATED: Grants/Grants Pending: Canon USA*; Patents (planned, pending or issued): Canon USA.* *Money paid to the institution.
References
- Received April 13, 2016.
- Accepted after revision July 8, 2016.
- © 2016 by American Journal of Neuroradiology