Abstract
BACKGROUND AND PURPOSE: The occurrence of medulloblastomas in adults is rare; nevertheless, these tumors can be subdivided into genetic and histologic entities each having distinct prognoses. This study aimed to identify MR imaging biomarkers to classify these entities and to uncover differences in MR imaging biomarkers identified in pediatric medulloblastomas.
MATERIALS AND METHODS: Eligible preoperative MRIs from 28 patients (11 women; 22–53 years of age) of the Multicenter Pilot-study for the Therapy of Medulloblastoma of Adults (NOA-7) cohort were assessed by 3 experienced neuroradiologists. Lesions and perifocal edema were volumetrized and multiparametrically evaluated for classic morphologic characteristics, location, hydrocephalus, and Chang criteria. To identify MR imaging biomarkers, we correlated genetic entities sonic hedgehog (SHH) TP53 wild type, wingless (WNT), and non-WNT/non-SHH medulloblastomas (in adults, Group 4), and histologic entities were correlated with the imaging criteria. These MR imaging biomarkers were compared with corresponding data from a pediatric study.
RESULTS: There were 19 SHH TP53 wild type (69%), 4 WNT-activated (14%), and 5 Group 4 (17%) medulloblastomas. Six potential MR imaging biomarkers were identified, 3 of which, hydrocephalus (P = .03), intraventricular macrometastases (P = .02), and hemorrhage (P = .04), when combined, could identify WNT medulloblastoma with 100% sensitivity and 88.3% specificity (95% CI, 39.8%–100.0% and 62.6%–95.3%). WNT-activated nuclear β-catenin accumulating medulloblastomas were smaller than the other entities (95% CI, 5.2–22.3 cm3 versus 35.1–47.6 cm3; P = .03). Hemorrhage was exclusively present in non-WNT/non-SHH medulloblastomas (P = .04; n = 2/5). MR imaging biomarkers were all discordant from those identified in the pediatric cohort. Desmoplastic/nodular medulloblastomas were more rarely in contact with the fourth ventricle (4/15 versus 7/13; P = .04).
CONCLUSIONS: MR imaging biomarkers can help distinguish histologic and genetic medulloblastoma entities in adults and appear to be different from those identified in children.
ABBREVIATIONS:
- AUC
- area under the curve
- CE
- contrast-enhanced
- CMB
- classic medulloblastoma
- DNMB
- desmoplastic/nodular medulloblastoma
- SHH
- sonic hedgehog
- WHO
- World Health Organization
- WNT
- wingless
Medulloblastomas (World Health Organization [WHO] grade IV) rarely occur in adults. According to the United States registry analysis from the Surveillance, Epidemiology, and End-Results data base, incidence rates around 0.6 cases per million have been recorded, which is >50 times lower than the incidence of glioblastoma.1⇓–3 A higher age at diagnosis is a negative prognostic factor for survival, with a median overall survival currently between 7.7 and 9.7 years, provided patients receive the best medical care.4 The 2016 revision of the WHO classification of CNS tumors introduced the concept of an integrative medulloblastoma diagnosis.5 The diagnosis includes 4 histologically and 4 genetically defined entities known to have an influence on the course of the disease in both children and adults.6⇓⇓⇓⇓⇓–12 The genetic entities that are currently defined are sonic hedgehog (SHH)-activated (and exist with or without TP53 mutation), wingless (WNT)-activated, and non-SHH/non-WNT (Groups 3 and 4) medulloblastomas. The defined histologic groups are classic (CMB), large cell anaplastic, desmoplastic/nodular (DNMB) medulloblastomas and medulloblastoma of extensive nodularity. The exact classification at the earliest possible time point is of great importance to evaluate prognosis and possible targeted therapies.
Radiogenomics is a dynamically evolving field in radiology based on standard diagnostic MR imaging. It seeks to identify so-called MR imaging biomarkers that may predict the genetic profile of a tumor, assuming that the genetic profile is reflected in a distinctive radiophenotype, and can also be of benefit when true genetic analysis is not available.13 Only 1 study has been published in a predominantly pediatric cohort, which investigated a radiogenomic approach to differentiate genetically defined medulloblastoma entities.14 The authors found that genetic entities were distinguishable by several MR imaging biomarkers such as tumor location or enhancement pattern. Different relative frequencies and varying prognostic influences of the genetic medulloblastoma entities between adult and pediatric cohorts suggest that MR imaging biomarkers identified in pediatric cohorts may be different from those in adult medulloblastoma.8⇓–10,12,15,16
The Multicenter Pilot-study for the Therapy of Medulloblastoma of Adults (NOA-07) is the first prospective trial of an adult medulloblastoma cohort that systematically evaluated radiochemotherapy as the first-line treatment and included, among others, imaging biomarkers in its analysis. The study presented here is dedicated to identifying MR imaging biomarkers that will allow differentiation of medulloblastoma genetic entities based on the entirely adult NOA-07 cohort. Identification of such MR imaging biomarkers may facilitate presurgical tumor assessment and assist in the categorization of the differences between adult and pediatric MR imaging biomarkers.
Materials and Methods
Patient and Imaging Data
This trial was registered at ClinicalTrials.gov (NCT01614132) and under the EudraCT number 2007-002560-10 (https://www.clinicaltrialsregister.eu/ctr-search/search?query=2007-002560-10) after approval by the ethics committee of the University of Regensburg, Regensburg, Germany (08-112-0058; substantial amendment of July 1, 2016) and of all participating sites. Between 2008 and 2014, 15 neuro-oncologic centers recruited 30 adult patients older than 21 years of age with suspected medulloblastomas. Medulloblastoma diagnosis was confirmed in all cases. Presurgical CT and MR imaging datasets were retrospectively analyzed involving multiparametric imaging criteria based on T1-weighted, T2-weighted, FLAIR, and contrast-enhanced T1-weighted sequences (available in n = 28/30) as well as apparent diffusion coefficient maps (n = 23/30, On-line Figure). In 2 of 30 cases, presurgical MR imaging data were not available.
Data were compared with that in the cohort of Perreault et al,14 in which 42 of 47 cases were pediatric (Table 1).
Adult and pediatric cohorts by histology and genetic status
Imaging Criteria Definition and Analysis
Three neuroradiologists (E.H., M.W.-M., V.C.K., with 26, 28, and 6 years of experience, respectively) who were blinded to the histologic and genetic backgrounds of each patient, evaluated non-contrast-enhanced CT scans and MR imaging datasets (1.5T or 3T). Criteria were grouped as follows: 1) classic morphologic imaging characteristics applied to describe brain tumors, 2) Chang medulloblastoma staging criteria and tumor location,17 3) diagnostic criteria of hydrocephalus or brain stem compression, and 4) tumor and edema volumes and respective ratios.
Classic morphologic characteristics of brain tumors were defined as the following: enhancement pattern (strong, weak, nonenhancing), signal homogeneity (homogeneous, inhomogeneous), signal intensity (hypointense, isointense, hyperintense to gray matter) on nonenhanced T1-weighted and T2-weighted images, the sharpness of tumor margins, and the presence of cyst formation and hemorrhage. The presence of hemorrhage was diagnosed from either additional SWI or T2-weighted gradient-echo sequences or on presurgical CT.
Location criteria were the following: infiltration of the vermis, the hemispheres, or the brain stem/peduncular region, including contact with the lower rhombic lip, the eighth cranial nerve, the fourth ventricle, or the cerebellopontine angle. In addition, the presence of hydrocephalus, brain stem compression, midline shift, supratentorial growth, and multifocality was evaluated.
Medulloblastoma volumes were determined with commercially available software (tumor tracking tool, Intellispace Portal 5.0; Philips Healthcare, Best, the Netherlands) on automatically aligned contrast-enhanced T1WI (CET1WI) and T2-weighted images on the basis of manually defined signal-intensity thresholds. Perifocal edema was quantified on FLAIR datasets. Volume ratios were established as the following: 1) tumor volume ratio, which was defined as the volume fraction of the tumor part with exclusive T2 signal elevation and without the contrast-enhancing volume fraction (T2-weighted volume minus CET1WI volume divided by T2-weighted volume); and 2) edema-tumor ratio, which was defined as edema volume on FLAIR images divided by T2-weighted tumor volume.
The criteria of Chang et al17 were determined on the basis of tumor margins on both T2-weighted and CET1-weighted images. Macroscopically visible subependymal/ventricular metastases (Chang M-stage ≥2) were determined on CET1WI. Micrometastases (Chang M-stage 1) were previously investigated in the CSF at the time of diagnosis on the basis of the evaluation of 2 board-certified neuropathologists in all cases.17
Neuropathologic Evaluation
Medulloblastoma diagnoses had been confirmed at the German Brain Tumor Reference Center of the German Society for Neuropathology and Neuroanatomy by at least 2 board-certified neuropathologists. Classification was performed with histologic, immunohistochemical, and genetic analyses according to the revised WHO Classification of Tumors of the CNS 2016 as described in current Best Practice Guidelines.18 Histologically and genetically defined medulloblastoma entities were available for all 28 patients. In addition, the whole genomic copy number and allelic distribution were analyzed by molecular inversion probe methodology without evidence of amplifications in MYCC, MYCN, or GLI2 genes. One SHH-activated tumor was found to exhibit copy number losses of 9q and chromosome 14, typical for SHH-activated entities. A subgroup of samples was classified according their epigenetic subgroup by methylation arrays (450k array), which confirmed the genetically defined subgroups in all cases.19,20 The complete analysis catalogue is listed in the On-line Figure.
Imaging criteria of the comparative pediatric cohort are listed in Table 2 extracted from the earlier publication.14
Comparative overview of MR imaging biomarkers for adult and pediatric cohorts regarding genetic entity discrimination
Statistics
Statistical analyses were designed by an independent biomathematician and performed with STATA 14.0 (StataCorp, College Station, Texas). Because no tentative assumptions regarding the relationship between imaging criteria and genetic or histologic status could be made, an exploratory statistical approach to identify possible MR imaging biomarkers involved testing imaging criteria against all other criteria with 2-sided Fisher exact or ANOVA testing (depending on whether categoric or continuous data were handled). Deliberately, no multiple testing correction approach was chosen to not overlook any possible significant associations.21 Statistical significance was reached at P < .05.
Imaging criteria that were significantly associated with genetic criteria, according to the exploratory tests, were defined as potential imaging biomarkers. These were then assessed regarding their sensitivity and specificity in the differentiation of genetic entities. Evaluation involved both single and combined MR imaging biomarker applications and was based on an area under the curve (AUC) analysis.
To investigate potential differences between adult and pediatric biomarkers, we compared imaging biomarkers identified in the present study with those of a previous study involving a predominantly pediatric cohort.14
Results
Patient Cohort
The cohort of adult patients with medulloblastoma in this NOA-07 substudy included 11 women and 17 men (detailed overview in Table 1). In this cohort, no SHH-activated TP53–mutated medulloblastomas were found, all were TP53 wild type. Histologically, no cases with large cell anaplastic histology or with extensive nodularity were present. All WNT-activated and non-WNT/non-SHH medulloblastomas were histologically CMB, while 4 of 15 SHH-activated medulloblastomas were DNMB (P = .001).
Imaging Biomarkers of Genetic Entities and Chromosomal Aberrations
Six categoric and continuous volumetric imaging criteria could be identified as MR imaging biomarkers to differentiate genetically defined entities of SHH TP53 wild type, WNT-activated, and non-SHH/non-WNT Group 4 medulloblastomas in this cohort (Table 3). Notably, apart from hemorrhage, none of the classic MR imaging criteria used for tumor description, as defined in the “Materials and Methods” section, were identified as suitable MR imaging biomarkers.
Imaging biomarkers for adult medulloblastoma genetic entity differentiationa
The absence of the biomarker “hydrocephalus” was observed in 4 of 4 WNT-activated medulloblastomas. Hence, normally sized ventricular spaces as a biomarker to differentiate WNT-activated lesions showed a high negative predictive value and a sensitivity of 100% (95% CI, 39.81%–100.0%; AUC, 0.83 [95% CI, 0.67–0.99]). However, the positive predictive value of having ruled out the other entities correctly was low (33.33%; 95% CI, 22.12%–46.8%), because 7 of 19 SHH-activated and 1 of 5 non-SHH/non-WNT medulloblastomas also showed no signs of hydrocephalus.
The absence of subependymal macrometastases (Chang M ≥2) identified WNT-activated lesions with 100% sensitivity (all WNT activated cases, Chang M <2; AUC, 0.73; 95% CI, 0.52–0.94). However, the positive predictive value was again low (21.0%; 95% CI, 16.42%–26.73%).
Hemorrhage was exclusively observed in non-SHH/non-WNT medulloblastomas (n = 2/5), resulting in a positive predictive value of 100% to identify this entity correctly; however, it had a low sensitivity (40.0%; 95% CI, 5.30%–85.34%; AUC, 0.70 [95% CI, 0.40–1.0]).
The combination of all 3 of the above-mentioned biomarkers to identify WNT-activated medulloblastomas increased the specificity to 88.30% (95% CI, 62.6%–95.32%) at a sensitivity of 100% (95% CI, 39.80%–100%) and a negative predictive value of 100%, because no WNT-activated lesions showed hemorrhage, hydrocephalus, or macrometastases (AUC, 0.92; 95% CI, 0.81–1.0). The positive predictive value was 50.0% (95% CI, 29.0%–71.0%) because 3 of 19 SHH-activated and 1 of 5 non-SHH/non-WNT lesions were also negative for all 3 criteria.
Differentiation of WNT-activated medulloblastomas from SHH-activated and non-SHH/non-WNT entities combined was also possible: WNT-activated nuclear β-catenin accumulating medulloblastomas were significantly smaller on T2-weighted images (median, 11.0 versus 37.7 cm3; 95% CI, 5.2–22.3 versus 35.1–47.6 cm3; P = .03). Tumor volume on T2-weighted images was at a 5% error limit not significantly different when comparing all 3 entities (P = .06, Table 3).
The differentiation of SHH-activated and non-SHH/non-WNT Group 4 lesions was possible on the basis of edema volumes (SHH-activated medulloblatoma: median edema, 5.1 cm3; 95% CI, 3.3–8.0 cm3; Group 4 median edema, 1.2 cm3; 95% CI, 0.0–6.9 cm3; AUC, 0.85 [95% CI, 0.63–1.0; P = .02]), while tumor at the fourth ventricle (SHH-activated medulloblastoma, 3 of 19 cases; Group 4, three of 5 cases; AUC, 7.21; 95% CI, 0.39–1.0), contact with the lower rhombic lip, and the presence of hemorrhage (both biomarkers: SHH-activated medulloblastoma in 0 of 19 cases; Group 4, two of 5 cases; AUC, 0.70; 95% CI, 0.39–1.0) were less reliable and not significant (P = .14, .18, and .18, respectively). A visualization of MR imaging biomarkers for all 3 genetically defined medulloblastoma entities is presented in Fig 1.
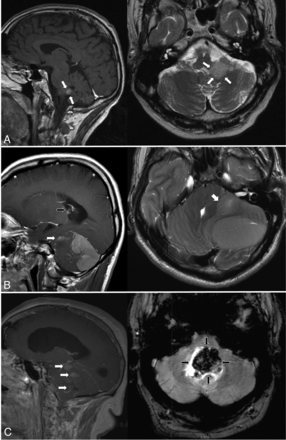
Characteristic imaging features of genetic medulloblastoma entities. Sagittal sections on the left; transverse sections on the right. A, Typical WNT-activated medulloblastoma, relatively small, in contact with the fourth ventricle (tumor margins marked with white arrows), no hydrocephalus. B, SHH-activated medulloblastoma with large tumor volume and macrometastases (sagittal image, white arrow) and large edema volume (transverse image, white arrow) and hydrocephalus (sagittal image, black arrow). C, Non-SHH/non-WNT Group 4 tumor showing macrometastases (sagittal image, white arrows), hydrocephalus, and hemorrhage (transverse image, black arrows) as distinguishing features.
No significant association could be identified for any of the imaging criteria and the chromosomal status of the tumor.
Comparison with Previously Reported MR Imaging Biomarkers for Medulloblastoma Entities in Pediatric Patients
Relative frequencies of histologically and genetically defined medulloblastoma entities were different between both cohorts: None of the MR imaging biomarkers identified in the adult cohort corresponded to those identified in the comparative predominantly pediatric cohort (Table 2). “Midline/fourth ventricle contact” was a significant biomarker to identify non-SHH/non-WNT medulloblastomas in the pediatric cohort. Midline location of the lesion was not significant in our cohort (criterion “vermis”). Contact with the fourth ventricle was analyzed separately for the present adult cohort and was determined as a biomarker with excess representation in WNT-activated medulloblastomas (Table 3).
Imaging Characteristics of Histologically Defined Entities DNMB and CMB
Perifocal edema volume was significantly larger in DNMB (n = 15) than in CMB [n = 13; median, 8.74 cm3 (95% CI, 5.71–11.73 cm3) versus 2.31 cm3 (95% CI, 1.12–5.74 cm3); P = .01; AUC 0.83 (95% CI, 0.68–0.98)]. DNMBs were less frequently in contact with the fourth ventricle than CMBs [4 of 15 versus 7 of 13; P = .04; positive predictive value to identify CMB correctly, 63.6% (95% CI, 39.7%–82.3%); AUC, 0.7 (95% CI, 0.5–0.9)].
Discussion
According to the recent revision of the WHO classification of brain tumors, the definition of genetic and histologic features of medulloblastoma is a prerequisite for a proper diagnosis of this tumor.5 In both pediatric and adult patients, genetically and histologically defined entities were determined to be relevant for prognosis and patient outcome. Therefore, precise characterization of medulloblastomas can facilitate the design of personalized therapies.14,15,20 Biomarkers based on standard MR imaging techniques have been successfully applied in the diagnosis of numerous brain lesions, including medulloblastomas.13,22⇓⇓⇓⇓–27
As part of the criteria of Chang et al,17 MR imaging morphometric analyses are well-established for medulloblastomas and are relevant in the prognosis of both adult and pediatric cohorts.28 However, entities of adult medulloblastomas seem to behave differently from the same entities in pediatric cohorts.10,12,14 To our knowledge, this is the first study to date that analyzes MR imaging biomarkers for genetic entity discrimination of exclusively adult patients with medulloblastoma in comparison with a pediatric cohort. The imaging criteria used in this study differ slightly from those presented in the study of Perreault et al.14 Because the present study was a multicenter study, sequence parameters and image quality varied among the centers; these differences make the analysis more difficult.14 In detail, the pediatric comparative cohort did not differentiate between 2 of the biomarkers in our cohort (“contact with the fourth ventricle” and “vermis/midline location”) and did not include the Chang criteria or hydrocephalus. Remarkably, no coherent MR imaging biomarkers for the same genetic entities could be identified between adult and pediatric cohorts.
The cohort of adult medulloblastomas included in this study did not contain any SHH-activated TP53 mutated cases or patients with large cell/anaplastic or extensively nodular histology, which are all common in pediatric cohorts.4 Similarly, the relative frequencies of genetically defined medulloblastoma entities in this cohort differed from those in pediatric cohorts but were concordant with other adult cohorts.9,12,14,16
As a main result of our study, WNT-activated genetic entities of adult patients with medulloblastoma could be differentiated from SHH-activated TP53 wild type and non-SHH/non-WNT Group 4 medulloblastomas by a combination of MR imaging biomarkers. Adult WNT-activated tumors were observed to be correlated to several imaging biomarkers, which might be described as their radiophenotype. At the time of tumor detection (and thus of neurologic symptoms), WNT tumors are in contact with the fourth ventricle, but without Chang M-stage ≥2 and are of small volume (lower Chang T-stages), in consequence without hydrocephalus. This finding might suggest that early midline location at the fourth ventricle induces neurologic symptoms, which allow diagnosis despite low tumor size. In contrast, most SHH-activated or non-SHH/non-WNT tumors were larger, multilocular, or focally metastatic and associated with hydrocephalus. Smaller, monofocal tumors can be more easily resected in total. This characteristic may also partially explain a frequently identified higher survival in these patients.27 Most interesting, an association of WNT-activated medulloblastomas in the lower rhombic lip could only be confirmed for a minority of tumors in this trial, despite the embryologic association of WNT-activation within the lower rhombus.
The combination of 3 of 6 identified imaging biomarkers, hemorrhage, subependymal macrometastases, and hydrocephalus, allowed an optimal discrimination of WNT-activated medulloblastomas from the other 2 genetic entities in our cohort. All MR imaging biomarkers relevant for genetic entity discrimination in this cohort were, however, different from those identified for the comparative pediatric cohort. The pediatric cohort did not differentiate between 2 of the biomarkers in our cohort (contact with the fourth ventricle and vermis/midline location). Contact with the fourth ventricle is, therefore, a pseudoconsistent biomarker between both cohorts because it was linked to the criterion midline location in the pediatric cohort, which was not identified as a biomarker in our study.
Commonly, medulloblastoma location is stratified into medial, cerebellopontine, and hemispheric lesions. However, attempts to use location criteria as biomarkers for differentiation of genetic medulloblastoma entities have yielded only inconsistent results.14,29⇓–31 Our cohort also exhibits this inconsistency, because no significant association between genetic entities and tumor location was observed. This may suggest that these location categories are not useful as biomarkers in adult medulloblastomas. A lack of association between cerebellar location and genetic entity is, to some extent, in conflict with findings that suggested that different medulloblastoma genetic entities had a spatially and cytologically distinct tumorigenesis.7,29,32
Classic morphologic tumor criteria such as signal homogeneity were not significantly associated with genetic entities in our study. This finding was strikingly different from the data of Perreault et al,14 in which sharpness of tumor margins, enhancement pattern, and location categories were highly predictive of genetic entities (though tumor margins later proved not an adequate MR imaging biomarker when tested with the validation cohort of the pediatric study). Hemorrhage was the only classic morphologic tumor criterion in our cohort that exclusively occurred in Group 4 tumors. However, hemorrhage was also reported in WNT-activated medulloblastomas.27,33 Therefore, this MR imaging biomarker should be re-evaluated in a larger independent cohort.
Considering the substantial amount of genetic and chromosomal factors investigated in this study, it remains surprising how many of these factors apparently do not result in a particular radiophenotype, especially when considering chromosomal aberrations. It can thus be postulated that the biologic impact of these factors cannot be quantified by standard MR imaging sequences. Quantitative MR imaging sequences and multifactorial computer-assisted image data analyses may, however, allow a better identification of biomarkers.34
As a limitation of our study, the comparative pediatric cohort comprised 4 adults (besides 43 children). However, the main limitation is the small size of our cohort. All findings regarding imaging biomarkers must, therefore, be taken as hypothesis-generating and should be tested for their validity in larger cohorts in the future. Different from our comparative pediatric cohort, in the discovery cohort of Perreault et al,14 no validation cohort was available to test the identified MR imaging biomarkers for adult medulloblastoma genetic entity differentiation in a blinded setting. Further research is needed to determine why biomarkers for medulloblastoma entities in pediatric patients differed from adult biomarkers identified in this study and also why classic tumor imaging criteria were usually not relevant as MR imaging biomarkers in this study. Possible reasons can be statistical effects due to a small cohort size, different methods of image assessment, but also a true lack of influence of the genetic entity on medulloblastoma radiophenotype. An international prospective trial with larger patient numbers is planned and will be an opportunity to elaborate on these open questions.
Conclusions
Several MR imaging biomarkers could be identified in this cohort of adult patients with medulloblastoma and allowed successful presurgical discrimination of genetically as well as histologically defined medulloblastoma entities. MR imaging biomarkers used in differentiation of medulloblastomas in adults seem distinct from biomarkers in children. Both findings will need to undergo further validation by radiogenomic analyses of larger medulloblastoma cohorts.
Footnotes
Elke Hattingen and Peter Hau contributed equally to this work.
Disclosures: Vera C. Keil—UNRELATED: Payment for Lectures Including Service on Speakers Bureaus: Philips Healthcare, Comments: 2015 invited talk in Switzerland, topic different from current study; Travel/Accommodations/Meeting Expenses Unrelated to Activities Listed: European Congress of Radiology, Vienna, Comments: oral presentation of study. Monika Warmuth-Metz—UNRELATED: Board Membership: Roche, Comments: adjudicator for the central review of imaging for the HERBY study (already closed); Consultancy: Italian Children's Hospital (Milano) and Russian Children's Cancer Hospital (Moscow), Comments: For the Milano hospital, I do the regular central evaluation for a diffuse intrinsic pontine glioma treatment study. For the Russian hospital, I do occasional evaluation of complicated cases of various brain tumors or brain lesions after treatment (eg, for leukemia); Expert Testimony: German Childhood Cancer Foundation (association of parents of affected children), Comments: funding for the central review of pediatric brain tumor imaging.* Torsten Pietsch—UNRELATED: Consulting Fee or Honorarium: Roche, Chugai Pharmaceutical Co, Comments: honoraria for talks; Support for Travel to Meetings for the Study or Other Purposes: Chugai Pharmaceutical Co, Affymetrix, Comments: travel support. Peter Hau—RELATED: Grant: Medac, Comments: educational grant for clinical trial, €5000 (US $5638.28)*; UNRELATED: Consultancy: Roche, Novocure, Eli Lilly and Company, Merck, Comments: consultant fees between €1000 (US $1141.65) and €2000 (US $2283.30); Payment for Lectures Including Service on Speakers Bureaus: Novocure, Comments: fees between €1000 (US $1141.65) and €2000 (US $2283.30); Travel/Accommodations/Meeting Expenses Unrelated to Activities Listed: Roche, Medac, Comments: Society for Neuro-Oncology meeting 2015 and 2017. *Money paid to the institution.
Prof. Hau received funding from Medac GmbH to conduct this study.
Paper previously presented at: Annual Meeting of the European Congress of Radiology, March 1–5, 2017 (SS711); Vienna, Austria.
References
- Received March 20, 2017.
- Accepted after revision May 24, 2017.
- © 2017 by American Journal of Neuroradiology