Abstract
BACKGROUND AND PURPOSE: The superior petrosal sinus terminates anteriorly at the cavernous sinus and posteriorly at the transverse sinus. Venous variations directly connecting the superior petrosal sinus and the emissary veins of the foramen ovale are not well-recognized. We present a connecting vein, provisionally named the petrobasal vein.
MATERIALS AND METHODS: Biplane cerebral angiography of the bilateral internal carotid arteries and the vertebral artery acquired in 267 patients was retrospectively reviewed by 2 neuroradiologists with special interest in the existence and course of the petrobasal vein.
RESULTS: The petrobasal vein was observed to lie anterior-posteriorly on the superior surface of the petrosal bone and connected to the midportion of the superior petrosal sinus and the emissary veins of the foramen ovale in 41 patients (15%) and sides (7.9%); it drained into the pterygoid plexus. The petrobasal vein was observed on VAG in 21 patients, on ICAG alone in 8 patients (9 sides), on both VAG and ICAG in 12 patients, and on ICAG in 1 patient. In the patients in whom the petrobasal vein was visualized on the ICAs, the superficial middle cerebral vein drained into a combination of the pterygoid plexus via the emissary veins of the foramen ovale and the superior petrosal sinus.
CONCLUSIONS: The petrobasal vein, an unknown vein directly connecting the superior petrosal sinus and the emissary veins of the foramen ovale and draining into the pterygoid plexus, can occasionally be identified on cerebral angiography as a variant drainage route from the cerebellum and brainstem veins and/or from the superficial middle cerebral vein. The petrobasal vein is thought to be a remnant of the primitive tentorial sinus.
ABBREVIATIONS:
- dAVF
- dural AVF
- EVFO
- emissary vein of the foramen ovale
- ICAG
- internal carotid angiography
- PBV
- petrobasal vein
- PTS
- primitive tentorial sinus
- SMCV
- superior middle cerebral vein
- SPS
- superior petrosal sinus
- VA
- vertebral artery
- VAG
- vertebral angiography
The superior petrosal sinus (SPS) is an important venous sinus that receives venous blood from the cerebellum and brainstem, and it drains posteriorly into the transverse sinus and anteriorly into the cavernous sinus. Some variations in the SPS, including the absence of either termination to the cavernous sinus or the transverse sinus, disconnection between the anterior and posterior parts of the SPS, and absence of the SPS, have been reported.1,2 The SPS occasionally receives a variant type of the superficial middle cerebral vein called the sphenopetrosal vein.3,4 The emissary vein of the foramen ovale (EVFO), a major emissary vein at the middle cranial fossa, connects medially to the cavernous sinus and laterally to the middle meningeal vein.5,6 It runs inferiorly through the foramen ovale and joins into the pterygoid plexus and/or the pharyngeal plexus. It also receives the superficial middle cerebral vein in individuals with a type of variant termination, the so-called sphenobasal vein. Blood from the SPS can drain into the cavernous sinus, and in some individuals, the SPS drains into the EVFO. There is another potential route indirectly connecting the SPS to the EVFO. The petrosquamous sinus, a rare remnant of the embryologic emissary vein, arises from the transverse sinus near the confluence with the SPS and runs anteriorly in the petrosquamous fissure or in an intraosseous canal at the most lateral part of the petrous bone to join the EVFO or the retromandibular vein through the postglenoid foramen.7,8 It is generally thought that there is no direct connection between the SPS and the EVFO. However, we observed on cerebral angiography a previously unrecognized venous channel that directly connects the SPS to the EVFO and serves as a drainage route for normal cerebral venous blood or as a potential access route to intracranial lesions, such as dural AVFs (dAVFs) (Figs 1–3); this vein is referred to as the petrobasal vein (PBV) in this article.
Schematic drawing of the potential venous pathway between the SPS and the EVFO. Two known routes include the medial route through the cavernous sinus (CS) and the lateral route through the petrosquamosal sinus (PSqS) and the middle meningeal sinus (MMS). There is another unknown route running on the superior surface of the petrosal bone connecting directly between the EVFO and the SPS (red dotted line). PV indicates petrosal vein; IPS, inferior petrosal; TS, tentorial sinus.
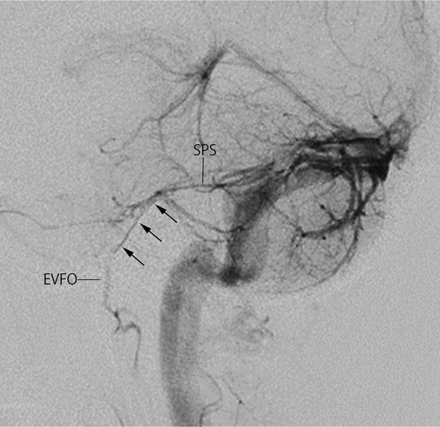
Lateral view on left vertebral angiography in the venous phase showing a venous channel (arrows) arising from the midportion of the SPS that runs anterior-inferiorly to join the EVFO. It is provisionally named the PBV.
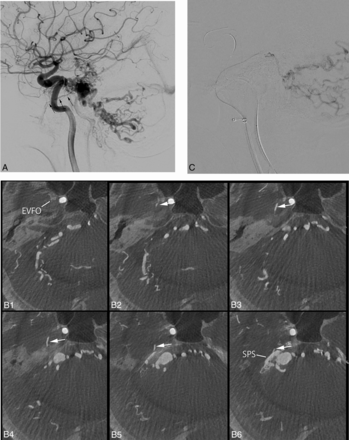
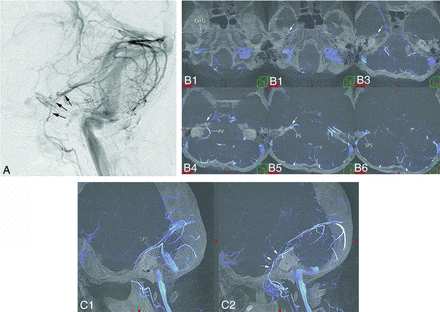
A case of a dAVF involving the SPS with a drainage route via the PBV. A, Lateral view on right internal carotid angiography shows a dAVF fed by multiple feeders from the meningohypophyseal trunk. The dAVF drains retrogradely via the various petrosal veins into the cerebellar cortical veins and the lateral mesencephalic vein. There is another drainage route via the PBV (arrows) into the emissary vein of the foramen rotundum. B, Axial MPR images based on rotational angiography of the right ICA show the dAVF involving the SPS. A drainage route (white arrow) via the PBV arising from the anterior aspect of the SPS to the EVFO is noted. C, Selective venography via a microcatheter shows transvenous introduction of the microcatheter into the petrosal vein via the PBV. Thereafter, the dAVF was completely occluded by transvenous embolization.
MATERIALS AND METHODS
This study was approved by the local ethics review board, which waived the requirement for written informed consent due to its retrospective nature. We retrospectively reviewed cerebral angiography images acquired at our institution (Oita University Hospital) from May 2015 to January 2018. After excluding patients with lesions affecting cerebral venous drainage and/or with suboptimal angiography imaging quality mainly due to motion artifacts, 267 sets of biplane cerebral angiographic images of the bilateral internal/common carotid and vertebral arteries were retrospectively reviewed by 2 neuroradiologists with special interest in visualization and the course of the PBV. The PBV was defined as a venous structure directly connecting the midportion of the SPS and the EVFO.
3D rotational angiography in the venous phase, CT angiography, and 3D T1 contrast-enhanced MR images were also reviewed when available. 3D rotational angiography was performed for selected patients who had brain tumors to depict detailed anatomic relationships of the tumors with adjacent venous structures before an operation, who had spontaneous hemorrhage with an unknown origin, and who were suspected of having venous abnormalities, including developmental venous anomalies. The patients included 111 men and 156 women, and the average age of the patients was 57.9 years. The included patients had various underlying diseases; there were 91 patients with brain or head and neck tumors, 80 patients with cerebral aneurysms, 54 patients with cerebral artery stenosis, 17 patients with dAVFs, 12 patients with cerebral arteriovenous malformations, 7 patients with spontaneous subarachnoid or cerebral hemorrhage, and 6 patients with other diseases. All angiographic procedures, including biplane DSA and 3D rotational angiography, were performed using biplane angiography equipment (Infinix VB; Toshiba Medical). For biplane angiography, the injection volume and rate in the ICA, common carotid artery, and vertebral artery (VA) were 8–9 mL at 4–4.5 mL/s, 9–10 mL at 4.5–5 mL/s, and 6–7 mL at 3–3.5 mL/s, respectively.
For 3D rotational angiography, the injection volume and rate were 18–24 mL at 3–4 mL/s in the ICA and 18–21 mL at 3-3.5 mL/s in the VA. The raw 3D rotational angiography data were transferred to a workstation (Ziostation; Ziosoft), and 3D and multiplanar reconstruction images consisting of 0.3- to 1-mm-thick sections obtained at a 0.5-mm interval were reconstructed.
RESULTS
Visualization of the PBV
The PBV was identified on 42 sides in 41 patients (7.9% and 15%) and was identified on vertebral angiography alone in 21 patients, on carotid angiography alone in 8 patients (9 sides), and on both vertebral and internal carotid angiography in 12 patients. The PBV originated at the anterior aspect of the midportion of the SPS and ran anteriorly to join the EVFO when it was visualized on vertebral angiography (Fig 4). In patients in whom the PBV was visualized on carotid angiography, the superior middle cerebral vein (SMCV) showed a combined type of sphenobasal and sphenopetrosal vein that drained into the 2 terminations of the EVFO and the SPS; additionally, the PBV formed part of the SMCV between the EVFO and the SPS (Fig 5). Among 21 patients in whom the PBV was visualized on vertebral angiography alone, the SMCV terminated in the cavernous sinus on 10 sides, in the EVFO in 6 patients, and in the transverse sinus in 1 patent, and the SMCV was absent in 2 patients.
PBV visualized on vertebral angiography alone. A, Lateral view on left vertebral angiography in the venous phase shows the PBV (arrows) from the superior petrosal sinus at the confluence of the petrosal vein. The PBV runs anterior-inferiorly to join the emissary vein of the foramen ovale. Axial (B) and sagittal (C) MPR images based on rotational angiography of the right VA show the PBV (arrows) from the anterior aspect of the SPS. It runs anteriorly on the superior surface of the petrosal bone to join the EVFO.
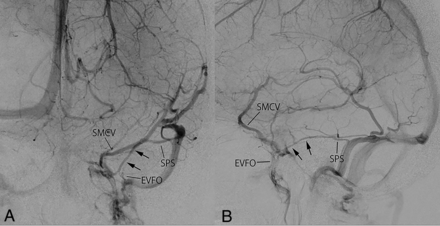
Internal carotid angiography showing the PBV as part of the drainage route of the superficial middle cerebral vein (combined type of the sphenobasal and the sphenopetrosal veins). Frontal (A) and lateral (B) views on left internal carotid angiography in the venous phase show the combined sphenobasal and sphenopetrosal drainage type of the SMCV. The PBV (arrows) composes part of the termination of the SMCV between the EVFO and the SPS. Axial (C, next page) and sagittal (D, next page) MPR images based on rotational angiography of the left ICA show the PBV (arrows) running anterior-posteriorly between the midportion of the SPS and the EVFO. The SMCV joins the EVFO.
Course of the PBV
The PBV lies anterior-posteriorly on the superior surface of the petrous bone and connects posteriorly to the anterior aspect of the SPS near the termination of the petrosal vein. Additionally, it runs inferiorly and anteromedially on the middle fossa to the foramen ovale to join the EVFO. The EVFO runs downward through the foramen ovale and joins the pterygoid plexus.
DISCUSSION
It is generally believed that no direct connection exists between the SPS and EVFO; however, in this study, we found that a connection between the SPS and the EVFO, known as the PBV, can be demonstrated in 15% of patients on cerebral angiography. The PBV has been unnoticed for a long time, probably due to its small size and the fact that it overlays other veins such as the inferior temporal vein on biplane angiography. Furthermore, the PBV runs on the surface of the petrosal bone; therefore, it can be hidden by the petrosal bone in areas with hyperdensity on CT angiography unless careful attention is paid to it.
During embryologic development, the SPS and EVFO are formed through a complicated process.8 In the early embryonic period, veins in the primitive brain initially drain by the primitive dural venous plexuses through the 3 anterior, middle, and posterior dural plexus stems into the primary head sinus. The primary head sinus also receives blood from the orbital and nasopharyngeal areas by the primitive maxillary vein and dorsal pharyngeal vein. According to the development of the brain, the anterior dural plexus stem disappears and the primary head sinus between the middle and the posterior dural plexus stem regresses due to enlargement of the trigeminal ganglion and otic vesicle; new dorsal drainage pathways develop through the transverse-sigmoid sinuses. The stem of the middle dural plexus becomes the prootic sinus. The remnant of the primary head sinus and prootic sinus forms the lateral part of the cavernous sinus and wing, the dorsal pharyngeal vein becomes the EVFO, and the emissary vein of the foramen Vesalius anastomoses with deep facial tributaries of the primitive maxillary vein.8 The EVFO also drains the middle meningeal sinus. The posterior part of the SPS develops as a drainage route of the metencephalic vein, becoming the petrosal vein. The metencephalic vein joins the distal portion of the stem of the middle dural plexus. The primitive tentorial sinus (PTS) drains into the anastomotic dural plexus between the anterior and middle dural plexuses during the same period.
A few weeks later, a venous ring surrounding the trigeminal nerve develops, and it connects the distal portion and proximal portion of the prootic sinus (Fig 6A).9 The anterior part of the middle dural plexus and inferior ramus of the peritrigeminal venous ring regresses and disappears due to further development of the trigeminal nerve and the otic vesicle. The remnant of the superior ramus of the venous ring and the residual posterior portion of the middle dural plexus stem form the primary SPS (Fig 6B). The PTS receives the superficial and deep telencephalic veins, and the ventral diencephalic vein (in the future, these become the SMCV and uncal vein) initially joins the anterior dural plexus stem and then drains into the transverse sinus after regression of the anterior dural plexus stem. The PTS deviates medially and stretches due to the development of the temporo-occipital lobes (Fig 6C). Finally, it connects and fuses to the lateral portion of the primary cavernous sinus. The midportion of the PTS fuses with the primary SPS1 and further evolves to constitute the final form of the cavernous sinus and the SPS (Fig 6D).
Schematic drawing of the embryologic development of the SPS, EVFO, and SMCV. A, Lateral view of an embryo at 18–26 mm in crown rump length. ADpxS indicates anterior dural plexus stem; PHS, primary head sinus; OV, otic vesicle; pSOV, primitive superior ophthalmic vein; pMax, primitive maxillary vein; DPV, dorsal pharyngeal vein; MDpxS, middle dural plexus; STV, superficial telencephalic vein; Met, metencephalic vein; IPS, inferior petrosal sinus; V, trigeminal nerve; VDV, ventral diencephalic vein. The dotted lines indicate regressed parts of the ADpxS and PHS. The dorsal part of the MDpxS forms the posterior part of the SPS in the future. The peritumoral venous ring surrounds the trigeminal nerve, and it connects the distal and proximal portions of the prootic sinus (proOS). B, View a few weeks after that shown in A. DTV indicates deep telencephalic vein; V3, mandibular nerve; Met, metencephalic vein; PSqS, petrosquamosal sinus; FO, foramen ovale; CS, cavernous sinus; SOV, superior ophthalmic vein. The anterior part of the MDpxS and inferior ramus of the paratrigeminal venous ring disappear, and the remnant of the superior ramus of the venous ring and the residual posterior portion of the MDpxS form the primary SPS. The DPV runs along the V3, which connects with the pterygoid venous plexus through the FO to become the EVFO. C, View just before or after birth. The PTS deviates inferomedially (red arrows) to the middle cranial fossa close to the FO secondary to further development of the temporal lobe. D, Common venous system type in adults. The PTS is further deviated medially (red arrows) and fuses to the CS, and the SMCV and uncal vein (UV) drain into the CS. The midportion of the PTS fuses to the primary SPS. A remnant of the posterior part of the PTS becomes the lateral and/or medial tentorial sinus. The metencephalic vein becomes the petrosal vein. E, The PBV combined with the sphenobasal vein. In this type, the anterior and midportions of the PTS remain at the middle cranial fossa and fuse to the EVFO. Venous blood from the SMCV drains into the EVFO and the SPS via the PBV. Venous blood from the SPS can also be drained via the PBV into the EVFO, depending on the pressure gradient. F, The PBV alone. In this type, the midportion of the PTS remains alone at the middle cranial fossa. The anterior part fuses to the CS. Venous blood from the SMCV drains into the CS. Venous blood from the SPS partially drains via the PBV into the EVFO.
There are some variations in the termination of the SMCV due to the incomplete fusion of the PTS to the cavernous sinus and the SPS. Two types of the variations are well-known. One is the sphenobasal vein in which the SMCV drains into the EVFO and the pterygoid plexus through the foramen ovale. Another is the sphenopetrosal vein, which drains into the SPS or the transverse sinus.3 Osborn4 reported that a combination of the 2 major variations was occasionally observed on cerebral angiography, in which the vein drains into both the EVFO and the SPS or the transverse sinus (Fig 6E). In our study, all patients in whom the PBV was visualized on carotid angiography demonstrated termination of the SMCV into either the EVFO or SPS; this appears to confirm the concept that the PBV is formed by incomplete fusion of the midportion of the PTS to the SPS and the cavernous sinus (Fig 6F).
Regarding the clinical relevance of the PBV, the PBV itself functions as an accessory or a main drainage route from the cerebellum and brainstem via the SPS; it also composes a part of the termination of a variant of the SMCV from the cerebral hemisphere. Furthermore, it can potentially serve as a collateral pathway of the cerebellar and brainstem venous systems in patients with sinus occlusion of the SPS or the transverse-sigmoid sinus. When a dAVF is present, the SPS and adjacent venous sinuses can serve as a drainage pathway, as shown in Fig 3. Additionally, the PBV can potentially be used as a transvenous access route to the SPS via the pterygoid plexus. Furthermore, knowledge of the detailed vascular anatomy and variations of the middle cranial fossa, including the PBV, may be important for potentially gaining access to the undersurface of the temporal lobe and the trigeminal nerve to perform endovascular neuromodulation and stimulation in the diagnosis and future endovascular treatment of epilepsy and trigeminal neuralgia.10⇓-12
Limitations
The PBV can be obscured by overlapping tributes on 2D angiography. We also reviewed 3D angiography, CT angiography, and 3D T1 contrast-enhanced MR images in addition to the 2D angiography when available. However, there were some cases evaluated by 2D angiography alone. We carefully reviewed angiographic images and eliminated the doubtful cases if possible. However, there is a potential risk of overdiagnosis of the PBV for such cases.
CONCLUSIONS
The PBV, an embryologic remnant of the PTS that is interposed between the SPS and EVFO, may occasionally be seen in the venous phase on carotid or vertebral angiography and may be considered a transvenous therapeutic access route in selected indications.
Acknowledgments
We would like to thank Drs Hisaya Hiramatsu and Mika Kamiya for providing an interesting case.
Footnotes
Disclosure forms provided by the authors are available with the full text and PDF of this article at www.ajnr.org.
Paper previously presented, in part, at: Annual Meeting of the Radiological Society of North America, November 27 to December 2, 2016; Chicago, Illinois.
References
- Received May 26, 2021.
- Accepted after revision September 14, 2021.
- © 2022 by American Journal of Neuroradiology